ABCA1
ATP-binding cassette transporter ABCA1 (member 1 of human transporter sub-family ABCA), also known as the cholesterol efflux regulatory protein (CERP) is a protein which in humans is encoded by the ABCA1 gene.[5] This transporter is a major regulator of cellular cholesterol and phospholipid homeostasis.
Tangier disease[edit]
It was discovered that a mutation in the ABCA1 protein is responsible for causing Tangier disease by several groups in 1998. Gerd Schmitz's group in Germany[6] and Michael Hayden's group in British Columbia[7] were using standard genetics techniques and DNA from family pedigrees to locate the mutation. Richard Lawn's group at CV Therapeutics in Palo Alto, CA used cDNA microarrays, which were relatively new at the time, to assess gene expression profiles from cell lines created from normal and affected individuals.[8] They showed cell lines from patients with Tangier's disease showed differential regulation of the ABCA1 gene. Subsequent sequencing of the gene identified the mutations. This group received an award from the American Heart Association for their discovery.[9] Tangier disease has been identified in nearly 100 patients worldwide, and patients have a broad range of biochemical and clinical phenotypes as over 100 different mutations have been identified in ABCA1 resulting in the disease.[10]
Function[edit]
The membrane-associated protein encoded by this gene is a member of the superfamily of ATP-binding cassette (ABC) transporters. ABC proteins transport various molecules across extra- and intracellular membranes. ABC genes are divided into seven distinct subfamilies (ABCA, MDR/TAP, MRP, ALD, OABP, GCN20, White). This protein is a member of the ABCA subfamily. Members of the ABCA subfamily comprise the only major ABC subfamily found exclusively in multicellular eukaryotes. With cholesterol as its substrate, this protein functions as a cholesterol efflux pump in the cellular lipid removal pathway.[11][12]
While the complete 3D-structure of ABCA1 remains relatively unknown, there has been some determination of the c-terminus. The ABCA1 c-terminus contains a PDZ domain, responsible for mediating protein-protein interactions, as well as a VFVNFA motif essential for lipid efflux activity.[10]
Physiological role[edit]
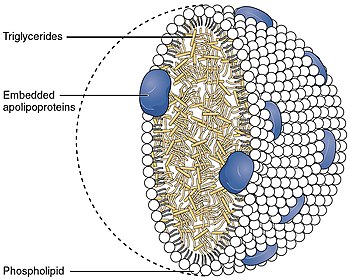
ABCA1 mediates the efflux of cholesterol and phospholipids to lipid-poor apolipoproteins (apoA1 and apoE) (reverse cholesterol transport), which then form nascent high-density lipoproteins (HDL). It also mediates the transport of lipids between Golgi and cell membrane. Since this protein is needed throughout the body it is expressed ubiquitously as a 220 kDa protein. It is present in higher quantities in tissues that shuttle or are involved in the turnover of lipids such as the liver, the small intestine and adipose tissue.[13]
Factors that act upon the ABCA1 transporter's expression or its posttranslational modification are also molecules that are involved in its subsequent function like fatty acids, cholesterol and also cytokines and cAMP.[14] Adiponectin induces reverse cholesterol transport by an ABCA1-dependent pathway.[15] Other endogenous metabolites more loosely related to the ABCA1 functions are also reported to influence the expression of this transporter, including glucose and bilirubin.[16][17]
Interactions between members of the apoliprotein family and ABCA1 activate multiple signalling pathways, including the JAK-STAT, PKA, and PKC pathways[18]
Overexpression of ABCA1 has been reported to induce resistance to the anti-inflammatory diarylheptanoid antioxidant curcumin.[19] Downregulation of ABCA1 in senescent macrophages disrupts the cell's ability to remove cholesterol from its cytoplasm, leading the cells to promote pathologic atherogenesis (blood vessel thickening/hardening) which "plays a central role in common age-associated diseases such as atherosclerosis, cancer, and macular degeneration"[20] Knockout mouse models of AMD treated with agonists that increase ABCA1 in loss of function and gain of function experiments demonstrated the protective role of elevating ABCA1 in regulating angiogenesis in eye disease. Human data from patients and controls were used to demonstrate the translation of mouse findings in human disease.[21]
Clinical significance[edit]
Mutations in this gene have been associated with Tangier disease and familial high-density lipoprotein deficiency. ABCA1 has been shown to be reduced in Tangier disease which features physiological deficiencies of HDL.[22][23] Leukocytes' ABCA1 gene expression is upregulated in postmenopausal women receiving hormone replacement therapy (HRP).[24] ABCA1 expression is also upregulated in tumor-associated astroctytes surrounding glioblastoma brain tumors, and is important to the tumor progression. [25][26]
Interactive pathway map[edit]
Click on genes, proteins and metabolites below to link to respective articles. [§ 1]
- ^ The interactive pathway map can be edited at WikiPathways: "Statin_Pathway_WP430".
