Clausius–Clapeyron relation
The Clausius–Clapeyron relation, in chemical thermodynamics, specifies the temperature dependence of pressure, most importantly vapor pressure, at a discontinuous phase transition between two phases of matter of a single constituent. It is named after Rudolf Clausius[1] and Benoît Paul Émile Clapeyron.[2] However, this relation was in fact originally derived by Sadi Carnot in his Reflections on the Motive Power of Fire, which was published in 1824 but largely ignored until it was rediscovered by Clausius, Clapeyron, and Lord Kelvin decades later.[3] Kelvin said of Carnot's argument that "nothing in the whole range of Natural Philosophy is more remarkable than the establishment of general laws by such a process of reasoning."[4]
Kelvin and his brother James Thomson confirmed the relation experimentally in 1849–50, and it was historically important as a very early successful application of theoretical thermodynamics.[5] Its relevance to meteorology and climatology is the increase of the water-holding capacity of the atmosphere by about 7% for every 1 °C (1.8 °F) rise in temperature.
Definition[edit]
Exact Clapeyron equation[edit]
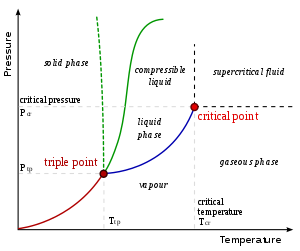
On a pressure–temperature (P–T) diagram, for any phase change the line separating the two phases is known as the coexistence curve. The Clapeyron relation[6] gives the slope of the tangents to this curve. Mathematically,
Clausius–Clapeyron equation[edit]
The Clausius–Clapeyron equation[7]: 509 applies to vaporization of liquids where vapor follows ideal gas law using the specific gas constant and liquid volume is neglected as being much smaller than vapor volume V. It is often used to calculate vapor pressure of a liquid.[8]
The equation expresses this in a more convenient form just in terms of the latent heat, for moderate temperatures and pressures.
Derivations[edit]
Derivation from state postulate[edit]
Using the state postulate, take the specific entropy for a homogeneous substance to be a function of specific volume and temperature .[7]: 508
The Clausius–Clapeyron relation describes a Phase transition in a closed system composed of two contiguous phases, condensed matter and ideal gas, of a single substance, in mutual thermodynamic equilibrium, at constant temperature and pressure. Therefore,[7]: 508
Using the appropriate Maxwell relation gives[7]: 508
Given constant pressure and temperature (during a phase change), we obtain[7]: 508
Substituting the definition of specific latent heat gives
Substituting this result into the pressure derivative given above (), we obtain[7]: 508 [11]
This result (also known as the Clapeyron equation) equates the slope of the coexistence curve to the function of the specific latent heat , the temperature , and the change in specific volume . Instead of the specific, corresponding molar values may also be used.
Derivation from Gibbs–Duhem relation[edit]
Suppose two phases, and , are in contact and at equilibrium with each other. Their chemical potentials are related by
Furthermore, along the coexistence curve,
One may therefore use the Gibbs–Duhem relation
Rearrangement gives
from which the derivation of the Clapeyron equation continues as in the previous section.
Ideal gas approximation at low temperatures[edit]
When the phase transition of a substance is between a gas phase and a condensed phase (liquid or solid), and occurs at temperatures much lower than the critical temperature of that substance, the specific volume of the gas phase greatly exceeds that of the condensed phase . Therefore, one may approximate
where is the pressure, is the specific gas constant, and is the temperature. Substituting into the Clapeyron equation
Let and be any two points along the coexistence curve between two phases and . In general, varies between any two such points, as a function of temperature. But if is approximated as constant,
These last equations are useful because they relate equilibrium or saturation vapor pressure and temperature to the latent heat of the phase change without requiring specific-volume data. For instance, for water near its normal boiling point, with a molar enthalpy of vaporization of 40.7 kJ/mol and R = 8.31 J/(mol⋅K),
Clapeyron's derivation[edit]
In the original work by Clapeyron, the following argument is advanced.[13] Clapeyron considered a Carnot process of saturated water vapor with horizontal isobars. As the pressure is a function of temperature alone, the isobars are also isotherms. If the process involves an infinitesimal amount of water, , and an infinitesimal difference in temperature , the heat absorbed is
Applications[edit]
Chemistry and chemical engineering[edit]
For transitions between a gas and a condensed phase with the approximations described above, the expression may be rewritten as
Meteorology and climatology[edit]
Atmospheric water vapor drives many important meteorologic phenomena (notably, precipitation), motivating interest in its dynamics. The Clausius–Clapeyron equation for water vapor under typical atmospheric conditions (near standard temperature and pressure) is
where
- is saturation vapor pressure,
- is temperature,
- is the specific latent heat of evaporation of water,
- is the gas constant of water vapor.
