Microcotyle
| Microcotyle | |
|---|---|
| |
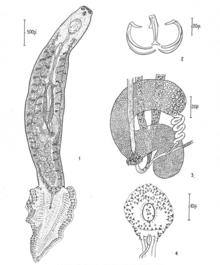
| Microcotyle donavini (Type-species of Microcotyle) | |
| Scientific classification | |
| Domain: | Eukaryota |
| Kingdom: | Animalia |
| Phylum: | Platyhelminthes |
| Class: | Monogenea |
| Order: | Mazocraeidea |
| Family: | Microcotylidae |
| Subfamily: | Microcotylinae |
| Genus: | Microcotyle Van Beneden & Hesse, 1863 |
Microcotyle is a genus which belongs to the phylum Platyhelminthes and class Monogenea. Species of Microcotyle are ectoparasites that affect their host by attaching themselves as larvae on the gills of the fish and grow into adult stage. This larval stage is called oncomiracidium, and is characterized as free swimming and ciliated.
Species of Microcotyle have only one host in their entire life cycle. Different species of Microcotyle inhabit marine and freshwater and they can also infect different species of fishes. They are uniquely characterized by their haptor having a lot of tiny clamps on the lateral margins.
Although not really known to cause that much damage in marine wildlife, some of them are reported to cause agricultural problems like Microcotyle sebastis as an example. Microcotyle sebastis commonly affects Sebastes schlegeli, a maricultured fish in Korea.[1]
Morphology[edit]
Individuals of species of Microcotyle, like any typical polyopisthocotylean monogenean, have an anterior organ called prohaptor, which is mainly used for feeding and attachment. Although the prohaptor is not the primary attachment organ, it is used to anchor the body of the parasite while the opisthaptor is being repositioned. The opisthaptor is found in all Microcotyle species and it is an important attachment organ that allows these ectoparasites to latch onto their hosts.
Located at the anterior part of the worm is the funnel-shaped mouth that is connected to the pharynx, followed by the esophagus that is smaller in diameter compared to the pharynx. About one tenth of the entire length of the worm, the esophagus then divides into intestinal crura, which extends further posteriorly. The cruca is divided into pouches, which extend between vitellaria. Vitallaria are glands that secrete yolk around the egg. This digestive pathway is observed to be continuous throughout the entire worm. Food particles were observed to be passing back and forth along these pathways in vivo.[2]
The worms are hermaphroditic, containing both male and female organs. Each worm has reproductive organs such as vas deferens, testis, uterus, vitelline duct, ovary, and vitellaria.[2] They also have flame cells that function as a kidney and remove waste material. A short duct that opens to the outside on the dorsal surface is composed of four canals on each side, two posterior and two anterior, that come together laterally to the cirrus. During observation on immature specimens, these canals can still be seen. However, on adults, these canals are concealed by vitellaria.[2]
Life cycle[edit]
Species of Microcotyle have a direct life cycle; no intermediate hosts are needed to complete its life cycle. Marine fishes are the definitive host of these parasites. The adults live on the gill of the fishes as ectoparasites. The eggs are released into water, hatch and develop as oncomiracidium. The oncomiracidia, free swimming form larvae, move around and attach to another fish and grow into adult stage. The life cycle of species of Microcotyle is completed and repeated again and again.
