Aspergillopepsin II
| Aspergilloglutamic peptidase | |||||||||
|---|---|---|---|---|---|---|---|---|---|
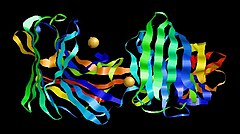 Aspergilloglutamic peptidase dimer | |||||||||
| Identifiers | |||||||||
| EC no. | 3.4.23.19 | ||||||||
| CAS no. | 9025-49-4 | ||||||||
| Databases | |||||||||
| IntEnz | IntEnz view | ||||||||
| BRENDA | BRENDA entry | ||||||||
| ExPASy | NiceZyme view | ||||||||
| KEGG | KEGG entry | ||||||||
| MetaCyc | metabolic pathway | ||||||||
| PRIAM | profile | ||||||||
| PDB structures | RCSB PDB PDBe PDBsum | ||||||||
| |||||||||
Aspergilloglutamic peptidase, also called aspergillopepsin II (EC 3.4.23.19, proctase A, Aspergillus niger acid proteinase A, Aspergillus niger var. macrosporus aspartic proteinase) is a proteolytic enzyme.[1][2] The enzyme was previously thought be an aspartic protease, but it was later shown to be a glutamic protease with a catalytic Glu residue at the active site, and was therefore renamed aspergilloglutamic peptidase.[3]
Determination of its molecular structure showed it to be a unique two-chain enzyme with a light chain and a heavy chain bound non-covalently with each other. The C-terminal region of the light chain of one molecule binds to the active site cleft of another molecule in the manner of a substrate.[4]
This enzyme catalyses the following chemical reaction
- Preferential cleavage in B chain of insulin: Asn3-Gln, Gly13-Ala, Tyr26-Thr
This enzyme is isolated from Aspergillus niger var. macrosporus.
References[edit]
- ^ Chang, W.J.; Horiuchi, S.; Takahashi, K.; Yamasaki, M.; Yamada, Y. (1976). "The structure and function of acid proteases. VI. Effects of acid protease-specific inhibitors on the acid proteases from Aspergillus niger var. macrosporus". J. Biochem. 80: 975–981. PMID 12156.
- ^ Iio, K.; Yamasaki, M. (1976). "Specificity of acid proteinase A from Aspergillus niger var. macrosporus towards B-chain of performic acid oxidized bovine insulin". Biochim. Biophys. Acta. 429: 912–924. doi:10.1016/0005-2744(76)90336-3. PMID 1268233.
- ^ Takahashi K (2013). "Structure and function studies on enzymes with a catalytic carboxyl group(s): from ribonuclease T1 to carboxyl peptidases". Proc Jpn Acad Ser B Phys Biol Sci. 89 (6): 201–25. doi:10.2183/pjab.89.201. PMC 3749792. PMID 23759941.
- ^ Sasaki H, Kubota K, Lee WC, Ohtsuka J, Kojima M, Iwata S, Nakagawa A, Takahashi K, Tanokura M (2012). "The crystal structure of an intermediate dimer of aspergilloglutamic peptidase that mimics the enzyme-activation product complex produced upon autoproteolysis". Journal of Biochemistry. 152 (1): 45–52. doi:10.1093/jb/mvs050. PMID 22569035.
External links[edit]
- Aspergillopepsin+II at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
