Carbon fibers
This article needs additional citations for verification. (February 2020) |

| Part of a series on |
| Fiber |
|---|
 |
| Natural fibers |
| Human-made fibers |
Carbon fibers or carbon fibres (alternatively CF, graphite fiber or graphite fibre) are fibers about 5 to 10 micrometers (0.00020–0.00039 in) in diameter and composed mostly of carbon atoms.[1] Carbon fibers have several advantages: high stiffness, high tensile strength, high strength to weight ratio, high chemical resistance, high-temperature tolerance, and low thermal expansion.[2] These properties have made carbon fiber very popular in aerospace, civil engineering, military, motorsports, and other competition sports.[3] However, they are relatively expensive compared to similar fibers, such as glass fiber, basalt fibers, or plastic fibers.[4]
To produce a carbon fiber, the carbon atoms are bonded together in crystals that are more or less aligned parallel to the fiber's long axis as the crystal alignment gives the fiber a high strength-to-volume ratio (in other words, it is strong for its size). Several thousand carbon fibers are bundled together to form a tow, which may be used by itself or woven into a fabric.
Carbon fibers are usually combined with other materials to form a composite. For example, when permeated with a plastic resin and baked, it forms carbon-fiber-reinforced polymer (often referred to as carbon fiber), which has a very high strength-to-weight ratio and is extremely rigid although somewhat brittle. Carbon fibers are also composited with other materials, such as graphite, to form reinforced carbon-carbon composites, which have a very high heat tolerance.
Carbon fiber-reinforced materials are used to make aircraft and spacecraft parts, racing car bodies, golf club shafts, bicycle frames, fishing rods, automobile springs, sailboat masts, and many other components where light weight and high strength are needed.
History[edit]

In 1860, Joseph Swan produced carbon fibers for the first time, for use in light bulbs.[5] In 1879, Thomas Edison baked cotton threads or bamboo slivers at high temperatures carbonizing them into an all-carbon fiber filament used in one of the first incandescent light bulbs to be heated by electricity.[6] In 1880, Lewis Latimer developed a reliable carbon wire filament for the incandescent light bulb, heated by electricity.[7]
In 1958, Roger Bacon created high-performance carbon fibers at the Union Carbide Parma Technical Center located outside of Cleveland, Ohio.[8] Those fibers were manufactured by heating strands of rayon until they carbonized. This process proved to be inefficient, as the resulting fibers contained only about 20% carbon. In the early 1960s, a process was developed by Dr. Akio Shindo at Agency of Industrial Science and Technology of Japan, using polyacrylonitrile (PAN) as a raw material. This had produced a carbon fiber that contained about 55% carbon. In 1960 Richard Millington of H.I. Thompson Fiberglas Co. developed a process (US Patent No. 3,294,489) for producing a high carbon content (99%) fiber using rayon as a precursor. These carbon fibers had sufficient strength (modulus of elasticity and tensile strength) to be used as a reinforcement for composites having high strength to weight properties and for high temperature resistant applications.
The high potential strength of carbon fiber was realized in 1963 in a process developed by W. Watt, L. N. Phillips, and W. Johnson at the Royal Aircraft Establishment at Farnborough, Hampshire. The process was patented by the UK Ministry of Defence, then licensed by the British National Research Development Corporation to three companies: Rolls-Royce, who were already making carbon fiber; Morganite; and Courtaulds. Within a few years, after successful use in 1968 of a Hyfil carbon-fiber fan assembly in the Rolls-Royce Conway jet engines of the Vickers VC10,[9] Rolls-Royce took advantage of the new material's properties to break into the American market with its RB-211 aero-engine with carbon-fiber compressor blades. Unfortunately, the blades proved vulnerable to damage from bird impact. This problem and others caused Rolls-Royce such setbacks that the company was nationalized in 1971. The carbon-fiber production plant was sold off to form Bristol Composite Materials Engineering Ltd[10] (often referred to as Bristol Composites).
In the late 1960s, the Japanese took the lead in manufacturing PAN-based carbon fibers. A 1970 joint technology agreement allowed Union Carbide to manufacture Japan's Toray Industries product. Morganite decided that carbon-fiber production was peripheral to its core business, leaving Courtaulds as the only big UK manufacturer. Courtaulds's water-based inorganic process made the product susceptible to impurities that did not affect the organic process used by other carbon-fiber manufacturers, leading Courtaulds ceasing carbon-fiber production in 1991.

