Diethylaluminium chloride
| |
| Names | |
|---|---|
| IUPAC name
Chlorodiethylalumane
| |
| Other names
Chlorodiethylaluminium
| |
| Identifiers | |
3D model (JSmol)
|
|
| 4123259 | |
| ChemSpider | |
| ECHA InfoCard | 100.002.253 |
| EC Number |
|
PubChem CID
|
|
| RTECS number |
|
| UNII | |
| UN number | 3394 |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C8H20Al2Cl2 | |
| Molar mass | 241.11 g·mol−1 |
| Appearance | Colorless liquid[1] |
| Density | 0.96 g/cm3[1] |
| Melting point | −74 °C (−101 °F; 199 K)[1] |
| Boiling point | 125 to 126 °C (257 to 259 °F; 398 to 399 K) at 50 mmHg |
| Reacts[1] | |
| Vapor pressure | 3 mmHg (at 60 °C) |
| Hazards | |
| GHS labelling: | |
 
| |
| Danger | |
| H225, H250, H260, H261, H314 | |
| P210, P222, P223, P231+P232, P233, P240, P241, P242, P243, P260, P264, P280, P301+P330+P331, P302+P334, P303+P361+P353, P304+P340, P305+P351+P338, P310, P321, P335+P334, P363, P370+P378, P402+P404, P403+P235, P405, P422, P501 | |
| NFPA 704 (fire diamond) | |
| Flash point | −18 °C (0 °F; 255 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
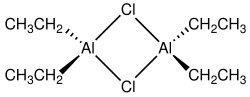
Diethylaluminium chloride, abbreviated DEAC, is an organoaluminium compound. Although often given the chemical formula (C2H5)2AlCl, it exists as a dimer, [(C2H5)2AlCl]2 It is a precursor to Ziegler-Natta catalysts employed for the production of polyolefins. The compound is also a Lewis acid, useful in organic synthesis. The compound is a colorless waxy solid, but is usually handled as a solution in hydrocarbon solvents. It is highly reactive, even pyrophoric.[2]
Structure and bonding[edit]
Compounds of the empirical formula AlR2Cl (R = alkyl, aryl) usually exist as dimers with the formula (R2Al)2(μ-Cl)2. The bridging ligands (indicated by "μ-") are halides, not the organic substituents. The aluminium adopts a tetrahedral geometry. Each Al(III) center follows the octet rule.[3][4] In contrast, triethylaluminium and trimethylaluminium feature bridging alkyl groups and these compounds violate the octet rule.
Production[edit]
Diethylaluminium chloride can be produced from ethylaluminium sesquichloride, (C2H5)3Al2Cl3, by reduction with sodium:[5]
- 2 (C2H5)3Al2Cl3 + 3 Na → 3 (C2H5)2AlCl + Al + 3 NaCl
It is also obtained from the reaction of triethylaluminium with hydrochloric acid:
- (C2H5)3Al + HCl → (C2H5)2AlCl + C2H6
Reproportionation reactions can also be used:
- 2 (C2H5)3Al + AlCl3 → 3 (C2H5)2AlCl
- (C2H5)3Al2Cl3 + (C2H5)3Al → 3 (C2H5)2AlCl
Uses[edit]
Diethylaluminium chloride and other organoaluminium compounds are used in combination with transition metal compounds as Ziegler–Natta catalysts for the polymerization of various alkenes.[6]
As a Lewis acid, diethylaluminium chloride also has uses in organic synthesis. For example, it is used to catalyze the Diels–Alder and ene reactions. Alternatively, it can react as a nucleophile or a proton scavenger.[2]
Safety[edit]
Diethylaluminium chloride is not only flammable but pyrophoric.
References[edit]
- Hu, Y. J.; Jiang, H. L.; Wang, H. H., "Preparation of highly branched polyethylene with acenaphthenediimine nickel chloride/diethylaluminum chloride catalyst". Chinese Journal of Polymer Science 2006, 24 (5), 483–488.
- Yao, Y. M.; Qi, G. Z.; Shen, Q.; Hu, J. Y.; Lin, Y. H., "Reactivity and structural characterization of divalent samarium aryloxide with diethylaluminum chloride". Chinese Science Bulletin 2003, 48 (20), 2164–2167.
- ^ a b c d John Rumble (June 18, 2018). CRC Handbook of Chemistry and Physics (99th ed.). CRC Press. pp. 4–40. ISBN 978-1138561632.
- ^ a b Snider, Barry B. (2001). "Diethylaluminum Chloride". Encyclopedia of Reagents for Organic Synthesis. doi:10.1002/047084289X.rd165. ISBN 0-471-93623-5.
- ^ Brendhaugen, Kristen; Haaland, Arne; Novak, David P.; Østvold, Terje; Bjørseth, Alf; Powell, D. L. (1974). "The Molecular Structure of Dimethylaluminium Chloride Dimer, [(CH3)2AlCl]2 Redetermined by Gas Phase Electron Diffraction". Acta Chemica Scandinavica. 28a: 45–47. doi:10.3891/acta.chem.scand.28a-0045.
- ^ McMahon, C. Niamh; Francis, Julie A.; Barron, Andrew R. (1997). "Molecular Atructure of [(t Bu)2Al(μ-Cl)]2". Journal of Chemical Crystallography. 27 (3): 191–194. doi:10.1007/BF02575988. S2CID 195242291.
- ^ Krause, Michael J.; Orlandi, Frank; Saurage, Alfred T.; Zietz, Joseph R. (2000), "Aluminum Compounds, Organic", Ullmann's Encyclopedia of Industrial Chemistry, Weinheim, Germany: Wiley-VCH Verlag GmbH & Co. KGaA, pp. 592–593, doi:10.1002/14356007.a01_543, ISBN 978-3-527-30673-2
- ^ Fisch, A. G. (2000). "Ziegler–Natta Catalysts". Kirk-Othmer Encyclopedia of Chemical Technology. Wiley. pp. 1–22. doi:10.1002/0471238961.2609050703050303.a01.pub2. ISBN 978-0-471-48494-3. S2CID 213111515.
External links[edit]
 Media related to Diethylaluminium chloride at Wikimedia Commons
Media related to Diethylaluminium chloride at Wikimedia Commons

