ABO blood group system

The ABO blood group system is used to denote the presence of one, both, or neither of the A and B antigens on erythrocytes.[1] For human blood transfusions, it is the most important of the 44 different blood type (or group) classification systems currently recognized by the International Society of Blood Transfusions (ISBT) as of December 2022.[2][3] A mismatch (very rare in modern medicine) in this, or any other serotype, can cause a potentially fatal adverse reaction after a transfusion, or an unwanted immune response to an organ transplant.[4] The associated anti-A and anti-B antibodies are usually IgM antibodies, produced in the first years of life by sensitization to environmental substances such as food, bacteria, and viruses.
The ABO blood types were discovered by Karl Landsteiner in 1901; he received the Nobel Prize in Physiology or Medicine in 1930 for this discovery.[5] ABO blood types are also present in other primates such as apes, monkeys and Old World monkeys.[6]
History[edit]
Discovery[edit]
The ABO blood types were first discovered by an Austrian physician, Karl Landsteiner, working at the Pathological-Anatomical Institute of the University of Vienna (now Medical University of Vienna). In 1900, he found that red blood cells would clump together (agglutinate) when mixed in test tubes with sera from different persons, and that some human blood also agglutinated with animal blood.[7] He wrote a two-sentence footnote:
The serum of healthy human beings not only agglutinates animal red cells, but also often those of human origin, from other individuals. It remains to be seen whether this appearance is related to inborn differences between individuals or it is the result of some damage of bacterial kind.[8]
This was the first evidence that blood variations exist in humans – it was believed that all humans have similar blood. The next year, in 1901, he made a definitive observation that blood serum of an individual would agglutinate with only those of certain individuals. Based on this he classified human blood into three groups, namely group A, group B, and group C. He defined that group A blood agglutinates with group B, but never with its own type. Similarly, group B blood agglutinates with group A. Group C blood is different in that it agglutinates with both A and B.[9]
This was the discovery of blood groups for which Landsteiner was awarded the Nobel Prize in Physiology or Medicine in 1930. In his paper, he referred to the specific blood group interactions as isoagglutination, and also introduced the concept of agglutinins (antibodies), which is the actual basis of antigen-antibody reaction in the ABO system.[10] He asserted:
[It] may be said that there exist at least two different types of agglutinins, one in A, another one in B, and both together in C. The red blood cells are inert to the agglutinins which are present in the same serum.[9]
Thus, he discovered two antigens (agglutinogens A and B) and two antibodies (agglutinins – anti-A and anti-B). His third group (C) indicated absence of both A and B antigens, but contains anti-A and anti-B.[10] The following year, his students Adriano Sturli and Alfred von Decastello discovered the fourth type (but not naming it, and simply referred to it as "no particular type").[11][12]

In 1910, Ludwik Hirszfeld and Emil Freiherr von Dungern introduced the term 0 (null) for the group Landsteiner designated as C, and AB for the type discovered by Sturli and von Decastello. They were also the first to explain the genetic inheritance of the blood groups.[13][14]
Classification systems[edit]

Czech serologist Jan Janský independently introduced blood type classification in 1907 in a local journal.[15] He used the Roman numerical I, II, III, and IV (corresponding to modern 0, A, B, and AB). Unknown to Janský, an American physician William L. Moss devised a slightly different classification using the same numerical;[16] his I, II, III, and IV corresponding to modern AB, A, B, and 0.[12]
These two systems created confusion and potential danger in medical practice. Moss's system was adopted in Britain, France, and US, while Janský's was preferred in most European countries and some parts of US. To resolve the chaos, the American Association of Immunologists, the Society of American Bacteriologists, and the Association of Pathologists and Bacteriologists made a joint recommendation in 1921 that the Jansky classification be adopted based on priority.[17] But it was not followed particularly where Moss's system had been used.[18]
In 1927, Landsteiner had moved to the Rockefeller Institute for Medical Research in New York. As a member of a committee of the National Research Council concerned with blood grouping, he suggested to substitute Janský's and Moss's systems with the letters O, A, B, and AB. (There was another confusion on the use of figure 0 for German null as introduced by Hirszfeld and von Dungern, because others used the letter O for ohne, meaning without or zero; Landsteiner chose the latter.[18]) This classification was adopted by the National Research Council and became variously known as the National Research Council classification, the International classification, and most popularly the "new" Landsteiner classification. The new system was gradually accepted and by the early 1950s, it was universally followed.[19]
Other developments[edit]
The first practical use of blood typing in transfusion was by an American physician Reuben Ottenberg in 1907. Large-scale application began during the First World War (1914–1915) when citric acid began to be used for blood clot prevention.[10] Felix Bernstein demonstrated the correct blood group inheritance pattern of multiple alleles at one locus in 1924.[20] Watkins and Morgan, in England, discovered that the ABO epitopes were conferred by sugars, to be specific, N-acetylgalactosamine for the A-type and galactose for the B-type.[21][22][23] After much published literature claiming that the ABH substances were all attached to glycosphingolipids, Finne et al. (1978) found that the human erythrocyte glycoproteins contain polylactosamine chains[24] that contains ABH substances attached and represent the majority of the antigens.[25][26][27] The main glycoproteins carrying the ABH antigens were identified to be the Band 3 and Band 4.5 proteins and glycophorin.[28] Later, Yamamoto's group showed the precise glycosyl transferase set that confers the A, B and O epitopes.[29]
-
Diagram showing the carbohydrate chains that determine the ABO blood group
-
Student blood test. Three drops of blood are mixed with anti-B (left) and anti-A (right) serum. Agglutination with anti-A suggests this individual is type A.