During the 1960s, experimental work to find alternative raw materials led to the introduction of carbon fibers made from a petroleum pitch derived from oil processing. These fibers contained about 85% carbon and had excellent flexural strength. Also, during this period, the Japanese Government heavily supported carbon fiber development at home and several Japanese companies such as Toray, Nippon Carbon, Toho Rayon and Mitsubishi started their own development and production. Since the late 1970s, further types of carbon fiber yarn entered the global market, offering higher tensile strength and higher elastic modulus. For example, T400 from Toray with a tensile strength of 4,000 MPa and M40, a modulus of 400 GPa. Intermediate carbon fibers, such as IM 600 from Toho Rayon with up to 6,000 MPa were developed. Carbon fibers from Toray, Celanese and Akzo found their way to aerospace application from secondary to primary parts first in military and later in civil aircraft as in McDonnell Douglas, Boeing, Airbus, and United Aircraft Corporation planes. In 1988, Dr. Jacob Lahijani invented balanced ultra-high Young's modulus (greater than 100 Mpsi) and high tensile strength pitch carbon fiber (greater than 500 kpsi) used extensively in automotive and aerospace applications. In March 2006, the patent was assigned to the University of Tennessee Research Foundation.[11]
Structure and properties[edit]

Carbon fiber is frequently supplied in the form of a continuous tow wound onto a reel. The tow is a bundle of thousands of continuous individual carbon filaments held together and protected by an organic coating, or size, such as polyethylene oxide (PEO) or polyvinyl alcohol (PVA). The tow can be conveniently unwound from the reel for use. Each carbon filament in the tow is a continuous cylinder with a diameter of 5–10 micrometers and consists almost exclusively of carbon. The earliest generation (e.g. T300, HTA and AS4) had diameters of 16–22 micrometers.[12] Later fibers (e.g. IM6 or IM600) have diameters that are approximately 5 micrometers.[12]
The atomic structure of carbon fiber is similar to that of graphite, consisting of sheets of carbon atoms arranged in a regular hexagonal pattern (graphene sheets), the difference being in the way these sheets interlock. Graphite is a crystalline material in which the sheets are stacked parallel to one another in regular fashion. The intermolecular forces between the sheets are relatively weak Van der Waals forces, giving graphite its soft and brittle characteristics.
Depending upon the precursor to make the fiber, carbon fiber may be turbostratic or graphitic, or have a hybrid structure with both graphitic and turbostratic parts present. In turbostratic carbon fiber the sheets of carbon atoms are haphazardly folded, or crumpled, together. Carbon fibers derived from polyacrylonitrile (PAN) are turbostratic, whereas carbon fibers derived from mesophase pitch are graphitic after heat treatment at temperatures exceeding 2200 °C. Turbostratic carbon fibers tend to have high ultimate tensile strength, whereas heat-treated mesophase-pitch-derived carbon fibers have high Young's modulus (i.e., high stiffness or resistance to extension under load) and high thermal conductivity.
Applications[edit]