Interactions[edit]
ABCA1 has been shown to interact with:
See also[edit]
References[edit]
- ^ a b c GRCh38: Ensembl release 89: ENSG00000165029 – Ensembl, May 2017
- ^ a b c GRCm38: Ensembl release 89: ENSMUSG00000015243 – Ensembl, May 2017
- ^ "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ "Mouse PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ Luciani MF, Denizot F, Savary S, Mattei MG, Chimini G (May 1994). "Cloning of two novel ABC transporters mapping on human chromosome 9". Genomics. 21 (1): 150–159. doi:10.1006/geno.1994.1237. PMID 8088782.
- ^ Bodzioch M, Orsó E, Klucken J, Langmann T, Böttcher A, Diederich W, et al. (August 1999). "The gene encoding ATP-binding cassette transporter 1 is mutated in Tangier disease". Nature Genetics. 22 (4): 347–351. doi:10.1038/11914. PMID 10431237. S2CID 26890624.
- ^ Brooks-Wilson A, Marcil M, Clee SM, Zhang LH, Roomp K, van Dam M, et al. (August 1999). "Mutations in ABC1 in Tangier disease and familial high-density lipoprotein deficiency". Nature Genetics. 22 (4): 336–345. doi:10.1038/11905. PMID 10431236. S2CID 1497231.
- ^ Lawn RM, Wade DP, Garvin MR, Wang X, Schwartz K, Porter JG, et al. (October 1999). "The Tangier disease gene product ABC1 controls the cellular apolipoprotein-mediated lipid removal pathway". The Journal of Clinical Investigation. 104 (8): R25–R31. doi:10.1172/JCI8119. PMC 481052. PMID 10525055.
- ^ "American Heart Association Selects CV Therapeutics' Discovery of Role Of 'Good' Cholesterol-Regulating Gene as Top Ten 1999 Research Advances In Heart Disease" (Press release). CV Therapeutics; Incyte Pharmaceuticals. January 3, 2000. Retrieved May 28, 2018.
- ^ a b Brunham LR, Singaraja RR, Hayden MR (2006). "Variations on a gene: rare and common variants in ABCA1 and their impact on HDL cholesterol levels and atherosclerosis". Annual Review of Nutrition. 26: 105–129. doi:10.1146/annurev.nutr.26.061505.111214. PMID 16704350.
- ^ "Entrez Gene: ABCA1 ATP-binding cassette, sub-family A (ABC1), member 1".
- ^ Schmitz G, Langmann T (April 2001). "Structure, function and regulation of the ABC1 gene product". Current Opinion in Lipidology. 12 (2): 129–140. doi:10.1097/00041433-200104000-00006. PMID 11264984. S2CID 23837673.
- ^ Wagner E, Basso F, Kim CS, Amar MJ (2014). "ABC lipid transporters". AccessScience. McGraw-Hill Education. doi:10.1036/1097-8542.801530.
- ^ Yokoyama S (February 2006). "ABCA1 and biogenesis of HDL". Journal of Atherosclerosis and Thrombosis. 13 (1): 1–15. doi:10.5551/jat.13.1. PMID 16505586.
