User:Mr. Ibrahem/Lisdexamfetamine
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Tyvense, Elvanse, Vyvanse, others |
| Other names | (2S)-2,6-Diamino-N-[(2S)-1-phenylpropan-2-yl]hexanamide N-[(2S)-1-Phenyl-2-propanyl]-L-lysinamide |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a607047 |
| License data |
|
| Pregnancy category |
|
| Dependence liability | High[1][2] |
| Addiction liability | Moderate |
| Routes of administration | By mouth (capsules) |
| Drug class | Central nervous system (CNS) stimulant[1][3] |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | 96.4%[4] |
| Metabolism | Hydrolysis by enzymes in red blood cells initially. Subsequent metabolism follows Amphetamine#Pharmacokinetics. |
| Onset of action | 2 h[5][6] |
| Elimination half-life | ≤1 h (prodrug molecule) 9–11 h (dextroamphetamine) |
| Duration of action | 10–12 h[2][5][6] |
| Excretion | Renal: ~2% |
| Identifiers | |
| |
| Chemical and physical data | |
| Formula | C15H25N3O |
| Molar mass | 263.385 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Lisdexamfetamine, sold under the brand name Vyvanse among others, is a medication that is used to treat attention deficit hyperactivity disorder (ADHD) in people over the age of five as well as for moderate to severe binge eating disorder in adults.[1] Lisdexamfetamine is taken by mouth.[1][8] In the United Kingdom it is usually less preferred than methylphenidate.[9] Its effects generally begin within 2 hours and last for up to 12 hours.[1]
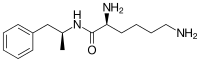
Common side effects include loss of appetite, anxiety, diarrhea, trouble sleeping, irritability, and nausea.[1] Rare but serious side effects include mania, sudden cardiac death in those with underlying heart problems, and psychosis.[1] It has a high potential for abuse per the DEA.[1][8] Serotonin syndrome may occur if used with certain other medications.[1] Its use during pregnancy may result in harm to the baby and use during breastfeeding is not recommended by the manufacturer.[9][1][8] Lisdexamfetamine is a central nervous system (CNS) stimulant that works after being converted by the body into dextroamphetamine.[1][3] Chemically, lisdexamfetamine is composed of the amino acid L-lysine, attached to dextroamphetamine.[10]
Lisdexamfetamine was approved for medical use in the United States in 2007.[1] A month's supply in the United Kingdom costs the British National Health Service about £58 as of 2019.[update][9] In the United States, the wholesale cost of this amount is about US$264.[11] In 2017, it was the 91st most commonly prescribed medication in the United States, with more than eight million prescriptions.[12][13] It is a Schedule II controlled substance in the United Kingdom and a Schedule II controlled substance in the United States.[9][14]
References[edit]
- ^ a b c d e f g h i j k l m n "Lisdexamfetamine Dimesylate Monograph for Professionals". Drugs.com. American Society of Health-System Pharmacists. Archived from the original on 8 June 2019. Retrieved 15 April 2019.
- ^ a b Stahl SM (March 2017). "Lisdexamfetamine". Prescriber's Guide: Stahl's Essential Psychopharmacology (6th ed.). Cambridge, United Kingdom: Cambridge University Press. pp. 379–384. ISBN 9781108228749.
- ^ a b Heal DJ, Smith SL, Gosden J, Nutt DJ (June 2013). "Amphetamine, past and present – a pharmacological and clinical perspective". J. Psychopharmacol. 27 (6): 479–496. doi:10.1177/0269881113482532. PMC 3666194. PMID 23539642.
- ^ "Public Assessment Report Decentralised Procedure" (PDF). MHRA. p. 14. Archived (PDF) from the original on 31 October 2018. Retrieved 23 August 2014.
- ^ a b Millichap JG (2010). "Chapter 9: Medications for ADHD". In Millichap JG (ed.). Attention Deficit Hyperactivity Disorder Handbook: A Physician's Guide to ADHD (2nd ed.). New York, USA: Springer. p. 112. ISBN 9781441913968.
Table 9.2 Dextroamphetamine formulations of stimulant medication
Dexedrine [Peak:2–3 h] [Duration:5–6 h] ...
Adderall [Peak:2–3 h] [Duration:5–7 h]
Dexedrine spansules [Peak:7–8 h] [Duration:12 h] ...
Adderall XR [Peak:7–8 h] [Duration:12 h]
Vyvanse [Peak:3–4 h] [Duration:12 h] - ^ a b Brams M, Mao AR, Doyle RL (September 2008). "Onset of efficacy of long-acting psychostimulants in pediatric attention-deficit/hyperactivity disorder". Postgrad. Med. 120 (3): 69–88. doi:10.3810/pgm.2008.09.1909. PMID 18824827.
Onset of efficacy was earliest for d-MPH-ER at 0.5 hours, followed by d, l-MPH-LA at 1 to 2 hours, MCD at 1.5 hours, d, l-MPH-OR at 1 to 2 hours, MAS-XR at 1.5 to 2 hours, MTS at 2 hours, and LDX at approximately 2 hours. ... MAS-XR, and LDX have a long duration of action at 12 hours postdose
- ^ "WHOCC - ATC/DDD Index". www.whocc.no. Archived from the original on 8 March 2021. Retrieved 9 September 2020.
- ^ a b c "Lisdexamfetamine (Vyvanse) Use During Pregnancy". Drugs.com. Archived from the original on 11 November 2020. Retrieved 16 April 2019.
- ^ a b c d British national formulary: BNF 76 (76 ed.). Pharmaceutical Press. 2018. pp. 348–349. ISBN 9780857113382.
- ^ Blick SK, Keating GM (2007). "Lisdexamfetamine". Paediatric Drugs. 9 (2): 129–135, discussion 136–138. doi:10.2165/00148581-200709020-00007. PMID 17407369.
- ^ "NADAC as of 2019-02-27". Centers for Medicare and Medicaid Services. Archived from the original on 6 March 2019. Retrieved 3 March 2019.
- ^ "The Top 300 of 2020". ClinCalc. Archived from the original on 12 February 2021. Retrieved 11 April 2020.
- ^ "Lisdexamfetamine Dimesylate - Drug Usage Statistics". ClinCalc. Archived from the original on 27 January 2021. Retrieved 11 April 2020.
- ^ Drugs of Abuse (PDF). Drug Enforcement Administration • U.S. Department of Justice. 2017. p. 22. Archived (PDF) from the original on 17 June 2021. Retrieved 16 April 2019.