Carbon fiber can have higher cost than other materials which has been one of the limiting factors of adoption. In a comparison between steel and carbon fiber materials for automotive materials, carbon fiber may be 10-12x more expensive. However, this cost premium has come down over the past decade from estimates of 35x more expensive than steel in the early 2000s.[13]
Composite materials[edit]
Carbon fiber is most notably used to reinforce composite materials, particularly the class of materials known as carbon fiber or graphite reinforced polymers. Non-polymer materials can also be used as the matrix for carbon fibers. Due to the formation of metal carbides and corrosion considerations, carbon has seen limited success in metal matrix composite applications. Reinforced carbon-carbon (RCC) consists of carbon fiber-reinforced graphite, and is used structurally in high-temperature applications. The fiber also finds use in filtration of high-temperature gases, as an electrode with high surface area and impeccable corrosion resistance, and as an anti-static component. Molding a thin layer of carbon fibers significantly improves fire resistance of polymers or thermoset composites because a dense, compact layer of carbon fibers efficiently reflects heat.[14]
The increasing use of carbon fiber composites is displacing aluminum from aerospace applications in favor of other metals because of galvanic corrosion issues.[15][16] Note, however, that carbon fiber does not eliminate the risk of galvanic corrosion.[17] In contact with metal, it forms "a perfect galvanic corrosion cell ..., and the metal will be subjected to galvanic corrosion attack" unless a sealant is applied between the metal and the carbon fiber.[18]
Carbon fiber can be used as an additive to asphalt to make electrically conductive asphalt concrete.[19] Using this composite material in the transportation infrastructure, especially for airport pavement, decreases some winter maintenance problems that lead to flight cancellation or delay due to the presence of ice and snow. Passing current through the composite material 3D network of carbon fibers dissipates thermal energy that increases the surface temperature of the asphalt, which is able to melt ice and snow above it.[20]
Textiles[edit]



Precursors for carbon fibers are polyacrylonitrile (PAN), rayon and pitch. Carbon fiber filament yarns are used in several processing techniques: the direct uses are for prepregging, filament winding, pultrusion, weaving, braiding, etc. Carbon fiber yarn is rated by the linear density (weight per unit length; i.e., 1 g/1000 m = 1 tex) or by number of filaments per yarn count, in thousands. For example, 200 tex for 3,000 filaments of carbon fiber is three times as strong as 1,000 carbon filament yarn, but is also three times as heavy. This thread can then be used to weave a carbon fiber filament fabric or cloth. The appearance of this fabric generally depends on the linear density of the yarn and the weave chosen. Some commonly used types of weave are twill, satin and plain. Carbon filament yarns can also be knitted or braided.
Microelectrodes[edit]
Carbon fibers are used for fabrication of carbon-fiber microelectrodes. In this application typically a single carbon fiber with diameter of 5–7 μm is sealed in a glass capillary.[21] At the tip the capillary is either sealed with epoxy and polished to make a carbon-fiber disk microelectrode, or the fiber is cut to a length of 75–150 μm to make a carbon-fiber cylinder electrode. Carbon-fiber microelectrodes are used either in amperometry or fast-scan cyclic voltammetry for detection of biochemical signaling.
Flexible heating[edit]

Despite being known for their electrical conductivity, carbon fibers can carry only very low currents on their own. When woven into larger fabrics, they can be used to reliably provide (infrared) heating in applications requiring flexible electrical heating elements and can easily sustain temperatures past 100 °C. Many examples of this type of application can be seen in DIY heated articles of clothing and blankets. Due to its chemical inertness, it can be used relatively safely amongst most fabrics and materials; however, shorts caused by the material folding back on itself will lead to increased heat production and can lead to a fire.
Synthesis[edit]