Treatment[edit]
Bath treatment using praziquantel is the effective methods of treating Microcotyle sebastis on rockfish.[3] An experiment was done to indicate that feeding a praziquantel-adsorbed diet significantly reduces the abundance of M. sebastis infestation, and bathing in 100 ppm praziquantel for 4 minutes is effective for controlling M. sebastis infestation in a practical rockfish culture system.[1]
Species[edit]
According to the World Register of Marine Species,[4] there are more than one hundred species in this genus:
- Microcotyle aigoi Ishii & Sawada, 1938 [5]
- Microcotyle algeriensis Ayadi, Gey, Justine & Tazerouti, 2017 [6]
- Microcotyle angelichthys MacCallum, 1913 [7]
- Microcotyle archosargi MacCallum, 1913 [7]
- Microcotyle argenticus Hadi & Bilqees, 2011 [8]
- Microcotyle arripis Sandars, 1945 [9]
- Microcotyle bassensis Murray, 1931 [10]
- Microcotyle bothi Yamaguti, 1968 [11]
- Microcotyle branchiostegi Yamaguti, 1937 [12]
- Microcotyle brevis Dillon & Hargis, 1965 [13]
- Microcotyle caudata Goto, 1984 [14]
- Microcotyle centrodonti Brown, 1929 [15]
- Microcotyle centropristis MacCallum, 1915.[16]
- Microcotyle cepolae Yamaguti, 1938 [17]
- Microcotyle constricta Robinson, 1961 [18]
- Microcotyle danielcarrioni (Martinez & Barrantes, 1977) Bouguerche, Gey, Justine and Tazerouti, 2019 [19]
- Microcotyle ditrematis Yamaguti, 1940 [20]
- Microcotyle donavini Van Beneden & Hesse, 1863 [21][22]
- Microcotyle elegans Goto, 1894 [14]
- Microcotyle emmelichthyops Yamaguti, 1968 [11]
- Microcotyle erythrini Van Beneden & Hesse, 1863 [21]
- Microcotyle eueides MacCallum & MacCallum, 1913 [23]
- Microcotyle fistulariae Mamaev, 1989 [24]
- Microcotyle furcata Linton, 1940 [25]
- Microcotyle fusiformis Goto, 1894 [14]
- Microcotyle gimpo Yamaguti, 1958 [26]
- Microcotyle guanabarensis Bravo-Hollis & Kohn, 1990 [27]
- Microcotyle hainanensis [28]
- Microcotyle helotes Sandars, 1944 [29]
- Microcotyle hiatulae Goto, 1899 [30]
- Microcotyle inglisi Gupta & Krishna, 1980 [31]
- Microcotyle jonii Hadi, Bilqees & Khatoon, 2011[32]
- Microcotyle justinei Ayadi & Tazerouti, 2023 [33]
- Microcotyle korathai Gupta & Krishna, 1980 [31]
- Microcotyle lichiae Ariola, 1899 [34]
- Microcotyle longirostri Robinson, 1961 [18]
- Microcotyle macropharynx Mamaev, 1989 [24]
- Microcotyle mouwoi Ishii & Sawada, 1938 [35]
- Microcotyle moyanoi (Villalba & Fernandes, 1986) Bouguerche, Gey, Justine and Tazerouti, 2019 [36]
- Microcotyle nemadactylus Dillon & Hargis, 1965 [13]
- Microcotyle neozealanica Dillon & Hargis, 1965 [13]
- Microcotyle oceanica Caballero, Bravo-Hollis & Grocott, 1953 [37]
- Microcotyle odacis Sandars, 1945 [9]
- Microcotyle omani Machkewskyi, Dmitrieva, Al-Jufaili & Al-Mazrooei, 2013 [38]
- Microcotyle otrynteri Pearse, 1949 [39]
- Microcotyle pacifica Crane, 1972 [40]
- Microcotyle pempheri Machida & Azaki, 1977 [41]
- Microcotyle pentapodi Sandars, 1944 [29]
- Microcotyle peprili Pearse, 1949 [39]
- Microcotyle polymixiae Yamaguti, 1968 [11]
- Microcotyle pomacanthi MacCallum, 1915 [16]
- Microcotyle pomatomi Goto, 1899 [30]
- Microcotyle pontica Pogorel'tseva, 1964 [42]
- Microcotyle poronoti MacCallum, 1915 [16]
- Microcotyle pseudopercis Amato & Cezar, 1994 [43]
- Microcotyle rubrum Hadi & Bilqees, 2010 [44]
- Microcotyle sebastis Goto, 1894 [14]
- Microcotyle sebastisci Yamaguti, 1958 [26]
- Microcotyle stenotomi Goto, 1899 [30]
- Microcotyle tampicensis (Caballero & Bravo-Hollis, 1972) Mamaev, 1986 [45]
- Microcotyle tanago Yamaguti, 1940 [20]
- Microcotyle toba Ishii & Sawada, 1938 [35]
- Microcotyle victoriae Woolcock, 1936 [46]