- ^ Hafiane A, Gasbarrino K, Daskalopoulou SS (November 2019). "The role of adiponectin in cholesterol efflux and HDL biogenesis and metabolism". Metabolism. 100: 153953. doi:10.1016/j.metabol.2019.153953. PMID 31377319. S2CID 203413137.
- ^ Mauerer R, Ebert S, Langmann T (February 2009). "High glucose, unsaturated and saturated fatty acids differentially regulate expression of ATP-binding cassette transporters ABCA1 and ABCG1 in human macrophages". Experimental & Molecular Medicine. 41 (2): 126–132. doi:10.3858/emm.2009.41.2.015. PMC 2679329. PMID 19287193.
- ^ Wang D, Tosevska A, Heiß EH, Ladurner A, Mölzer C, Wallner M, et al. (April 2017). "Bilirubin Decreases Macrophage Cholesterol Efflux and ATP-Binding Cassette Transporter A1 Protein Expression". Journal of the American Heart Association. 6 (5): e005520. doi:10.1161/JAHA.117.005520. PMC 5524097. PMID 28455345.
- ^ Luu W, Sharpe LJ, Gelissen IC, Brown AJ (August 2013). "The role of signalling in cellular cholesterol homeostasis". IUBMB Life. 65 (8): 675–684. doi:10.1002/iub.1182. PMID 23847008. S2CID 23391447.
- ^ Bachmeier BE, Iancu CM, Killian PH, Kronski E, Mirisola V, Angelini G, et al. (December 2009). "Overexpression of the ATP binding cassette gene ABCA1 determines resistance to Curcumin in M14 melanoma cells". Molecular Cancer. 8: 129. doi:10.1186/1476-4598-8-129. PMC 2804606. PMID 20030852.
- ^ Sene A, Khan AA, Cox D, Nakamura RE, Santeford A, Kim BM, et al. (April 2013). "Impaired cholesterol efflux in senescent macrophages promotes age-related macular degeneration". Cell Metabolism. 17 (4): 549–561. doi:10.1016/j.cmet.2013.03.009. PMC 3640261. PMID 23562078.
- ^ http://www.faqs.org/patents/app/20130317090[full citation needed][permanent dead link]
- ^ Ordovas JM (March 2000). "ABC1: the gene for Tangier disease and beyond". Nutrition Reviews. 58 (3 Pt 1): 76–79. doi:10.1111/j.1753-4887.2000.tb01843.x. PMID 10812922.
- ^ Oram JF, Vaughan AM (June 2000). "ABCA1-mediated transport of cellular cholesterol and phospholipids to HDL apolipoproteins". Current Opinion in Lipidology. 11 (3): 253–260. doi:10.1097/00041433-200006000-00005. PMID 10882340.
- ^ Darabi M, Rabbani M, Ani M, Zarean E, Panjehpour M, Movahedian A (September 2011). "Increased leukocyte ABCA1 gene expression in post-menopausal women on hormone replacement therapy". Gynecological Endocrinology. 27 (9): 701–705. doi:10.3109/09513590.2010.507826. PMID 20807164. S2CID 203464.
- ^ Perelroizen R, Philosof B, Budick-Harmelin N, Chernobylsky T, Ron A, Katzir R, et al. (July 2022). "Astrocyte immunometabolic regulation of the tumour microenvironment drives glioblastoma pathogenicity". Brain. 145 (9): 3288–3307. doi:10.1093/brain/awac222. PMC 10233310. PMID 35899587.
- ^ Murk K, Hülse R (August 2022). "Forced but effective partners in crime: How astrocytes drive the progression of glioblastoma". Brain. 145 (9): 2952–2954. doi:10.1093/brain/awac302. PMID 35978482.
- ^ Fitzgerald ML, Morris AL, Rhee JS, Andersson LP, Mendez AJ, Freeman MW (September 2002). "Naturally occurring mutations in the largest extracellular loops of ABCA1 can disrupt its direct interaction with apolipoprotein A-I". The Journal of Biological Chemistry. 277 (36): 33178–33187. doi:10.1074/jbc.M204996200. PMID 12084722.
