From Wikipedia, the free encyclopedia
Chemical compound
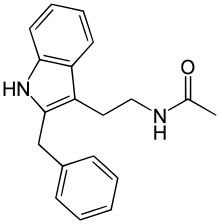
Luzindole (N-0774 ), (N -acetyl-2-benzyltryptaminedrug used in scientific research to study the role of melatonin in the body. Luzindole acts as a selective melatonin receptor antagonist ,[1] affinity for the MT2 over the MT1 receptor .[2] [3] circadian rhythm as well as produce antidepressant effects.[2] [4]
References [ edit ]
^ Dubocovich ML (September 1988). "Luzindole (N-0774): a novel melatonin receptor antagonist" . The Journal of Pharmacology and Experimental Therapeutics . 246 (3): 902–10. PMID 2843633 . ^ a b Dubocovich ML, Yun K, Al-Ghoul WM, Benloucif S, Masana MI (September 1998). "Selective MT2 melatonin receptor antagonists block melatonin-mediated phase advances of circadian rhythms" . The FASEB Journal . 12 (12): 1211–20. doi :10.1096/fasebj.12.12.1211 PMID 9737724 . S2CID 566199 . ^ Browning C, Beresford I, Fraser N, Giles H (March 2000). "Pharmacological characterization of human recombinant melatonin mt(1) and MT(2) receptors" . British Journal of Pharmacology 129 (5): 877–86. doi :10.1038/sj.bjp.0703130 . PMC 1571913 PMID 10696085 . ^ Dubocovich ML, Mogilnicka E, Areso PM (July 1990). "Antidepressant-like activity of the melatonin receptor antagonist, luzindole (N-0774), in the mouse behavioral despair test". European Journal of Pharmacology . 182 (2): 313–25. doi :10.1016/0014-2999(90)90290-M . PMID 2168835 .
MT1 Tooltip Melatonin receptor 1 MT2 Tooltip Melatonin receptor 2 Unsorted
1-Methylpsilocin 2,alpha-DMT 2-Me-DET 2-Methyl-5-HT 2,N,N-TMT 4,5-DHP-DMT 4,5-MDO-DMT 4,5-MDO-DiPT 4-AcO-DALT 4-AcO-DET 4-AcO-DMT 4-AcO-DiPT 4-AcO-EPT 4-AcO-NMT 4-AcO-MALT 4-AcO-MET 4-AcO-DPT 4-AcO-MiPT 4-F-5-MeO-DMT 4-HO-5-MeO-DMT 4-HO-DALT 4-HO-DBT 4-HO-DET 4-HO-DiPT 4-HO-DPT 4-HO-DSBT 4-HO-EPT 4-HO-MALT 4-HO-MET 4-HO-McPT 4-HO-McPeT 4-HO-MiPT 4-HO-MPMI 4-HO-MPT 4-HO-MsBT 4-HO-NMT 4-HO-PiPT 4-HO-pyr-T 4-HO-αMT 4-Me-αET 4-Me-αMT 4-MeO-DiPT 4-MeO-DMT 4-MeO-MiPT 4-PrO-DMT 5,6-MeO-MiPT 5,6-MDO-DiPT 5,6-MDO-DMT 5,6-MDO-MiPT 5,7-Dihydroxytryptamine 5-BT 5-Bromo-DMT 5-CT 5-Chloro-αMT 5-Chloro-DMT 5-Ethoxy-αMT 5-Ethoxy-DMT 5-Ethyl-DMT 5-Fluoro-AET 5-Fluoro-αMT 5-Fluoro-DET 5-Fluoro-DMT 5-Fluoro-EPT 5-Fluoro-MET 5-HO-αMT 5-HO-DiPT 5-HTP 5-iPrO-AMT 5-MeS-DMT 5-Methoxytryptamine 5-MeO-7,N,N-TMT 5-Methyl-αET 5-MeO-2-TMT 5-MeO-αET 5-MeO-αMT 5-MeO-DALT 5-MeO-DBT 5-MeO-DET 5-MeO-DiPT 5-MeO-DMT 5-MeO-DPT 5-MeO-EiPT 5-MeO-EPT 5-MeO-MALT 5-MeO-MET 5-MeO-MiPT 5-MeO-MPMI 5-MeO-NMT 5-MeO-pyr-T 5-MeO-NBpBrT 5-Methyl-DMT 5-(Nonyloxy)tryptamine 6-Fluoro-αMT 6-Fluoro-DMT 6-Hydroxymelatonin 6-MeO-THH 7-Chloro-AMT 7-Methyl-α-ethyltryptamine 7-Methyl-DMT Acetryptine Aeruginascin αET Alpha,N-DMT α,N,N-Trimethyltryptamine Alpha,N,O-TMS AL-37350A αMT Baeocystin BNC-210 Bufotenidine Bufotenin (5-HO-DMT) BW-723C86 Convolutindole A CP-132,484 DALT DBT Desformylflustrabromine DET DiPT DPT E-6801 E-6837 Ethocybin EiPT EMDT EPT FGIN-127 FGIN-143 Harmaline HIOC Ibogaine Idalopirdine Indorenate Iprocin Lespedamine Luzindole MET Methylbutyltryptamine MiPT MPT Miprocin Melatonin MPMI MS-245 NAS N-Ethyltryptamine N-Feruloylserotonin NMT DMT Norbaeocystin Normelatonin N-t-Butyltryptamine O-4310 Oxypertine Plakohypaphorine PiPT Psilocin (4-HO-DMT) Psilocybin (4-PO-DMT) Pyr-T Rizatriptan RU-28306 Serotonin ST-1936 Sumatriptan Tryptamine Tryptophan Yohimbine Yuremamine Zolmitriptan