-
There are three basic variants of immunoglobulin antigens in humans that share a very similar chemical structure but are distinctly different. Red circles show where there are differences in chemical structure in the antigen-binding site (sometimes called the antibody-combining site) of human immunoglobulin. Notice the O-type antigen does not have a binding site.[30]
Genetics[edit]
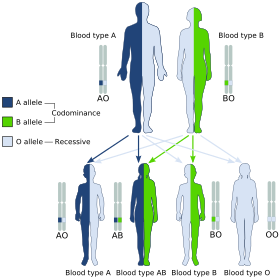

Blood groups are inherited from both parents. The ABO blood type is controlled by a single gene (the ABO gene) with three types of alleles inferred from classical genetics: i, IA, and IB. The I designation stands for isoagglutinogen, another term for antigen.[31] The gene encodes a glycosyltransferase—that is, an enzyme that modifies the carbohydrate content of the red blood cell antigens. The gene is located on the long arm of the ninth chromosome (9q34).[32]
The IA allele gives type A, IB gives type B, and i gives type O. As both IA and IB are dominant over i, only ii people have type O blood. Individuals with IAIA or IAi have type A blood, and individuals with IBIB or IBi have type B. IAIB people have both phenotypes, because A and B express a special dominance relationship: codominance, which means that type A and B parents can have an AB child. A couple with type A and type B can also have a type O child if they are both heterozygous (IBi and IAi). The cis-AB phenotype has a single enzyme that creates both A and B antigens. The resulting red blood cells do not usually express A or B antigen at the same level that would be expected on common group A1 or B red blood cells, which can help solve the problem of an apparently genetically impossible blood group.[33]
| Blood group inheritance | |||||||
| Blood type | O | A | B | AB | |||
|---|---|---|---|---|---|---|---|
| Genotype | ii (OO) | IAi (AO) | IAIA (AA) | IBi (BO) | IBIB (BB) | IAIB (AB) | |
| O | ii (OO) | O OO OO OO OO |
O or A AO OO AO OO |
A AO AO AO AO |
O or B BO OO BO OO |
B BO BO BO BO |
A or B AO BO AO BO |
| A | IAi (AO) | O or A AO AO OO OO |
O or A AA AO AO OO |
A AA AA AO AO |
O, A, B or AB AB AO BO OO |
B or AB AB AB BO BO |
A, B or AB AA AB AO BO |
| IAIA (AA) | A AO AO AO AO |
A AA AO AA AO |
A AA AA AA AA |
A or AB AB AO AB AO |
AB AB AB AB AB |
A or AB AA AB AA AB | |
| B | IBi (BO) | O or B BO BO OO OO |
O, A, B or AB AB BO AO OO |
A or AB AB AB AO AO |
O or B BB BO BO OO |
B BB BB BO BO |
A, B or AB AB BB AO BO |
| IBIB (BB) | B BO BO BO BO |
B or AB AB BO AB BO |
AB AB AB AB AB |
B BB BO BB BO |
B BB BB BB BB |
B or AB AB BB AB BB | |
| AB | IAIB (AB) | A or B AO AO BO BO |
A, B or AB AA AO AB BO |
A or AB AA AA AB AB |
A, B or AB AB AO BB BO |
B or AB AB AB BB BB |
A, B, or AB AA AB AB BB |
Individuals with the rare Bombay phenotype (hh) produce antibodies against the A, B, and O groups and can only receive transfusions from other hh individuals. The table above summarizes the various blood groups that children may inherit from their parents.[34][35] Genotypes are shown in the second column and in small print for the offspring: AO and AA both test as type A; BO and BB test as type B. The four possibilities represent the combinations obtained when one allele is taken from each parent; each has a 25% chance, but some occur more than once. The text above them summarizes the outcomes.