The temperature dependence of the latent heat cannot be neglected in this application. Fortunately, the August–Roche–Magnus formula provides a very good approximation:[14][15]
This is also sometimes called the Magnus or Magnus–Tetens approximation, though this attribution is historically inaccurate.[16] But see also the discussion of the accuracy of different approximating formulae for saturation vapour pressure of water.
Under typical atmospheric conditions, the denominator of the exponent depends weakly on (for which the unit is degree Celsius). Therefore, the August–Roche–Magnus equation implies that saturation water vapor pressure changes approximately exponentially with temperature under typical atmospheric conditions, and hence the water-holding capacity of the atmosphere increases by about 7% for every 1 °C rise in temperature.[17]
Example[edit]
One of the uses of this equation is to determine if a phase transition will occur in a given situation. Consider the question of how much pressure is needed to melt ice at a temperature below 0 °C. Note that water is unusual in that its change in volume upon melting is negative. We can assume
- (latent heat of fusion for water),
- (absolute temperature in kelvins),
- (change in specific volume from solid to liquid),
we obtain
To provide a rough example of how much pressure this is, to melt ice at −7 °C (the temperature many ice skating rinks are set at) would require balancing a small car (mass ~ 1000 kg[18]) on a thimble (area ~ 1 cm2). This shows that ice skating cannot be simply explained by pressure-caused melting point depression, and in fact the mechanism is quite complex.[19]
Second derivative[edit]
While the Clausius–Clapeyron relation gives the slope of the coexistence curve, it does not provide any information about its curvature or second derivative. The second derivative of the coexistence curve of phases 1 and 2 is given by[20]
See also[edit]
References[edit]
- ^ Clausius, R. (1850). "Ueber die bewegende Kraft der Wärme und die Gesetze, welche sich daraus für die Wärmelehre selbst ableiten lassen" [On the motive power of heat and the laws which can be deduced therefrom regarding the theory of heat]. Annalen der Physik (in German). 155 (4): 500–524. Bibcode:1850AnP...155..500C. doi:10.1002/andp.18501550403. hdl:2027/uc1.$b242250.
- ^ Clapeyron, M. C. (1834). "Mémoire sur la puissance motrice de la chaleur". Journal de l'École polytechnique (in French). 23: 153–190. ark:/12148/bpt6k4336791/f157.
- ^ Feynman, Richard (1963). "Illustrations of Thermodynamics". The Feynman Lectures on Physics. California Institute of Technology. Retrieved 13 December 2023.
This relationship was deduced by Carnot, but it is called the Clausius-Clapeyron equation.
- ^ Thomson, William (1849). "An Account of Carnot's Theory of the Motive Power of Heat; with Numerical Results deduced from Regnault's Experiments on Steam". Transactions of the Edinburgh Royal Society. 16 (5): 541–574. doi:10.1017/S0080456800022481.
- ^ Pippard, Alfred B. (1981). Elements of classical thermodynamics: for advanced students of physics (Repr ed.). Cambridge: Univ. Pr. p. 116. ISBN 978-0-521-09101-5.
- ^ Koziol, Andrea; Perkins, Dexter. "Teaching Phase Equilibria". serc.carleton.edu. Carleton University. Retrieved 1 February 2023.
- ^ a b c d e f g h i Wark, Kenneth (1988) [1966]. "Generalized Thermodynamic Relationships". Thermodynamics (5th ed.). New York, NY: McGraw-Hill, Inc. ISBN 978-0-07-068286-3.
- ^ Clausius; Clapeyron. "The Clausius-Clapeyron Equation". Bodner Research Web. Purdue University. Retrieved 1 February 2023.
- ^ a b Çengel, Yunus A.; Boles, Michael A. (1998) [1989]. Thermodynamics – An Engineering Approach. McGraw-Hill Series in Mechanical Engineering (3rd ed.). Boston, MA.: McGraw-Hill. ISBN 978-0-07-011927-7.
- ^ Salzman, William R. (2001-08-21). "Clapeyron and Clausius–Clapeyron Equations". Chemical Thermodynamics. University of Arizona. Archived from the original on 2007-06-07. Retrieved 2007-10-11.
