GLUT2
| glucose transporter, type 2 | |||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Identifiers | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Aliases | Glc_transpt_2IPR002440GLUT2Glucosamine/glucose uniporterGlut-2Glucose Transporter Type 2 | ||||||||||||||||||||||||||||||||||||||||||||||||||
| External IDs | GeneCards: [1] | ||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| Wikidata | |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
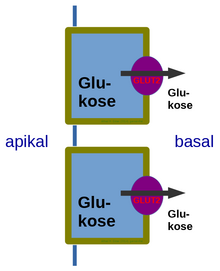
Glucose transporter 2 (GLUT2) also known as solute carrier family 2 (facilitated glucose transporter), member 2 (SLC2A2) is a transmembrane carrier protein that enables protein facilitated glucose movement across cell membranes. It is the principal transporter for transfer of glucose between liver and blood[1] Unlike GLUT4, it does not rely on insulin for facilitated diffusion.
In humans, this protein is encoded by the SLC2A2 gene.[2][3]
Tissue distribution[edit]
GLUT2 is found in cellular membranes of:
- liver (Primary)
- pancreatic β cell (Primary in mice, tertiary in humans after GLUT1 and GLUT3)[4]
- hypothalamus (Not overly significant)
- basolateral membrane of small intestine and apical GLUT2 is also suggested.[5]
- basolateral membrane of renal tubular cells[6][7]
Function[edit]
GLUT2 has high capacity for glucose but low affinity (high KM, ca. 15–20 mM) and thus functions as part of the "glucose sensor" in the pancreatic β-cells of rodents, though in human β-cells the role of GLUT2 seems to be a minor one.[8] It is a very efficient carrier for glucose.[9][10] Similarly, a recent study showed that lack of GLUT2 in β-cells doesn't impair glucose homeostasis or glucose-stimulated insulin secretion in mice.[11]
GLUT2 also carries glucosamine.[12]
When the glucose concentration in the lumen of the small intestine goes above 30 mM, such as occurs in the fed-state, GLUT2 is up-regulated at the brush border membrane, enhancing the capacity of glucose transport. Basolateral GLUT2 in enterocytes also aids in the transport of fructose into the bloodstream through glucose-dependent cotransport. Recent studies show that renal GLUT2 contributes to systemic glucose homeostasis by regulating glucose reabsorption.[7] Lack of renal Glut2 reversed features of diabetes and obesity in mice. In addition, renal Glut2 deficiency caused knockdown of renal Sglt2 through the transcription factor Hnf1α.[7]
Clinical significance[edit]
Defects in the SLC2A2 gene are associated with a particular type of glycogen storage disease called Fanconi-Bickel syndrome.[13]
In drug-treated diabetic pregnancies in which glucose levels in the woman are uncontrolled, neural tube and cardiac defects in the early-developing brain, spine, and heart depend upon functional GLUT2 carriers, and defects in the GLUT2 gene have been shown to be protective against such defects in rats.[14] However, whilst a lack of GLUT2 adaptability[15] is negative, it is important to remember the fact that the main result of untreated gestational diabetes appears to cause babies to be of above-average size, which may well be an advantage that is managed very well with a healthy GLUT2 status.
Maintaining a regulated osmotic balance of sugar concentration between the blood circulation and the interstitial spaces is critical in some cases of edema including cerebral edema.
GLUT2 appears to be particularly important to osmoregulation, and preventing edema-induced stroke, transient ischemic attack or coma, especially when blood glucose concentration is above average.[16] GLUT2 could reasonably be referred to as the "diabetic glucose transporter" or a "stress hyperglycemia glucose transporter."
SLC2A2 was associated with clinical stages and independently associated with overall survival in patients with Hepatocellular carcinoma, and could be considered a new prognostic factor for HCC.[17]
Interactive pathway map[edit]
Click on genes, proteins and metabolites below to link to respective articles.[§ 1]
- ^ The interactive pathway map can be edited at WikiPathways: "GlycolysisGluconeogenesis_WP534".
See also[edit]
References[edit]
- ^ Gwyn W. Gould; Helen M. Thomas; Thomas J. Jess; Graeme I. Bell (May 1991). "Expression of human glucose transporters in Xenopus oocytes: kinetic characterization and substrate specificities of the erythrocyte, liver, and brain isoforms". Biochemistry. 30 (21): 5139–5145. doi:10.1021/bi00235a004. PMID 2036379.
- ^ Froguel P, Zouali H, Sun F, Velho G, Fukumoto H, Passa P, Cohen D (July 1991). "CA repeat polymorphism in the glucose transporter GLUT 2 gene". Nucleic Acids Research. 19 (13): 3754. doi:10.1093/nar/19.13.3754-a. PMC 328421. PMID 1852621.
- ^ Uldry M, Thorens B (February 2004). "The SLC2 family of facilitated hexose and polyol transporters" (PDF). Pflügers Archiv. 447 (5): 480–9. doi:10.1007/s00424-003-1085-0. PMID 12750891. S2CID 25539725.
- ^ McCulloch LJ, van de Bunt M, Braun M, Frayn KN, CLark A, Gloyn AL (December 2011). "GLUT2 (SLC2A2) is not the principal glucose transporter in human pancreatic beta cells: implications for understanding genetic association signals at this locus". Molecular Genetics and Metabolism. 104 (4): 648–53. doi:10.1016/j.ymgme.2011.08.026. PMID 21920790.