| Blood group inheritance by phenotype only | ||||
| Blood type | O | A | B | AB |
|---|---|---|---|---|
| O | O | O or A | O or B | A or B |
| A | O or A | O or A | O, A, B or AB | A, B or AB |
| B | O or B | O, A, B or AB | O or B | A, B or AB |
| AB | A or B | A, B or AB | A, B or AB | A, B or AB |
Historically, ABO blood tests were used in paternity testing, but in 1957 only 50% of American men falsely accused were able to use them as evidence against paternity.[36] Occasionally, the blood types of children are not consistent with expectations—for example, a type O child can be born to an AB parent—due to rare situations, such as Bombay phenotype and cis AB.[37]
Subgroups[edit]
The A blood type contains about 20 subgroups, of which A1 and A2 are the most common (over 99%). A1 makes up about 80% of all A-type blood, with A2 making up almost all of the rest.[38] These two subgroups are not always interchangeable as far as transfusion is concerned, as some A2 individuals produce antibodies against the A1 antigen. Complications can sometimes arise in rare cases when typing the blood.[38]
With the development of DNA sequencing, it has been possible to identify a much larger number of alleles at the ABO locus, each of which can be categorized as A, B, or O in terms of the reaction to transfusion, but which can be distinguished by variations in the DNA sequence. There are six common alleles in white individuals of the ABO gene that produce one's blood type:[39][40]
| A | B | O |
|---|---|---|
| A101 (A1) A201 (A2) |
B101 (B1) | O01 (O1) O02 (O1v) O03 (O2) |
The same study also identified 18 rare alleles, which generally have a weaker glycosylation activity. People with weak alleles of A can sometimes express anti-A antibodies, though these are usually not clinically significant as they do not stably interact with the antigen at body temperature.[41]
Cis AB is another rare variant, in which A and B genes are transmitted together from a single parent.[42]
Distribution and evolutionary history[edit]
The distribution of the blood groups A, B, O and AB varies across the world according to the population. There are also variations in blood type distribution within human subpopulations.[citation needed]
In the UK, the distribution of blood type frequencies through the population still shows some correlation to the distribution of placenames and to the successive invasions and migrations including Celts, Norsemen, Danes, Anglo-Saxons, and Normans who contributed the morphemes to the placenames and the genes to the population. The native Celts tended to have more type O blood, while the other populations tended to have more type A.[43]
The two common O alleles, O01 and O02, share their first 261 nucleotides with the group A allele A01.[44] However, unlike the group A allele, a guanosine base is subsequently deleted. A premature stop codon results from this frame-shift mutation. This variant is found worldwide, and likely predates human migration from Africa. The O01 allele is considered to predate the O02 allele.[citation needed]
Some evolutionary biologists theorize that there are four main lineages of the ABO gene and that mutations creating type O have occurred at least three times in humans.[45] From oldest to youngest, these lineages comprise the following alleles: A101/A201/O09, B101, O02 and O01. The continued presence of the O alleles is hypothesized to be the result of balancing selection.[45] Both theories contradict the previously held theory that type O blood evolved first.[citation needed]
Origin theories[edit]
It is possible that food and environmental antigens (bacterial, viral, or plant antigens) have epitopes similar enough to A and B glycoprotein antigens. The antibodies created against these environmental antigens in the first years of life can cross-react with ABO-incompatible red blood cells that it comes in contact with during blood transfusion later in life. Anti-A antibodies are hypothesized to originate from immune response towards influenza virus, whose epitopes are similar enough to the α-D-N-galactosamine on the A glycoprotein to be able to elicit a cross-reaction. Anti-B antibodies are hypothesized to originate from antibodies produced against Gram-negative bacteria, such as E. coli, cross-reacting with the α-D-galactose on the B glycoprotein.[46]
However, it is more likely that the force driving evolution of allele diversity is simply negative frequency-dependent selection; cells with rare variants of membrane antigens are more easily distinguished by the immune system from pathogens carrying antigens from other hosts. Thus, individuals possessing rare types are better equipped to detect pathogens. The high within-population diversity observed in human populations would, then, be a consequence of natural selection on individuals.[47]
Clinical relevance[edit]
The carbohydrate molecules on the surfaces of red blood cells have roles in cell membrane integrity, cell adhesion, membrane transportation of molecules, and acting as receptors for extracellular ligands, and enzymes. ABO antigens are found having similar roles on epithelial cells as well as red blood cells.[48][49]
Bleeding and thrombosis (von Willebrand factor)[edit]
The ABO antigen is also expressed on the von Willebrand factor (vWF) glycoprotein,[50] which participates in hemostasis (control of bleeding). In fact, having type O blood predisposes to bleeding,[51] as 30% of the total genetic variation observed in plasma vWF is explained by the effect of the ABO blood group,[52] and individuals with group O blood normally have significantly lower plasma levels of vWF (and Factor VIII) than do non-O individuals.[53][54] In addition, vWF is degraded more rapidly due to the higher prevalence of blood group O with the Cys1584 variant of vWF (an amino acid polymorphism in VWF):[55] the gene for ADAMTS13 (vWF-cleaving protease) maps to human chromosome 9 band q34.2, the same locus as ABO blood type. Higher levels of vWF are more common amongst people who have had ischemic stroke (from blood clotting) for the first time. The results of this study found that the occurrence was not affected by ADAMTS13 polymorphism, and the only significant genetic factor was the person's blood group.[56]
ABO(H) blood group antigens are also carried by other hemostatically relevant glycoproteins, such as platelet glycoprotein Ibα, which is a ligand for vWF on platelets.[57] The significance of ABO(H) antigen expression on these other hemostatic glycoproteins is not fully defined, but may also be relevant for bleeding and thrombosis.
