Endochondral ossification
| Endochondral ossification | |
|---|---|
 A schematic representation of endochondral ossification. | |
| Anatomical terminology |
Endochondral ossification[1][2] is one of the two essential pathways by which bone tissue is produced during fetal development of the mammalian skeletal system, the other pathway being intramembranous ossification. Both endochondral and intramembranous processes initiate from a precursor mesenchymal tissue, but their transformations into bone are different. In intramembranous ossification, mesenchymal tissue is directly converted into bone. On the other hand, endochondral ossification starts with mesenchymal tissue turning into an intermediate cartilage stage, which is eventually substituted by bone.[3]
Endochondral ossification is responsible for development of most bones including long and short bones,[4] the bones of the axial (ribs and vertebrae) and the appendicular skeleton (e.g. upper and lower limbs),[5] the bones of the skull base (including the ethmoid and sphenoid bones)[6] and the medial end of the clavicle.[7] In addition, endochondral ossification is not exclusively confined to embryonic development; it also plays a crucial role in the healing of fractures.[3]
Formation of the cartilage model[edit]
The initiation of endochondral ossification starts by proliferation and condensation of mesenchymal cells in the area where the bone will eventually be formed. Subsequently, these mesenchymal progenitor cells differentiate into chondroblasts, which actively synthesize cartilage matrix components. Thus, the initial hyaline cartilage template is formed, which has the same basic shape and outline as the future bone.[8]
| Interstitial growth | Appositional growth | |
|---|---|---|
| Cellular protagonists | Chondrocytes present within the existing cartilage. | Chondroblasts that develop from the perichondrium. |
| Mechanism | Chondrocytes proliferate and lay down matrix. | Chondroblasts differentiate into chondrocytes and lay down matrix. |
| Site of expansion | From within. | From the external surface of existing cartilage. |
| Outcome | Increase in length. | Increase in width and thickness. |
Primary center of ossification[edit]

In developing bones, ossification commences within the primary ossification center located in the center of the diaphysis (bone shaft),[5] where the following changes occur:
- The perichondrium surrounding the cartilage model transforms into the periosteum. During this transformation, special cells within the perichondrium switch gears. Instead of becoming cartilage cells (chondrocytes), they mature into bone-building osteoblasts.[5] This newly formed bone can be called "periosteal bone" as it originates from the transformed periosteum. However, considering its developmental pathway, it could be classified as "intramembranous bone".[8]
- After the formation of the periosteum, chondrocytes in the primary center of ossification begin to grow (hypertrophy). They begin secreting:[10][11]
- Collagen type X, which causes stiffness and compression of the extracellular matrix.
- Matrix metalloproteinases.
- Vascular endothelial growth factor (VEGF), which controls forthcoming vascular invasion.
- Alkaline phosphatase, which causes calcification of the cartilage matrix. This calcification prevents passage of nutrients to chondrocytes leading to their death.
- When chondrocytes die, matrix metalloproteinases result in catabolism of various components within the extracellular matrix and the physical boundaries between neighboring lacunae (the spaces housing chondrocytes) weaken. This can lead to the merging of these lacunae, creating larger empty spaces.[8][9]
- Blood vessels arising from the periosteum invade these empty spaces and mesenchymal stem cells migrate guided by penetrating blood vessels. Following the invading blood vessels, mesenchymal stem cells reach these empty spaces and undergo differentiation into osteoprogenitor cells. These progenitors further mature into osteoblasts, that deposit unmineralized bone matrix, termed osteoid. Mineralization subsequently follows leading to formation of bone trabeculae (Endochondral bone formation).[11]