- Polymerization of acrylonitrile to PAN,
- Cyclization during the low-temperature process,
- High-temperature oxidative treatment of carbonization (hydrogen is removed). After this process of graphitization starts where nitrogen is removed and chains are joined into graphite planes.
Each carbon filament is produced from a polymer such as polyacrylonitrile (PAN), rayon, or petroleum pitch. All these polymers are known as a precursor. For synthetic polymers such as PAN or rayon, the precursor is first spun into filament yarns, using chemical and mechanical processes to initially align the polymer molecules in a way to enhance the final physical properties of the completed carbon fiber. Precursor compositions and mechanical processes used during spinning filament yarns may vary among manufacturers. After drawing or spinning, the polymer filament yarns are then heated to drive off non-carbon atoms (carbonization), producing the final carbon fiber. The carbon fibers filament yarns may be further treated to improve handling qualities, then wound on to bobbins.[22]
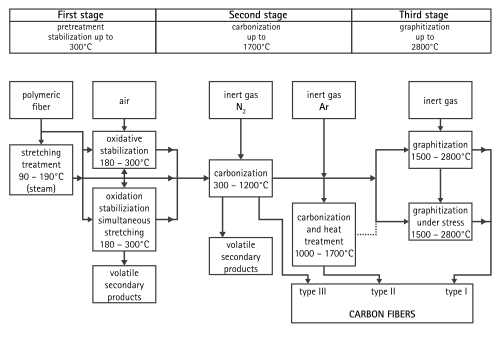