ABO hemolytic disease of the newborn[edit]
ABO blood group incompatibilities between the mother and child do not usually cause hemolytic disease of the newborn (HDN) because antibodies to the ABO blood groups are usually of the IgM type, which do not cross the placenta. However, in an O-type mother, IgG ABO antibodies are produced and the baby can potentially develop ABO hemolytic disease of the newborn.[citation needed]
Clinical applications[edit]
In human cells, the ABO alleles and their encoded glycosyltransferases have been described in several oncologic conditions.[58] Using anti-GTA/GTB monoclonal antibodies, it was demonstrated that a loss of these enzymes was correlated to malignant bladder and oral epithelia.[59][60] Furthermore, the expression of ABO blood group antigens in normal human tissues is dependent the type of differentiation of the epithelium. In most human carcinomas, including oral carcinoma, a significant event as part of the underlying mechanism is decreased expression of the A and B antigens.[61] Several studies have observed that a relative down-regulation of GTA and GTB occurs in oral carcinomas in association with tumor development.[61][62] More recently, a genome wide association study (GWAS) has identified variants in the ABO locus associated with susceptibility to pancreatic cancer.[63] In addition, another large GWAS study has associated ABO-histo blood groups as well as FUT2 secretor status with the presence in the intestinal microbiome of specific bacterial species. In this case the association was with Bacteroides and Faecalibacterium spp. Bacteroides of the same OTU (operational taxonomic unit) have been shown to be associated with inflammatory bowel disease,[64][65] thus the study suggests an important role for the ABO histo-blood group antigens as candidates for direct modulation of the human microbiome in health and disease.[66]
Clinical marker[edit]
A multi-locus genetic risk score study based on a combination of 27 loci, including the ABO gene, identified individuals at increased risk for both incident and recurrent coronary artery disease events, as well as an enhanced clinical benefit from statin therapy. The study was based on a community cohort study (the Malmo Diet and Cancer study) and four additional randomized controlled trials of primary prevention cohorts (JUPITER and ASCOT) and secondary prevention cohorts (CARE and PROVE IT-TIMI 22).[67]
Alteration of ABO antigens for transfusion[edit]
In April 2007, an international team of researchers announced in the journal Nature Biotechnology an inexpensive and efficient way to convert types A, B, and AB blood into type O.[68] This is done by using glycosidase enzymes from specific bacteria to strip the blood group antigens from red blood cells. The removal of A and B antigens still does not address the problem of the Rh blood group antigen on the blood cells of Rh positive individuals, and so blood from Rh negative donors must be used. The modified blood is named "enzyme converted to O" (ECO blood) but despite the early success of converting B- to O-type RBCs and clinical trials without adverse effects transfusing into A- and O-type patients,[69] the technology has not yet become clinical practice.[70]
Another approach to the blood antigen problem is the manufacture of artificial blood, which could act as a substitute in emergencies.[71]
Pseudoscience[edit]
During the 1930s, connecting blood groups to personality types became popular in Japan and other areas of the world.[72] Studies of this association have yet to confirm its existence definitively.[73]
Other popular but unsupported ideas include the use of a blood type diet, claims that group A causes severe hangovers, group O is associated with perfect teeth, and those with blood group A2 have the highest IQs. Scientific evidence in support of these concepts is limited at best.[74]
See also[edit]
- Secretor status – secretion of ABO antigens in body fluids
- Omegaverse - also known as A/B/O
References[edit]
- ^ Schreiber BA, Curley R, Gaur A, Rodriguez E, Rogers K, Sinha S (18 July 2017). "ABO blood group system". Encyclopædia Britannica. Encyclopædia Britannica, Inc. Retrieved 26 October 2017.
- ^ "ISBT: Red Cell Immunogenetics and Blood Group Terminology". www.isbtweb.org. Archived from the original on 2 February 2022. Retrieved 25 April 2023.
- ^ Storry JR, Castilho L, Chen Q, Daniels G, Denomme G, Flegel WA, et al. (August 2016). "International society of blood transfusion working party on red cell immunogenetics and terminology: report of the Seoul and London meetings". ISBT Science Series. 11 (2): 118–122. doi:10.1111/voxs.12280. PMC 5662010. PMID 29093749.
- ^ Muramatsu M, Gonzalez HD, Cacciola R, Aikawa A, Yaqoob MM, Puliatti C (March 2014). "ABO incompatible renal transplants: Good or bad?". World Journal of Transplantation. 4 (1): 18–29. doi:10.5500/wjt.v4.i1.18. PMC 3964193. PMID 24669364.
- ^ Maton A, Hopkins J, McLaughlin CW, Johnson S, Warner MQ, LaHart D, Wright JD (1993). Human Biology and Health. Englewood Cliffs, New Jersey, USA: Prentice Hall. ISBN 978-0-13-981176-0.
- ^ Ségurel L, Thompson EE, Flutre T, Lovstad J, Venkat A, Margulis SW, et al. (November 2012). "The ABO blood group is a trans-species polymorphism in primates". Proceedings of the National Academy of Sciences of the United States of America. 109 (45): 18493–18498. arXiv:1208.4613. Bibcode:2012PNAS..10918493S. doi:10.1073/pnas.1210603109. PMC 3494955. PMID 23091028.
- ^ Landsteiner K (1900). "Zur Kenntnis der antifermentativen, lytischen und agglutinierenden Wirkungen des Blutserums und der Lymphe". Zentralblatt für Bakteriologie, Parasitenkunde und Infektionskrankheiten. 27: 357–362.
