User:Mr. Ibrahem/Asenapine
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Saphris, Sycrest, Secuado, others |
| Other names | Asenapine maleate, ORG-5222 |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a610015 |
| License data | |
| Pregnancy category |
|
| Routes of administration | Under the tongue |
| Drug class | Atypical antipsychotic |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 35% (sublingual), <2% (by mouth)[3][4][1][5] |
| Protein binding | 95%[3][4][1][5] |
| Metabolism | hepatic (glucurinodation by UGT1A4 and oxidative metabolism by CYP1A2)[3][4][1][5] |
| Elimination half-life | 24 hours[3][4][1][5] |
| Excretion | Kidney (50%), Faecal (40%; ~5–16% as unchanged drug in faeces)[3][4][1][5] |
| Identifiers | |
| |
| Chemical and physical data | |
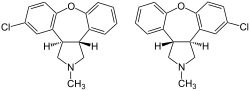
| Formula | C17H16ClNO |
| Molar mass | 285.77 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Asenapine, sold under the brand name Saphris among others, is an atypical antipsychotic used to treat schizophrenia and mania in bipolar disorder.[6][2] Use in older people with dementia related psychosis may increase the risk of death.[6] It is used under the tongue.[7]
Common side effects include sleepiness, dizziness, weight gain, movement disorders, and numbness within the mouth.[6] Other side effects may include stroke, neuroleptic malignant syndrome, tardive dyskinesia, diabetes, low white blood cells, seizures, and QT prolongation.[6] How it works is not clear, but is believed to involve blocking serotonin and dopamine receptors.[2]
Asenapine was approved for medical use in the United States in 2009 and Europe in 2010.[6][2] The first generic versions were approved in 2020.[8] In the United Kingdom it costs the NHS about £100 per month as of 2021.[9] This amount in the United States is about 200 USD.[10]
References[edit]
- ^ a b c d e f "Sycrest 5mg sublingual tablets - Summary of Product Characteristics (SmPC)". (emc). Archived from the original on 23 September 2020. Retrieved 9 September 2020.
- ^ a b c d e "Sycrest EPAR". European Medicines Agency (EMA). Archived from the original on 15 August 2020. Retrieved 9 September 2020.
- ^ a b c d e "Product Information Saphris (asenapine maleate)" (PDF). TGA eBusiness Services. Merck Sharp & Dohme (Australia) Pty Limited. 14 January 2013. Archived from the original on 2 April 2019. Retrieved 23 October 2013.
- ^ a b c d e "Saphris (asenapine maleate) tablet". DailyMed. Organon Pharmaceuticals. March 2013. Archived from the original on 3 December 2013. Retrieved 23 October 2013.
- ^ a b c d e "Product information Sycrest – EMEA/H/C/001177 –II/0012" (PDF). European Medicines Agency. N.V. Organon. 21 February 2013. Archived (PDF) from the original on 28 July 2017. Retrieved 23 October 2013.
- ^ a b c d e f g "DailyMed - SAPHRIS- asenapine maleate tablet". dailymed.nlm.nih.gov. Archived from the original on 13 November 2021. Retrieved 16 January 2022.
- ^ "Asenapine". LiverTox: Clinical and Research Information on Drug-Induced Liver Injury. National Institute of Diabetes and Digestive and Kidney Diseases. 2012. Archived from the original on 16 January 2022. Retrieved 16 January 2022.
- ^ Research, Center for Drug Evaluation and (23 February 2021). "2020 First Generic Drug Approvals". FDA. Archived from the original on 26 September 2021. Retrieved 16 January 2022.
- ^ BNF 81: March-September 2021. BMJ Group and the Pharmaceutical Press. 2021. p. 376. ISBN 978-0857114105.
- ^ "Asenapine Prices, Coupons & Savings Tips". GoodRx. Archived from the original on 23 October 2016. Retrieved 16 January 2022.
