User:Mr. Ibrahem/Minocycline
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Minocin, Minomycin, Akamin, others |
| AHFS/Drugs.com | Systemic: Monograph Topical: Monograph Mouth Monograph |
| MedlinePlus | a682101 |
| License data |
|
| Pregnancy category |
|
| Routes of administration | By mouth, intravenous, topical |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 90–100%[2] |
| Protein binding | 70–75%[3] |
| Metabolism | Liver[3] |
| Elimination half-life | 14–22[3] (11–26[2]) hours |
| Excretion | Mostly fecal, 10–15% renal[3] |
| Identifiers | |
| |
| Chemical and physical data | |
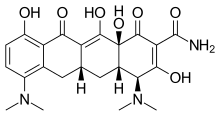
| Formula | C23H27N3O7 |
| Molar mass | 457.483 g·mol−1 |
| 3D model (JSmol) | |
| Specific rotation | = −166°[3] |
| Solubility in water | Low |
| |
| |
| | |
Minocycline, sold under the brand name Minocin among others, is a tetracycline antibiotic used to treat a number of bacterial infections such as pneumonia.[2][6] It is generally less preferred than the tetracycline doxycycline.[2][6] It is also used for the treatment of acne and rheumatoid arthritis.[6][7] It is taken by mouth or applied to the skin.[2][7]
Common side effects include nausea, diarrhea, dizziness, allergic reactions, and kidney problems.[2] Serious side effects may include anaphylaxis, a lupus-like syndrome, and easy sunburning.[2] Use in the later part of pregnancy may harm the baby and safety during breastfeeding is unclear.[8] It works by decreasing a bacterium's ability to make protein thus stopping its growth.[2]
Minocycline was patented in 1961 and came into commercial use in 1971.[9] It is available as a generic medication.[6] A month supply in the United Kingdom costs the NHS about £14 as of 2019.[6] In the United States the wholesale cost of this amount is about US$12.[10] In 2017, it was the 237th most commonly prescribed medication in the United States, with more than two million prescriptions.[11][12]
References[edit]
- ^ a b "Minocycline Use During Pregnancy". Drugs.com. 4 December 2018. Archived from the original on 29 November 2020. Retrieved 16 May 2020.
- ^ a b c d e f g h "Minocycline Hydrochloride Monograph for Professionals". Drugs.com. American Society of Health-System Pharmacists. Archived from the original on 7 January 2019. Retrieved 23 March 2019.
- ^ a b c d e Dinnendahl, V; Fricke, U, eds. (2010). Arzneistoff-Profile (in German). Vol. 7 (24 ed.). Eschborn, Germany: Govi Pharmazeutischer Verlag. Minocyclin. ISBN 978-3-7741-9846-3.
- ^ "DrugBank: DB01017 (Minocycline)". Archived from the original on 2007-01-01. Retrieved 2007-12-17.
- ^ "WHOCC - ATC/DDD Index". www.whocc.no. Archived from the original on 4 August 2020. Retrieved 9 September 2020.
- ^ a b c d e British national formulary : BNF 76 (76 ed.). Pharmaceutical Press. 2018. p. 556. ISBN 9780857113382.
- ^ a b "Minocycline topical foam" (PDF). FDA. Archived (PDF) from the original on 22 February 2020. Retrieved 23 February 2020.
- ^ "Minocycline Use During Pregnancy". Drugs.com. Archived from the original on 29 November 2020. Retrieved 3 March 2019.
- ^ Fischer, Janos; Ganellin, C. Robin (2006). Analogue-based Drug Discovery. John Wiley & Sons. p. 489. ISBN 9783527607495. Archived from the original on 2020-08-01. Retrieved 2020-08-05.
- ^ "NADAC as of 2019-02-27". Centers for Medicare and Medicaid Services. Archived from the original on 2019-03-06. Retrieved 3 March 2019.
- ^ "The Top 300 of 2020". ClinCalc. Archived from the original on 12 February 2021. Retrieved 11 April 2020.
- ^ "Minocycline Hydrochloride - Drug Usage Statistics". ClinCalc. Archived from the original on 8 July 2020. Retrieved 11 April 2020.

![{\displaystyle [\alpha ]_{D}^{25}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/fc055b4d62c591651f8a4adbc6f6b2e9e71ce021)