- ^ Kantha SS (September 1995). "The blood revolution initiated by the famous footnote of Karl Landsteiner's 1900 paper" (PDF). The Ceylon Medical Journal. 40 (3): 123–125. PMID 8536328. Archived from the original (PDF) on 30 August 2018. Retrieved 1 June 2018.
- ^ a b Landsteiner K (1961) [1901]. "On agglutination of normal human blood". Transfusion. 1 (1): 5–8. doi:10.1111/j.1537-2995.1961.tb00005.x. PMID 13758692. S2CID 40158397.Originally published in German in Wiener Klinische Wochenschrift, 46, 1132–1134
- ^ a b c Durand JK, Willis MS (2010). "Karl Landsteiner, MD: Transfusion Medicine". Laboratory Medicine. 41 (1): 53–55. doi:10.1309/LM0MICLH4GG3QNDC. ISSN 0007-5027.
- ^ Von Decastello AS, Sturli A (1902). "Concerning isoagglutinins in serum of healthy and sick humans". Munchener Medizinische Wochenschrift. 26: 1090–1095.
- ^ a b Farr AD (April 1979). "Blood group serology--the first four decades (1900--1939)". Medical History. 23 (2): 215–226. doi:10.1017/S0025727300051383. PMC 1082436. PMID 381816.
- ^ Farhud DD, Zarif Yeganeh M (2013). "A brief history of human blood groups". Iranian Journal of Public Health. 42 (1): 1–6. PMC 3595629. PMID 23514954.)
- ^ Von Dungern E, Hirschfeld L (1910). "Über Vererbung gruppenspezifischer Strukturen des Blutes". Zeitschrift für Immunitätsforschung und Experimentelle Therapie. G. Fischer.: 284–292.
Die Vererbung der durch Isoagglutinine nachweisbaren spezifischen Strukturen A und B der menschlichen Blutkfirper erfolgt nach der Mendelschen Regel, wobei die Eigenschaft der Struktur dominant ist, das Fehlen der Struktur rezessiv. Die Tatsache, dafi die nachweisbaren Bestandteile der Blutkorper niemals rezessiv sind und daher bei den Kindern nie erscheinen, wenn nicht einer der Eltern sie enthait, ist forensisch zu verwerten.
- ^ Janský J (1907). "Haematologick studie u. psychotiku". Sborn. Klinick (in Czech). 8: 85–139.
- ^ Moss WL (1910). "Studies on isoagglutinins and isohemolysins". Bulletin of the Johns Hopkins Hospital. 21: 63–70.
- ^ Doan CA (1927). "The Transfusion problem". Physiological Reviews. 7 (1): 1–84. doi:10.1152/physrev.1927.7.1.1. ISSN 0031-9333.
- ^ a b Schmidt P, Okroi M (2001). "Also sprach Landsteiner – Blood Group 'O' or Blood Group 'NULL'". Transfusion Medicine and Hemotherapy. 28 (4): 206–208. doi:10.1159/000050239. ISSN 1660-3796. S2CID 57677644.
- ^ Garratty G, Dzik W, Issitt PD, Lublin DM, Reid ME, Zelinski T (April 2000). "Terminology for blood group antigens and genes-historical origins and guidelines in the new millennium". Transfusion. 40 (4): 477–489. doi:10.1046/j.1537-2995.2000.40040477.x. PMID 10773062. S2CID 23291031.
- ^ Crow JF (January 1993). "Felix Bernstein and the first human marker locus". Genetics. 133 (1): 4–7. doi:10.1093/genetics/133.1.4. PMC 1205297. PMID 8417988.
- ^ Morgan WT, Watkins WM (January 1969). "Genetic and biochemical aspects of human blood-group A-, B-, H-, Le-a- and Le-b-specificity". British Medical Bulletin. 25 (1): 30–34. doi:10.1093/oxfordjournals.bmb.a070666. PMID 5782756.
- ^ Watkins WM (1980). "Biochemistry and Genetics of the ABO, Lewis, and P Blood Group Systems". In Harris H, Hirschhorn K (eds.). Advances in Human Genetics 10. Vol. 10. New York: Plenum. pp. 1–136. doi:10.1007/978-1-4615-8288-5_1. ISBN 9781461582885. PMID 6156588.
- ^ Watkins WM, Morgan WT (April 1959). "Possible genetical pathways for the biosynthesis of blood group mucopolysaccharides". Vox Sanguinis. 4 (2): 97–119. doi:10.1111/j.1423-0410.1959.tb04023.x. PMID 13669421. S2CID 45265348.
- ^ Finne J, Krusius T, Rauvala H, Kekomaki R, Myllyla G (May 1978). "Alkali-stable blood group A- and B-active poly(glycosyl)-peptides from human erythrocyte membrane". FEBS Letters. 89 (1): 111–115. doi:10.1016/0014-5793(78)80534-1. PMID 26599. S2CID 825499.
- ^ Krusius T, Finne J, Rauvala H (December 1978). "The poly(glycosyl) chains of glycoproteins. Characterisation of a novel type of glycoprotein saccharides from human erythrocyte membrane". European Journal of Biochemistry. 92 (1): 289–300. doi:10.1111/j.1432-1033.1978.tb12747.x. PMID 729592.
