Caesium azide
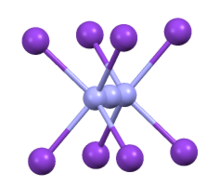 Coordination sphere of azide in CsN3
| |
| Names | |
|---|---|
| IUPAC name
caesium azide
| |
| Other names
cesium azide
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.041.072 |
| EC Number |
|
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties[1] | |
| CsN3 | |
| Molar mass | 174.926 g/mol |
| Appearance | colorless needles |
| Density | 3.5 g/cm3 |
| Melting point | 310 °C (590 °F; 583 K) |
| 224.2 g/100 mL (0 °C) | |
| Structure[2] | |
| tetragonal | |
| I4/mcm, No. 140 | |
a = 6.5412 Å, c = 8.0908 Å
| |
Formula units (Z)
|
4 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Caesium azide or cesium azide is an inorganic compound of caesium and nitrogen. It is a salt of azide with the formula CsN3.
Structure[edit]
CsN3 adopts the same structure as KN3, RbN3, and TlN3, crystallizing in a tetragonal distorted caesium chloride structure where each azide ion coordinates to eight metal cations, and each metal cation coordinates to eight terminal N centers.[2] When heated to 151 °C, it transitions to a cubic structure.[3]
Preparation and reactions[edit]
Caesium azide can be prepared from the neutralization reaction between hydrazoic acid and caesium hydroxide:[4]
CsOH + HN3 → CsN3 + H2O
Caesium carbonate can also be used as the base:
Cs2CO3 + HN3 → CsN3 + CO2 + H2O
Caesium sulfate reacts with barium azide to form insoluble barium sulfate and caesium azide:
Cs2SO4 + Ba(N3)2 → 2CsN3 + BaSO4↓
The thermal decomposition of CsN3 in vacuo can be used as a method of generating high purity caesium metal:[5]
2 CsN3 → 2 Cs + 3 N2
References[edit]
- ^ Perry, Dale L. (18 May 2011). Handbook of Inorganic Compounds. CRC Press. p. 110. ISBN 978-1-4398-1461-1.
- ^ a b Müller, Ulrich (1972). "Verfeinerung der Kristallstrukturen von KN3, RbN3, CsN3 und TIN3". Zeitschrift für anorganische und allgemeine Chemie. 392 (2): 159–166. doi:10.1002/zaac.19723920207.
- ^ McIntyre, J.E. (ed.). Dictionary of Inorganic Compounds Volume 3: C46 – Zr. Chapman & Hill. p. 3096. ISBN 0-412-30120-2.
- ^ Steudel, R.; Schenk, P. W. (1975). "Stickstoff". In Brauer, Georg (ed.). Handbuch der Präparativen Anorganischen Chemie, 3. Auflage, Band I. p. 458. ISBN 3-432-02328-6.
- ^ Blatter, Fritz; Schumacher, Ernst (15 January 1986). "Production of high purity caesium". Journal of the Less Common Metals. 115 (2): 307–313. doi:10.1016/0022-5088(86)90153-0.
