User:Mr. Ibrahem/Ketotifen
 | |
| Clinical data | |
|---|---|
| Trade names | Zaditor,[1] Zaditen[2] |
| Other names | Ketotifen fumarate |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a604033 |
| Pregnancy category |
|
| Routes of administration | By mouth (tablets), topical eye drops |
| Drug class | H1-antihistamine, mast cell stabilizer[3] |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | 60% |
| Protein binding | 75% |
| Metabolism | Hepatic |
| Elimination half-life | 12 hours |
| Identifiers | |
| |
| Chemical and physical data | |
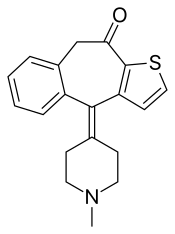
| Formula | C19H19NOS |
| Molar mass | 309.43 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
Ketotifen, sold under the brand name Zaditor among others, is a medication used to treat allergic conjunctivitis and allergic rhinitis.[2][1] It is taken by mouth or applied as an eye drop.[2][1]
Common side effects, when taken by mouth, include anxiety, trouble sleeping, and irritability.[2] Common side effects, when used as eye drops, include redness of the eyes, runny nose, and headache.[3] While use is often not recommended, there is no evidence of harm in early pregnancy.[2] It is a H1-antihistamine and mast cell stabilizer.[3]
Ketotifen was patented in 1970 and came into medical use in 1976.[4] In the United Kingdom 60 pills of 1 mg costs the NHS about £8.[2] In the United States a 5 ml bottle of eye drops costs about 7 USD.[5]
References[edit]
- ^ a b c d "Zaditor- ketotifen fumarate solution". DailyMed. 13 February 2020. Archived from the original on 11 June 2021. Retrieved 4 September 2020.
- ^ a b c d e f g h BNF 81: March-September 2021. BMJ Group and the Pharmaceutical Press. 2021. p. 302. ISBN 978-0857114105.
- ^ a b c d "Ketotifen Monograph for Professionals". Drugs.com. Archived from the original on 11 June 2021. Retrieved 1 December 2021.
- ^ Fischer, Jnos; Ganellin, C. Robin (2006). Analogue-based Drug Discovery. John Wiley & Sons. p. 548. ISBN 9783527607495. Archived from the original on 2020-08-13. Retrieved 2021-07-12.
- ^ "Compare Ketotifen Prices - GoodRx". GoodRx. Archived from the original on 2020-11-23. Retrieved 2021-12-01.