- ^ Järnefelt J, Rush J, Li YT, Laine RA (November 1978). "Erythroglycan, a high molecular weight glycopeptide with the repeating structure [galactosyl-(1 leads to 4)-2-deoxy-2-acetamido-glucosyl(1 leads to 3)] comprising more than one-third of the protein-bound carbohydrate of human erythrocyte stroma". The Journal of Biological Chemistry. 253 (22): 8006–8009. doi:10.1016/S0021-9258(17)34351-X. PMID 711733.
- ^ Laine RA, Rush JS (1988). "Chemistry of Human Erythrocyte Polylactosamine Glycopeptides (Erythroglycans) as Related to ABH Blood Group Antigenic Determinants". In Wu A, Kabat E (eds.). The Molecular Immunology of Complex Carbohydrates. Advances in Experimental Medicine and Biology. Vol. 228. Plenum Publishing Corporation. pp. 331–347. doi:10.1007/978-1-4613-1663-3_12. ISBN 9781461316633. PMID 2459929.
- ^ Finne J (February 1980). "Identification of the blood-group ABH-active glycoprotein components of human erythrocyte membrane". European Journal of Biochemistry. 104 (1): 181–189. doi:10.1111/j.1432-1033.1980.tb04414.x. PMID 6768549.
- ^ Yamamoto F, Clausen H, White T, Marken J, Hakomori S (May 1990). "Molecular genetic basis of the histo-blood group ABO system". Nature. 345 (6272): 229–233. Bibcode:1990Natur.345..229Y. doi:10.1038/345229a0. PMID 2333095. S2CID 4237562.
- ^ Hara A, Imamura A, Ando H, Ishida H, Kiso M (December 2013). "A new chemical approach to human ABO histo-blood group type 2 antigens". Molecules. 19 (1): 414–437. doi:10.3390/molecules19010414. PMC 6270767. PMID 24384923.
- ^ Klug WS, Cummings MR (1997). Concepts of Genetics (5th ed.). Upper Saddle River, NJ: Prentice Hall. p. 83. ISBN 978-0135310625.
- ^ Ferguson-Smith MA, Aitken DA, Turleau C, de Grouchy J (September 1976). "Localisation of the human ABO: Np-1: AK-1 linkage group by regional assignment of AK-1 to 9q34". Human Genetics. 34 (1): 35–43. doi:10.1007/BF00284432. PMID 184030. S2CID 12384399.
- ^ Yazer MH, Olsson ML, Palcic MM (July 2006). "The cis-AB blood group phenotype: fundamental lessons in glycobiology". Transfusion Medicine Reviews. 20 (3): 207–217. doi:10.1016/j.tmrv.2006.03.002. PMID 16787828.
- ^ "ABO inheritance patterns". Inheritance patterns of blood groups. Australian Red Cross Blood Service. Retrieved 30 October 2013.
- ^ "ABO blood group system". Abobloodtypes.webnode.com. Retrieved 2 February 2015.
- ^ Cited in "Misattributed paternity rates and non-paternity rates". Archived from the original on 26 August 2023. Retrieved 10 February 2022.
- ^ "Understanding genetics". Stanford. Archived from the original on 10 August 2020.
- ^ a b Blood Group A Suptypes, The Owen Foundation. Retrieved 1 July 2008.
- ^ Seltsam A, Hallensleben M, Kollmann A, Blasczyk R (October 2003). "The nature of diversity and diversification at the ABO locus". Blood. 102 (8): 3035–3042. doi:10.1182/blood-2003-03-0955. PMID 12829588.
- ^ Ogasawara K, Bannai M, Saitou N, Yabe R, Nakata K, Takenaka M, et al. (June 1996). "Extensive polymorphism of ABO blood group gene: three major lineages of the alleles for the common ABO phenotypes". Human Genetics. 97 (6): 777–783. doi:10.1007/BF02346189. PMID 8641696. S2CID 12076999.
- ^ Shastry S, Bhat S (October 2010). "Imbalance in A₂ and A₂B phenotype frequency of ABO group in South India". Blood Transfusion = Trasfusione del Sangue. 8 (4): 267–270. doi:10.2450/2010.0147-09. PMC 2957492. PMID 20967168.
- ^ Chun S, Choi S, Yu H, Cho D (March 2019). "Cis-AB, the Blood Group of Many Faces, Is a Conundrum to the Novice Eye". Annals of Laboratory Medicine. 39 (2): 115–120. doi:10.3343/alm.2019.39.2.115. PMC 6240514. PMID 30430772.
- ^ Potts WT (1979). "History and Blood Groups in the British Isles". In Sawyer PH (ed.). English Medieval Settlement. St. Martin's Press. ISBN 978-0-7131-6257-8.
- ^ Cserti CM, Dzik WH (October 2007). "The ABO blood group system and Plasmodium falciparum malaria". Blood. 110 (7): 2250–2258. doi:10.1182/blood-2007-03-077602. PMID 17502454.
- ^ a b Calafell F, Roubinet F, Ramírez-Soriano A, Saitou N, Bertranpetit J, Blancher A (September 2008). "Evolutionary dynamics of the human ABO gene". Human Genetics. 124 (2): 123–135. doi:10.1007/s00439-008-0530-8. PMID 18629539. S2CID 12450238.