- ^ Masterton, William L.; Hurley, Cecile N. (2008). Chemistry : principles and reactions (6th ed.). Cengage Learning. p. 230. ISBN 9780495126713. Retrieved 3 April 2020.
- ^ Clapeyron, E (1834). "Mémoire sur la puissance motrice de la chaleur". Journal de l ́École Polytechnique. XIV: 153–190.
- ^ Alduchov, Oleg; Eskridge, Robert (1997-11-01), Improved Magnus' Form Approximation of Saturation Vapor Pressure, NOAA, doi:10.2172/548871 Equation 25 provides these coefficients.
- ^ Alduchov, Oleg A.; Eskridge, Robert E. (1996). "Improved Magnus Form Approximation of Saturation Vapor Pressure". Journal of Applied Meteorology. 35 (4): 601–609. Bibcode:1996JApMe..35..601A. doi:10.1175/1520-0450(1996)035<0601:IMFAOS>2.0.CO;2. Equation 21 provides these coefficients.
- ^ Lawrence, M. G. (2005). "The Relationship between Relative Humidity and the Dewpoint Temperature in Moist Air: A Simple Conversion and Applications" (PDF). Bulletin of the American Meteorological Society. 86 (2): 225–233. Bibcode:2005BAMS...86..225L. doi:10.1175/BAMS-86-2-225.
- ^ IPCC, Climate Change 2007: Working Group I: The Physical Science Basis, "FAQ 3.2 How is Precipitation Changing?". Archived 2018-11-02 at the Wayback Machine.
- ^ Zorina, Yana (2000). "Mass of a Car". The Physics Factbook.
- ^ Liefferink, Rinse W.; Hsia, Feng-Chun; Weber, Bart; Bonn, Daniel (2021-02-08). "Friction on Ice: How Temperature, Pressure, and Speed Control the Slipperiness of Ice". Physical Review X. 11 (1): 011025. doi:10.1103/PhysRevX.11.011025.
- ^ Krafcik, Matthew; Sánchez Velasco, Eduardo (2014). "Beyond Clausius–Clapeyron: Determining the second derivative of a first-order phase transition line". American Journal of Physics. 82 (4): 301–305. Bibcode:2014AmJPh..82..301K. doi:10.1119/1.4858403.
Bibliography[edit]
- Yau, M. K.; Rogers, R. R. (1989). Short Course in Cloud Physics (3rd ed.). Butterworth–Heinemann. ISBN 978-0-7506-3215-7.
- Iribarne, J. V.; Godson, W. L. (2013). "4. Water-Air systems § 4.8 Clausius–Clapeyron Equation". Atmospheric Thermodynamics. Springer. pp. 60–. ISBN 978-94-010-2642-0.
- Callen, H. B. (1985). Thermodynamics and an Introduction to Thermostatistics. Wiley. ISBN 978-0-471-86256-7.
Notes[edit]
- ^ In the original work, was simply called the Carnot function and was not known in this form. Clausius determined the form 30 years later and added his name to the eponymous Clausius–Clapeyron relation.





















































![{\displaystyle P_{\text{vap}}(T)\cong 1~{\text{bar}}\cdot \exp \left[-{\frac {40\,700~{\text{K}}}{8.31}}\left({\frac {1}{T}}-{\frac {1}{373~{\text{K}}}}\right)\right].}](https://wikimedia.org/api/rest_v1/media/math/render/svg/14efba4091ecdf4bf388b40be1d429cc526d996a)

























![{\displaystyle {\begin{aligned}{\frac {\mathrm {d} ^{2}P}{\mathrm {d} T^{2}}}&={\frac {1}{v_{2}-v_{1}}}\left[{\frac {c_{p2}-c_{p1}}{T}}-2(v_{2}\alpha _{2}-v_{1}\alpha _{1}){\frac {\mathrm {d} P}{\mathrm {d} T}}\right]\\{}&+{\frac {1}{v_{2}-v_{1}}}\left[(v_{2}\kappa _{T2}-v_{1}\kappa _{T1})\left({\frac {\mathrm {d} P}{\mathrm {d} T}}\right)^{2}\right],\end{aligned}}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/2bc93e293ca2eaa0ab9ba9882bde1693b49c2b41)