A common method of manufacture involves heating the spun PAN filaments to approximately 300 °C in air, which breaks many of the hydrogen bonds and oxidizes the material. The oxidized PAN is then placed into a furnace having an inert atmosphere of a gas such as argon, and heated to approximately 2000 °C, which induces graphitization of the material, changing the molecular bond structure. When heated in the correct conditions, these chains bond side-to-side (ladder polymers), forming narrow graphene sheets which eventually merge to form a single, columnar filament. The result is usually 93–95% carbon. Lower-quality fiber can be manufactured using pitch or rayon as the precursor instead of PAN. The carbon can become further enhanced, as high modulus, or high strength carbon, by heat treatment processes. Carbon heated in the range of 1500–2000 °C (carbonization) exhibits the highest tensile strength (5,650 MPa, or 820,000 psi), while carbon fiber heated from 2500 to 3000 °C (graphitizing) exhibits a higher modulus of elasticity (531 GPa, or 77,000,000 psi).
See also[edit]
- Basalt fiber
- Carbon fiber reinforced polymer
- Carbon fiber reinforced ceramic material
- Carbon nanotube
- ESD materials
- Graphene
References[edit]
- ^ Chaudhary, Anisha; Gupta, Vinay; Teotia, Satish; Nimanpure, Subhash; Rajak, Dipen K. (2021-01-01), "Electromagnetic Shielding Capabilities of Metal Matrix Composites", in Brabazon, Dermot (ed.), Encyclopedia of Materials: Composites, Oxford: Elsevier, pp. 428–441, ISBN 978-0-12-819731-8, retrieved 2022-02-14
- ^ Bhatt, Pooja (2017). Carbon Fibres: Production, Properties and Potential Use (Thesis). Archived from the original on 2021-04-30. Retrieved 2021-07-25.
- ^ "What is the Carbon Fiber Plate in Shoes? Let's Make It All Clear Here!". clarco.com. 19 November 2022. Retrieved 2022-11-19.
- ^ Hasiotis, Theodoros; Badogiannis, Efstratios; Tsouvalis, Nicolaos Georgios (2011). "Application of Ultrasonic C-Scan Techniques for Tracing Defects in Laminated Composite Materials". Strojniški vestnik – Journal of Mechanical Engineering. 57 (3): 192–203. doi:10.5545/sv-jme.2010.170. ISSN 2536-3948. Retrieved 2023-02-21.
- ^ Deng, Yuliang (2007). Carbon Fiber Electronic Interconnects (PDF) (Thesis). Archived (PDF) from the original on 2019-04-04. Retrieved 2017-03-02.
- ^ "High Performance Carbon Fibers". National Historic Chemical Landmarks. American Chemical Society. 2003. Archived from the original on 2014-04-27. Retrieved 2014-04-26.
- ^ "The Gifted Men Who Worked for Edison". National Park Service. Archived from the original on 2015-02-07. Retrieved 2014-12-01.
- ^ US 2957756, Bacon, Roger, "Filamentary graphite and method for producing the same", issued 1960-10-25
- ^ "Stand Points". Flight International: 481. 1968-09-26. Archived from the original on 2014-08-14. Retrieved 2014-08-14 – via Flight Global Archive.
- ^ "Rolls-Royce - Graces Guide". www.gracesguide.co.uk. Retrieved 2020-09-22.
- ^ US 4915926, Lahijani, Jacob, "Balanced ultra-high modulus and high tensile strength carbon fibers", published 1990-04-10
- ^ a b Cantwell, W.J.; Morton, J. (1991). "The impact resistance of composite materials – a review". Composites. 22 (5): 347–362. doi:10.1016/0010-4361(91)90549-V.
- ^ Bregar, Bill (5 August 2014). "Price keeping carbon fiber from mass adoption - Plastics News". Plastics News. Atlanta: Crain Communications, Inc. Archived from the original on 2016-12-09. Retrieved 2017-05-25.
- ^ Zhao, Z.; Gou, J. (2009). "Improved fire retardancy of thermoset composites modified with carbon nanofibers". Sci. Technol. Adv. Mater. 10 (1): 015005. Bibcode:2009STAdM..10a5005Z. doi:10.1088/1468-6996/10/1/015005. PMC 5109595. PMID 27877268.
- ^ Banis, David; Marceau, J. Arthur; Mohaghegh, Michael (July 1999). "Design for Corrosion". Aero. No. 7. Boeing. Archived from the original on 2013-09-02. Retrieved 2018-05-07.
- ^ Warwick, Graham; Norris, Guy (2013-05-06). "Metallics Make Comeback With Manufacturing Advances". Aviation Week & Space Technology. Archived from the original on 2015-04-27.
- ^ Banis, David; Marceau, J. Arthur; Mohaghegh, Michael (July 1999). "Design for Corrosion". Aero. No. 7. Boeing. Archived from the original on 2013-09-02. Retrieved 2018-05-07.
- ^ Song, Guang-Ling; Chi, Zhang; Xiaodong, Chen (March 2021). "Galvanic activity of carbon fiber reinforced polymers and electrochemical behavior of carbon fiber". Corrosion Communications. Vol. 1, no. 1. Elsevier B.V. pp. 26–39. doi:10.1016/j.corcom.2021.05.003. Retrieved 2023-01-22.
- ^ Notani, Mohammad Ali; Arabzadeh, Ali; Ceylan, Halil; Kim, Sunghwan (June 2019). "Effect of Carbon-Fiber Properties on Volumetrics and Ohmic Heating of Electrically Conductive Asphalt Concrete". Journal of Materials in Civil Engineering. 31 (9). US: 04019200. doi:10.1061/(ASCE)MT.1943-5533.0002868. S2CID 198395022.
- ^ Arabzadeh, Ali; Notani, Mohammad Ali; Zadeh, Ayoub Kazemiyan; Nahvi, Ali; Sassani, Alireza; Ceylan, Halil (2019-09-15). "Electrically conductive asphalt concrete: An alternative for automating the winter maintenance operations of transportation infrastructure". Composites Part B: Engineering. 173. US: 106985. doi:10.1016/j.compositesb.2019.106985. S2CID 189994116.
- ^ Pike, Carolyn M.; Grabner, Chad P.; Harkins, Amy B. (2009-05-04). "Fabrication of Amperometric Electrodes". Journal of Visualized Experiments (27): 1040. doi:10.3791/1040. PMC 2762914. PMID 19415069.
- ^ "How is Carbon Fiber Made?". Zoltek. 2017-08-10. Archived from the original on 2015-03-19.
External links[edit]
- Making Carbon Fiber
- How carbon fiber is made
- "Carbon Fibres – the First 5 naked years". Flight International. 1971-09-09. p. 406. Archived from the original on 2012-02-04.