Secondary center of ossification[edit]
During the postnatal life, a secondary ossification center appears in each end (epiphysis) of long bones. In these secondary centers, cartilage is converted to bone similarly to that occurring in a primary ossification center.[8] As the secondary ossification centers enlarge, residual cartilage persists in two distinct locations:[11]
- Articular cartilage: This layer coats the bone ends, concerned with joint movement.
- Epiphyseal growth plate: This transverse layer lies between the epiphysis and diaphysis. It’s composed of highly active chondrocytes and responsible for longitudinal bone growth. Consequently, the bone elongates at this growth plate until closure occurs at skeletal maturity.
At the end of an individual’s growth period, the production of new cartilage in the epiphyseal plate stops. After this point, existing cartilage within the plate turns into mature bone tissue.[8]
Histology[edit]
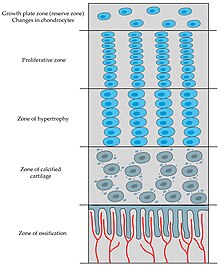
During endochondral ossification, five distinct zones can be seen at the light-microscope level:[3]
| Name | Definition |
|---|---|
| Zone of resting cartilage | This zone contains normal, resting hyaline cartilage. |
| Zone of proliferation / cell columns | In this zone, chondrocytes undergo rapid mitosis, forming distinctive looking columns. |
| Zone of maturation / hypertrophy | In this zone, the chondrocytes undergo hypertrophy (become enlarged). Chondrocytes contain large amounts of glycogen and begin to secrete vascular endothelial growth factor to initiate vascular invasion. |
| Zone of calcification | In this zone, chondrocytes are either dying or dead, leaving cavities that will later become invaded by bone-forming cells. Chondrocytes here die when they can no longer receive nutrients or eliminate wastes via diffusion. This is because the calcified matrix is much less hydrated than hyaline cartilage. |
| Zone of ossification | Osteoprogenitor cells invade the area and differentiate into osteoblasts, which elaborate matrix that becomes calcified on the surface of calcified cartilage. |
Fracture healing[edit]
For complete recovery of a fractured bone’s biomechanical functionality, the bone healing process needs to culminate in the formation of lamellar bone at the fracture site to withstand the same forces and stresses it did before the fracture. Indirect fracture healing, the most common type of bone repair,[10] relies heavily on endochondral ossification. In this type of healing, endochondral ossification occurs within the fracture gap and external to the periosteum. In contrast, intramembranous ossification takes place directly beneath the periosteum, adjacent to the broken bone’s ends.[10][12]

Additional images[edit]
-
Masson Goldner trichrome stain of growth plate in a rabbit tibia.
-
Section of fetal bone of cat. ir. Irruption of the subperiosteal tissue. p. Fibrous layer of the periosteum. o. Layer of osteoblasts. im. Subperiosteal bony deposit.
-
Process of endochondral ossification.
-
Drawing of part of a longitudinal section of the developing femur of a rabbit. a. Flattened cartilage cells. b. Enlarged cartilage cells. c, d. Newly formed bone. e. Osteoblasts. f. Giant cells or osteoclasts. g, h. Shrunken cartilage cells.
References[edit]
- ^ Etymology from Greek: ἔνδον/endon, "within", and χόνδρος/chondros, "cartilage"
- ^ "Etymology of the English word endochondral". myEtymology. Archived from the original on July 14, 2011.
{{cite web}}: CS1 maint: unfit URL (link) - ^ a b c Šromová, V; Sobola, D; Kaspar, P (5 November 2023). "A Brief Review of Bone Cell Function and Importance". Cells. 12 (21): 2576. doi:10.3390/cells12212576. PMC 10648520. PMID 37947654.
 This article incorporates text available under the CC BY 4.0 license.
This article incorporates text available under the CC BY 4.0 license.
- ^ Cowan, PT; Kahai, P (2023), "Anatomy, Bones", StatPearls, Treasure Island, Florida (FL): StatPearls Publishing, PMID 30725884
- ^ a b c Blumer, Michael J. F. (1 May 2021). "Bone tissue and histological and molecular events during development of the long bones". Annals of Anatomy - Anatomischer Anzeiger. 235: 151704. doi:10.1016/j.aanat.2021.151704. ISSN 0940-9602. PMID 33600952.
- ^ Sadler, T.W. (2023). Langman's medical embryology (15th ed.). Wolters Kluwer Health. ISBN 978-1975179960.
- ^ Hyland, S; Charlick, M; Varacallo, M (2023), "Anatomy, Shoulder and Upper Limb, Clavicle", StatPearls, Treasure Island, Florida FL): StatPearls Publishing, PMID 30252246
- ^ a b c d e f Pawlina, Wojciech (2024). Histology: a text and atlas: with correlated cell and molecular biology (9th ed.). Wolters Kluwer. ISBN 9781975181574.
- ^ a b Mescher, Anthony L. (2023). Junqueira's Basic Histology: Text and Atlas (17th ed.). McGraw-Hill Education. ISBN 978-1264930395.
- ^ a b c Richard, Marsell; Thomas A, Einhorn (1 June 2012). "The biology of fracture healing". Injury. 42 (6): 551–555. doi:10.1016/j.injury.2011.03.031. PMC 3105171. PMID 21489527.
- ^ a b c Chagin, AS; Chu, TL (December 2023). "The Origin and Fate of Chondrocytes: Cell Plasticity in Physiological Setting". Current Osteoporosis Reports. 21 (6): 815–824. doi:10.1007/s11914-023-00827-1. PMC 10724094. PMID 37837512.
- ^ Bahney, Chelsea S.; Hu, Diane P.; Miclau, Theodore; Marcucio, Ralph S. (5 February 2015). "The Multifaceted Role of the Vasculature in Endochondral Fracture Repair". Frontiers in Endocrinology. 6: 4. doi:10.3389/fendo.2015.00004. ISSN 1664-2392. PMC 4318416. PMID 25699016.





