Amidine
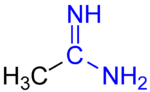
Amidines are organic compounds with the functional group RC(NR)NR2, where the R groups can be the same or different. They are the imine derivatives of amides (RC(O)NR2). The simplest amidine is formamidine, HC(=NH)NH2.
Examples of amidines include:
- DBU
- diminazene
- benzamidine
- Pentamidine
- Paranyline
Preparation[edit]
A common route to primary amidines is the Pinner reaction. Reaction of the nitrile with alcohol in the presence of acid gives an iminoether. Treatment of the resulting compound with ammonia then completes the conversion to the amidine.[1] Instead of using a Bronsted acid, Lewis acids such as aluminium trichloride promote the direct amination of nitriles.[2] They are also generated by amination of an imidoyl chloride.[3] They are also prepared by the addition of organolithium reagents to diimines, followed by protonation or alkylation.
Dimethylformamide acetal reacts with primary amines to give amidines:[4]
- Me2NC(H)(OMe)2 + RNH2 → Me2NC=NHR + 2 MeOH
Acid-base chemistry[edit]
Amidines are much more basic than amides and are among the strongest uncharged/unionized bases.[5][6]
Protonation occurs at the sp2-hybridized nitrogen. This occurs because the positive charge can be delocalized onto both nitrogen atoms. The resulting cationic species is known as an amidinium ion[7] and possesses identical C-N bond lengths.

Applications[edit]
Several drug or drug candidates feature amidine substituents. Examples include the antiprotozoal Imidocarb, the insecticide amitraz , the anthelmintic tribendimidine, and xylamidine, an antagonist at the 5HT2A receptor.[8]
Formamidinium (see below) may be reacted with a metal halide to form the light-absorbing semiconducting material in perovskite solar cells. Formamidinium (FA) cations or halides may partially or fully replace methylammonium halides in forming perovskite absorber layers in photovoltaic devices.
Nomenclature[edit]
Formally, amidines are a class of oxoacids. The oxoacid from which an amidine is derived must be of the form RnE(=O)OH, where R is a substituent. The −OH group is replaced by an −NH2 group and the =O group is replaced by =NR, giving amidines the general structure RnE(=NR)NR2.[9][10][11] When the parent oxoacid is a carboxylic acid, the resulting amidine is a carboxamidine or carboximidamide (IUPAC name). Carboxamidines are frequently referred to simply as amidines, as they are the most commonly encountered type of amidine in organic chemistry.
Derivatives[edit]
Formamidinium cations[edit]

A notable subclass of amidinium ions are the formamidinium cations; which can be represented by the chemical formula [R
2N−CH=NR
2]+
. Deprotonation of these gives stable carbenes which can be represented by the chemical formula R
2N−C:−NR
2.[12][13]
Amidinate salts[edit]

An amidinate salt has the general structure M+[RNRCNR]− and can be accessed by reaction of a carbodiimide with an organometallic compound such as methyl lithium.[15] They are used widely as ligands in organometallic complexes.
See also[edit]
- Guanidines — a similar group of compounds where the central carbon atom is bonded to three nitrogen atoms.
- Imidazolines contain a cyclic amidine.
References[edit]
- ^ A. W. Dox (1928). "Acetamidine Hydrochloride". Organic Syntheses. 8: 1. doi:10.15227/orgsyn.008.0001.
- ^ "N-Phenylbenzamidine". Organic Syntheses. 36: 64. 1956. doi:10.15227/orgsyn.036.0064.
- ^ Arthur C. Hontz, E. C. Wagner (1951). "N,N-Diphenylbenzamidine". Organic Syntheses. 31: 48. doi:10.15227/orgsyn.031.0048.
- ^ Daniel A. Dickman; Michael Boes; Albert I. Meyers (1989). "(S)-N,N-Dimethyl-N'-(1-tert-Butoxy-3-Methyl-2-Butyl)formamidine". Organic Syntheses. 67: 52. doi:10.15227/orgsyn.067.0052.
- ^ Roche VF. Improving Pharmacy Students’ Understanding and Long-term Retention of Acid-Base Chemistry. American Journal of Pharmaceutical Education. 2007;71(6):122.
- ^ Clayden; Greeves; Warren (2001). Organic chemistry. Oxford university press. p. 202. ISBN 978-0-19-850346-0.
- ^ Schrader, Thomas; Hamilton, Andrew D., eds. (2005). Functional synthetic receptors. Wiley-VCH. p. 132. ISBN 3-527-30655-2.
- ^ Greenhill, John V.; Lue, Ping (1993). 5 Amidines and Guanidines in Medicinal Chemistry. Progress in Medicinal Chemistry. Vol. 30. pp. 203–326. doi:10.1016/S0079-6468(08)70378-3. ISBN 9780444899897. PMID 7905649.
- ^ IUPAC, Compendium of Chemical Terminology, 2nd ed. (the "Gold Book") (1997). Online corrected version: (2006–) "amidines". doi:10.1351/goldbook.A00267
- ^ IUPAC, Compendium of Chemical Terminology, 2nd ed. (the "Gold Book") (1997). Online corrected version: (2006–) "carboxamidines". doi:10.1351/goldbook.C00851
- ^ IUPAC, Compendium of Chemical Terminology, 2nd ed. (the "Gold Book") (1997). Online corrected version: (2006–) "sulfinamidines". doi:10.1351/goldbook.S06107
- ^ Alder, Roger W.; Blake, Michael E.; Bufali, Simone; Butts, Craig P.; Orpen, A. Guy; Schütz, Jan; Williams, Stuart J. (2001). "Preparation of tetraalkylformamidinium salts and related species as precursors to stable carbenes". Journal of the Chemical Society, Perkin Transactions 1 (14): 1586–1593. doi:10.1039/B104110J.
- ^ Edward C. Taylor; Wendell A. Ehrhart; M. Kawanisi (1966). "Formamidine Acetate". Organic Syntheses. 46: 39. doi:10.15227/orgsyn.046.0039.
- ^ Keaton, Richard J.; Jayaratne, Kumudini C.; Henningsen, David A.; Koterwas, Lisa A.; Sita, Lawrence R. (2001). "Dramatic Enhancement of Activities for Living Ziegler−Natta Polymerizations Mediated by "Exposed" Zirconium Acetamidinate Initiators: The Isospecific Living Polymerization of Vinylcyclohexane". Journal of the American Chemical Society. 123 (25): 6197–6198. doi:10.1021/ja0057326. PMID 11414862.
- ^ Ulrich, Henri (2007). Chemistry and technology of carbodiimides. Chichester, England: John Wiley & Sons. ISBN 9780470065105.
