Gamma helix

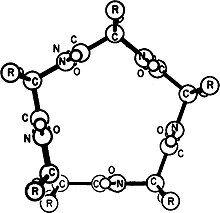
Gamma helix (or γ-helix)[2][3] is a type of secondary structure in proteins that has been predicted by Pauling, Corey, and Branson,[1][4] but has never been observed in natural proteins.[3] The hydrogen bond in this type of helix was predicted to be between N-H group of one amino acid and the C=O group of the amino acid six residues earlier (or, as described by Pauling, Corey, Branson, "to the fifth amide group beyond it"). This can also be described as i + 6 → i bond and would be a continuation of the series (310 helix, alpha helix, pi helix and gamma helix). This theoretical helix contains 5.1 residues per turn.[1]However, a fully developed gamma helix has characteristics of a structure that has 2.2 amino acid residues per turn, a rise of 2.75Å per residue, and a pseudo-cyclic (C7) structure closed by intramolecular H-bond. Depending on the amino acid's side chain (R) involved in this main-chain reversal motif, two stereoisomers can occur with their Cα-substituent located either in the axial or in the equatorial position relative to the H-bonded pseudo-cycle.[5]
References[edit]
- ^ a b c d Pauling L, Corey RB, Branson HR (April 1951). "The structure of proteins; two hydrogen-bonded helical configurations of the polypeptide chain". Proceedings of the National Academy of Sciences of the United States of America. 37 (4): 205–211. Bibcode:1951PNAS...37..205P. doi:10.1073/pnas.37.4.205. PMC 1063337. PMID 14816373.
- ^ Dunitz JD (November 2001). "Pauling's Left-Handed α-Helix". Angewandte Chemie International Edition in English. 40 (22): 4167–4173. doi:10.1002/1521-3773(20011119)40:22<4167::AID-ANIE4167>3.0.CO;2-Q. PMID 29712120.
- ^ a b Berndt KD (May 1996). "3.1 Helices". Stockholm, Sweden: Karolinska Institute. Retrieved 2022-03-21 – via www.cryst.bbk.ac.uk.
- ^ Pauling L, Corey RB (1950). "Two hydrogen-bonded spiral configurations of the polypeptide chain". Journal of the American Chemical Society. 72 (11): 5349. doi:10.1021/ja01167a545. ISSN 0002-7863.
- ^ Mazzier, Daniela; Crisma, Marco; Grassi, Luigi; Moretto, Alessandro; Alemán, Carlos; Formaggio, Fernando; Toniolo, Claudio (22 December 2016). "En route towards the peptide γ-helix: X-ray diffraction analyses and conformational energy calculations of Adm-rich short peptides". Peptide Science. 23 (4): 346–362. doi:10.1002/psc.2957. hdl:2117/105291.