- ^ Kellett GL, Brot-Laroche E (October 2005). "Apical GLUT2: a major pathway of intestinal sugar absorption". Diabetes. 54 (10): 3056–62. doi:10.2337/diabetes.54.10.3056. PMID 16186415.
- ^ Freitas HS, Schaan BD, Seraphim PM, Nunes MT, Machado UF (June 2005). "Acute and short-term insulin-induced molecular adaptations of GLUT2 gene expression in the renal cortex of diabetic rats". Molecular and Cellular Endocrinology. 237 (1–2): 49–57. doi:10.1016/j.mce.2005.03.005. PMID 15869838. S2CID 44856595.
- ^ a b c de Souza Cordeiro, Leticia Maria; Bainbridge, Lauren; Devisetty, Nagavardhini; McDougal, David H.; Peters, Dorien J. M.; Chhabra, Kavaljit H. (2022). "Loss of function of renal Glut2 reverses hyperglycaemia and normalises body weight in mouse models of diabetes and obesity". Diabetologia. 65 (6): 1032–1047. doi:10.1007/s00125-022-05676-8. PMC 9081162. PMID 35290476.
- ^ McCulloch LJ, van de Bunt M, Braun M, Frayn KN, Clark A, Gloyn AL (December 2011). "GLUT2 (SLC2A2) is not the principal glucose transporter in human pancreatic beta cells: implications for understanding genetic association signals at this locus". Molecular Genetics and Metabolism. 104 (4): 648–53. doi:10.1016/j.ymgme.2011.08.026. PMID 21920790.
- ^ Guillam MT, Hümmler E, Schaerer E, Yeh JI, Birnbaum MJ, Beermann F, Schmidt A, Dériaz N, Thorens B, Wu JY (November 1997). "Early diabetes and abnormal postnatal pancreatic islet development in mice lacking Glut-2". Nature Genetics. 17 (3): 327–30. doi:10.1038/ng1197-327. PMID 9354799. S2CID 37328600.
- ^ Efrat S (November 1997). "Making sense of glucose sensing". Nature Genetics. 17 (3): 249–50. doi:10.1038/ng1197-249. PMID 9354775. S2CID 13219161.
- ^ Bathina, Siresha; Faniyan, Tumininu S.; Bainbridge, Lauren; Davis, Autumn; Chhabra, Kavaljit H. (2023). "Normal β-Cell Glut2 Expression Is not Required for Regulating Glucose-Stimulated Insulin Secretion and Systemic Glucose Homeostasis in Mice". Biomolecules. 13 (3): 540. doi:10.3390/biom13030540. PMC 10046365. PMID 36979475.
- ^ Uldry M, Ibberson M, Hosokawa M, Thorens B (July 2002). "GLUT2 is a high affinity glucosamine transporter". FEBS Letters. 524 (1–3): 199–203. doi:10.1016/S0014-5793(02)03058-2. PMID 12135767. S2CID 40913482.
- ^ Santer R, Groth S, Kinner M, Dombrowski A, Berry GT, Brodehl J, Leonard JV, Moses S, Norgren S, Skovby F, Schneppenheim R, Steinmann B, Schaub J (January 2002). "The mutation spectrum of the facilitative glucose transporter gene SLC2A2 (GLUT2) in patients with Fanconi-Bickel syndrome". Human Genetics. 110 (1): 21–9. doi:10.1007/s00439-001-0638-6. PMID 11810292. S2CID 1767168.
- ^ Li R, Thorens B, Loeken MR (March 2007). "Expression of the gene encoding the high-Km glucose transporter 2 by the early postimplantation mouse embryo is essential for neural tube defects associated with diabetic embryopathy". Diabetologia. 50 (3): 682–9. doi:10.1007/s00125-006-0579-7. PMID 17235524.
- ^ Thomson AB, Wild G (March 1997). "Adaptation of intestinal nutrient transport in health and disease. Part I". Digestive Diseases and Sciences. 42 (3): 453–69. doi:10.1023/A:1018807120691. PMID 9073126. S2CID 25371741.
- ^ Stolarczyk E, Le Gall M, Even P, Houllier A, Serradas P, Brot-Laroche E, Leturque A (December 2007). Maedler K (ed.). "Loss of sugar detection by GLUT2 affects glucose homeostasis in mice". PLOS ONE. 2 (12): e1288. Bibcode:2007PLoSO...2.1288S. doi:10.1371/journal.pone.0001288. PMC 2100167. PMID 18074013.

- ^ Kim, Yun Hak; Jeong, Dae Cheon; Pak, Kyoungjune; Han, Myoung-Eun; Kim, Ji-Young; Liangwen, Liu; Kim, Hyun Jin; Kim, Tae Woo; Kim, Tae Hwa; Hyun, Dong Woo; Oh, Sae-Ock (2017-09-15). "SLC2A2 (GLUT2) as a novel prognostic factor for hepatocellular carcinoma". Oncotarget. 8 (40): 68381–68392. doi:10.18632/oncotarget.20266. ISSN 1949-2553. PMC 5620264. PMID 28978124.
External links[edit]
- Glucose+Transporter+Type+2 at the U.S. National Library of Medicine Medical Subject Headings (MeSH)