- ^ Buechler C, Bared SM, Aslanidis C, Ritter M, Drobnik W, Schmitz G (November 2002). "Molecular and functional interaction of the ATP-binding cassette transporter A1 with Fas-associated death domain protein". The Journal of Biological Chemistry. 277 (44): 41307–41310. doi:10.1074/jbc.C200436200. PMID 12235128.
- ^ Buechler C, Boettcher A, Bared SM, Probst MC, Schmitz G (May 2002). "The carboxyterminus of the ATP-binding cassette transporter A1 interacts with a beta2-syntrophin/utrophin complex". Biochemical and Biophysical Research Communications. 293 (2): 759–765. doi:10.1016/S0006-291X(02)00303-0. PMID 12054535.
- ^ Shimizu Y, Iwai S, Hanaoka F, Sugasawa K (January 2003). "Xeroderma pigmentosum group C protein interacts physically and functionally with thymine DNA glycosylase". The EMBO Journal. 22 (1): 164–173. doi:10.1093/emboj/cdg016. PMC 140069. PMID 12505994.
Further reading[edit]
- Tam SP, Mok L, Chimini G, Vasa M, Deeley RG (September 2006). "ABCA1 mediates high-affinity uptake of 25-hydroxycholesterol by membrane vesicles and rapid efflux of oxysterol by intact cells". American Journal of Physiology. Cell Physiology. 291 (3): C490–C502. doi:10.1152/ajpcell.00055.2006. PMID 16611739. S2CID 24019526.
- Oram JF (August 2002). "ATP-binding cassette transporter A1 and cholesterol trafficking". Current Opinion in Lipidology. 13 (4): 373–381. doi:10.1097/00041433-200208000-00004. PMID 12151852. S2CID 20345477.
- Hong SH, Rhyne J, Zeller K, Miller M (October 2002). "ABCA1(Alabama): a novel variant associated with HDL deficiency and premature coronary artery disease". Atherosclerosis. 164 (2): 245–250. doi:10.1016/S0021-9150(02)00106-5. PMID 12204794.
- Kozak M (August 2002). "Emerging links between initiation of translation and human diseases". Mammalian Genome. 13 (8): 401–410. doi:10.1007/s00335-002-4002-5. PMID 12226704. S2CID 25690586.
- Joyce C, Freeman L, Brewer HB, Santamarina-Fojo S (June 2003). "Study of ABCA1 function in transgenic mice". Arteriosclerosis, Thrombosis, and Vascular Biology. 23 (6): 965–971. doi:10.1161/01.ATV.0000055194.85073.FF. PMID 12615681.
- Singaraja RR, Brunham LR, Visscher H, Kastelein JJ, Hayden MR (August 2003). "Efflux and atherosclerosis: the clinical and biochemical impact of variations in the ABCA1 gene". Arteriosclerosis, Thrombosis, and Vascular Biology. 23 (8): 1322–1332. doi:10.1161/01.ATV.0000078520.89539.77. PMID 12763760.
- Nofer JR, Remaley AT (October 2005). "Tangier disease: still more questions than answers". Cellular and Molecular Life Sciences. 62 (19–20): 2150–2160. doi:10.1007/s00018-005-5125-0. PMID 16235041. S2CID 279676.
- Yokoyama S (February 2006). "ABCA1 and biogenesis of HDL". Journal of Atherosclerosis and Thrombosis. 13 (1): 1–15. doi:10.5551/jat.13.1. PMID 16505586.
- Schmitz G, Schambeck CM (2006). "Molecular defects in the ABCA1 pathway affect platelet function". Pathophysiology of Haemostasis and Thrombosis. 35 (1–2): 166–174. doi:10.1159/000093563. PMID 16855366. S2CID 71978568.
External links[edit]
- ABCA1 human gene location in the UCSC Genome Browser.
- ABCA1 human gene details in the UCSC Genome Browser.