- ^ van Oss CJ (August 2004). ""Natural" versus regular antibodies". The Protein Journal. 23 (6): 357, author reply 359-357, author reply 360. doi:10.1023/B:JOPC.0000039625.56296.6e. PMID 15517982. S2CID 189929325. Archived from the original on 14 July 2011. Retrieved 28 November 2009.
- ^ Seymour RM, Allan MJ, Pomiankowski A, Gustafsson K (May 2004). "Evolution of the human ABO polymorphism by two complementary selective pressures". Proceedings. Biological Sciences. 271 (1543): 1065–1072. doi:10.1098/rspb.2004.2674. PMC 1691687. PMID 15293861.
- ^ Reid ME, Mohandas N (April 2004). "Red blood cell blood group antigens: structure and function". Seminars in Hematology. 41 (2): 93–117. doi:10.1053/j.seminhematol.2004.01.001. PMID 15071789.
- ^ Mohandas N, Narla A (March 2005). "Blood group antigens in health and disease". Current Opinion in Hematology. 12 (2): 135–140. doi:10.1097/01.moh.0000153000.09585.79. PMID 15725904. S2CID 19866834.
- ^ Sarode R, Goldstein J, Sussman II, Nagel RL, Tsai HM (June 2000). "Role of A and B blood group antigens in the expression of adhesive activity of von Willebrand factor". British Journal of Haematology. 109 (4): 857–864. doi:10.1046/j.1365-2141.2000.02113.x. PMID 10929042. S2CID 25343413.
- ^ O'Donnell J, Laffan MA (August 2001). "The relationship between ABO histo-blood group, factor VIII and von Willebrand factor". Transfusion Medicine. 11 (4): 343–351. doi:10.1046/j.1365-3148.2001.00315.x. PMID 11532189. S2CID 23603864.
- ^ O'Donnell J, Boulton FE, Manning RA, Laffan MA (February 2002). "Amount of H antigen expressed on circulating von Willebrand factor is modified by ABO blood group genotype and is a major determinant of plasma von Willebrand factor antigen levels". Arteriosclerosis, Thrombosis, and Vascular Biology. 22 (2): 335–341. doi:10.1161/hq0202.103997. PMID 11834538. S2CID 325644.
- ^ Gill JC, Endres-Brooks J, Bauer PJ, Marks WJ, Montgomery RR (June 1987). "The effect of ABO blood group on the diagnosis of von Willebrand disease". Blood. 69 (6): 1691–1695. doi:10.1182/blood.V69.6.1691.1691. PMID 3495304.
- ^ Shima M, Fujimura Y, Nishiyama T, Tsujiuchi T, Narita N, Matsui T, et al. (1995). "ABO blood group genotype and plasma von Willebrand factor in normal individuals". Vox Sanguinis. 68 (4): 236–240. doi:10.1111/j.1423-0410.1995.tb02579.x. PMID 7660643. S2CID 11582510.
- ^ Bowen DJ, Collins PW, Lester W, Cumming AM, Keeney S, Grundy P, et al. (March 2005). "The prevalence of the cysteine1584 variant of von Willebrand factor is increased in type 1 von Willebrand disease: co-segregation with increased susceptibility to ADAMTS13 proteolysis but not clinical phenotype". British Journal of Haematology. 128 (6): 830–836. doi:10.1111/j.1365-2141.2005.05375.x. PMID 15755288. S2CID 45434815.
- ^ Bongers TN, de Maat MP, van Goor ML, Bhagwanbali V, van Vliet HH, Gómez García EB, et al. (November 2006). "High von Willebrand factor levels increase the risk of first ischemic stroke: influence of ADAMTS13, inflammation, and genetic variability". Stroke. 37 (11): 2672–2677. doi:10.1161/01.STR.0000244767.39962.f7. PMID 16990571.
- ^ Hollenhorst, Marie A.; Tiemeyer, Katherine H.; Mahoney, Keira E.; Aoki, Kazuhiro; Ishihara, Mayumi; Lowery, Sarah C.; Rangel-Angarita, Valentina; Bertozzi, Carolyn R.; Malaker, Stacy A. (April 2023). "Comprehensive analysis of platelet glycoprotein Ibα ectodomain glycosylation". Journal of Thrombosis and Haemostasis. 21 (4): 995–1009. doi:10.1016/j.jtha.2023.01.009. PMC 10065957. PMID 36740532.
- ^ Goldman M (February 2007). "Translational mini-review series on Toll-like receptors: Toll-like receptor ligands as novel pharmaceuticals for allergic disorders". Clinical and Experimental Immunology. 147 (2): 208–216. doi:10.1111/j.1365-2249.2006.03296.x. PMC 1810467. PMID 17223960.
- ^ Kay HE (November 1982). "Bone marrow transplantation: 1982". British Medical Journal. 285 (6351): 1296–1298. doi:10.1136/bmj.285.6351.1296. PMC 1500229. PMID 6812684.
- ^ Hakomori S (December 1999). "Antigen structure and genetic basis of histo-blood groups A, B and O: their changes associated with human cancer". Biochimica et Biophysica Acta (BBA) - General Subjects. 1473 (1): 247–266. doi:10.1016/s0304-4165(99)00183-x. PMID 10580143.
- ^ a b Dabelsteen E, Gao S (January 2005). "ABO blood-group antigens in oral cancer". Journal of Dental Research. 84 (1): 21–28. doi:10.1177/154405910508400103. PMID 15615870. S2CID 16975373.
- ^ Dabelsteen E (February 2002). "ABO blood group antigens in oral mucosa. What is new?". Journal of Oral Pathology & Medicine. 31 (2): 65–70. doi:10.1046/j.0904-2512.2001.00004.x. PMID 11896825.
- ^ Amundadottir LT (1 January 2016). "Pancreatic Cancer Genetics". International Journal of Biological Sciences. 12 (3): 314–325. doi:10.7150/ijbs.15001. PMC 4753160. PMID 26929738.
- ^ McGovern DP, Jones MR, Taylor KD, Marciante K, Yan X, Dubinsky M, et al. (September 2010). "Fucosyltransferase 2 (FUT2) non-secretor status is associated with Crohn's disease". Human Molecular Genetics. 19 (17): 3468–3476. doi:10.1093/hmg/ddq248. PMC 2916706. PMID 20570966.
- ^ de Lange KM, Moutsianas L, Lee JC, Lamb CA, Luo Y, Kennedy NA, et al. (February 2017). "Genome-wide association study implicates immune activation of multiple integrin genes in inflammatory bowel disease". Nature Genetics. 49 (2): 256–261. doi:10.1038/ng.3760. PMC 5289481. PMID 28067908.
- ^ Rühlemann MC, Hermes BM, Bang C, Doms S, Moitinho-Silva L, Thingholm LB, et al. (February 2021). "Genome-wide association study in 8,956 German individuals identifies influence of ABO histo-blood groups on gut microbiome". Nature Genetics. 53 (2): 147–155. doi:10.1038/s41588-020-00747-1. PMID 33462482. S2CID 231641761.
- ^ Mega JL, Stitziel NO, Smith JG, Chasman DI, Caulfield M, Devlin JJ, et al. (June 2015). "Genetic risk, coronary heart disease events, and the clinical benefit of statin therapy: an analysis of primary and secondary prevention trials". Lancet. 385 (9984): 2264–2271. doi:10.1016/S0140-6736(14)61730-X. PMC 4608367. PMID 25748612.
- ^ Liu QP, Sulzenbacher G, Yuan H, Bennett EP, Pietz G, Saunders K, et al. (April 2007). "Bacterial glycosidases for the production of universal red blood cells". Nature Biotechnology. 25 (4): 454–464. doi:10.1038/nbt1298. PMID 17401360. S2CID 29804004.
- ^ Kruskall MS, AuBuchon JP, Anthony KY, Herschel L, Pickard C, Biehl R, et al. (November 2000). "Transfusion to blood group A and O patients of group B RBCs that have been enzymatically converted to group O". Transfusion. 40 (11): 1290–1298. doi:10.1046/j.1537-2995.2000.40111290.x. PMID 11099655. S2CID 22740438.
- ^ Rahfeld P, Withers SG (January 2020). "Toward universal donor blood: Enzymatic conversion of A and B to O type". J Biol Chem. 295 (2): 325–334. doi:10.1074/jbc.REV119.008164. PMC 6956546. PMID 31792054.
- ^ "Scientists create 'plastic' blood". BBC. 11 May 2007. Retrieved 7 April 2019.
- ^ Southern California Blood Services Region. "Answers to Commonly Asked Questions About Blood and Blood Banking". Blood: The Basics (PDF). American Red Cross. p. 4. Archived (PDF) from the original on 29 November 2007. Retrieved 16 November 2007.
- ^ Ryu SI, Sohn YW (2007). "A Review of sociocultural, behavioral, biochemical analyzes on ABO blood-groups typology". Korean Journal of Social and Personality Psychology. 21 (3): 27–55.
- ^ Klein HG (7 March 2005). "Why Do People Have Different Blood Types?". Scientific American. Retrieved 16 November 2007.
Further reading[edit]
- Dean L (2005). "Chapter 5: The ABO blood group". Blood Groups and Red Cell Antigens. Archived from the original on 18 February 2009. Retrieved 24 March 2007.
- Farr AD (April 1979). "Blood group serology--the first four decades (1900--1939)". Medical History. 23 (2): 215–226. doi:10.1017/s0025727300051383. PMC 1082436. PMID 381816.
External links[edit]
- ABO at BGMUT Blood Group Antigen Gene Mutation Database at NCBI, NIH
- Encyclopædia Britannica, ABO blood group system
- National Blood Transfusion Service



![There are three basic variants of immunoglobulin antigens in humans that share a very similar chemical structure but are distinctly different. Red circles show where there are differences in chemical structure in the antigen-binding site (sometimes called the antibody-combining site) of human immunoglobulin. Notice the O-type antigen does not have a binding site.[30]](http://upload.wikimedia.org/wikipedia/commons/thumb/a/aa/AP-Biology_Final_Project.svg/147px-AP-Biology_Final_Project.svg.png)