- Microcotyle visa Bouguerche, Gey, Justine & Tazerouti, 2019 [47]
- Microcotyle zalembius Crane, 1972 [40]
References[edit]
- ^ a b Kim, Ki. H, and Eun. Lee, and Se. Kwon, and Jae. Cho. (2001). Treatment of Microcotyle sebastis infestation in cultured rockfish Sebastes schlegeli by oral administration of praziquantel in combination with cimetidine. Diseases of Aquatic Organisms, 44 (2), pp. 133–136
- ^ a b c Remley, L. W. 1942. Morphology and life history studies of Microcotyle spinicirrus MacCallum 1918, a Monogenetic Trematode parasitic on the gills of Aplodinotus grunniens. Transactions of the American Microscopical Society, Vol. 61, No. 2 (Apr., 1942), pp141–155
- ^ Kim, Ki. H, and Jae. Cho. (2000). Treatment of Microcotyle sebastis (Monogenea: Polyopisthocotylea) infestation with praziquantel in an experimental cage simulating commercial rockfish Sebastes schlegeli culture conditions.. Diseases of Aquatic Organisms, Vol.40: 229-231, pp. 229–231
- ^ WoRMS (2018). Microcotyle Van Beneden & Hesse, 1863. Accessed at: http://www.marinespecies.org/aphia.php?p=taxdetails&id=119381 on 2018-11-29
- ^ 石井信太郎・澤田利貞 (1937). 外部寄生性吸蟲類ノ研究. 日本寄生虫学会記事 9: 93-97. (Ishii, N. and Sawada, T. (1937). [Studies on the ectoparasitic trematodes]. Nihon Kiseichū Gakkai Kiji 9: 3-97. [In Japanese])
- ^ Ayadi, Zouhour El Mouna; Gey, Delphine; Justine, Jean-Lou; Tazerouti, Fadila (2017). "A new species of Microcotyle (Monogenea: Microcotylidae) from Scorpaena notata (Teleostei: Scorpaenidae) in the Mediterranean Sea" (PDF). Parasitology International. 66 (2): 37–42. doi:10.1016/j.parint.2016.11.004. ISSN 1383-5769. PMID 27840197.
- ^ a b MacCallum, G.A. (1913) Further notes on the genus Microcotyle. Zool Jahrb Jena, 35, 389-402.
- ^ Hadi, R. & Bilqees, F.M. (2011) Microcotyle argenticus n. sp. (Monogenea) on the fish Pampus argenticus from Karachi Coast. Proceedings of Parasitology, 51, 99-111.
- ^ a b Sandars, D. F. (1945). Five new microcotylids from fish from Western Australian waters. Journal of the Royal Society of Western Australia, 29, 107–135.
- ^ Murray, Florence V. (1931). "Gill Trematodes from some Australian Fishes". Parasitology. 23 (4): 492–506. doi:10.1017/S0031182000013883. ISSN 0031-1820. S2CID 85731699.
- ^ a b c Yamaguti, S. (1968). Monogenetic trematodes of Hawaiian fishes. Honolulu: University of Hawaii Press. ISBN 978-0870228919
- ^ Yamaguti, S. (1937a). Studies on the Helminth Fauna of Japan. Part 17. Trematodes From a Marine Fish, Branchiostegus japonicus (Houttuyn). Published by the author. pp. 1–19.
- ^ a b c Dillon, W. A., & Hargis, W. J. (1965). Monogenetic trematodes from the southern Pacific Ocean. 2. Polyopisthocotyleids from New Zealand fishes: the families Discocotylidae, Microcotylidae, Axinidae and Gastrocotylidae. Antarctic Research Series, 5, 251-280.
- ^ a b c d Goto, Seitaro. (1894). "Studies on the ectoparasitic Trematodes of Japan". doi:10.5962/bhl.title.56506. hdl:2027/coo.31924003409327.
{{cite journal}}: Cite journal requires|journal=(help) - ^ Brown, Eleanor M. (1929). "On a new species of Monogenetic Trematode from the gills of Pagellus centrodontus". Proceedings of the Zoological Society of London. 99 (1): 67–83. doi:10.1111/j.1469-7998.1929.tb07687.x. ISSN 0370-2774.
- ^ a b c MacCallum, 1915. Notes on the genus Microcotyle III. Zoologische Jahrbücher. Abteilung für Systematik, Geographie und Biologie der Tiere. 38, 71-78
- ^ Yamaguti, S. (1938). Studies on the helminth fauna of Japan. Part 24. Trematodes of fishes, V. Japanese Journal of Zoology, 8(1), 15-74.
- ^ a b Robinson, Edward S. (1961). "Some Monogenetic Trematodes from Marine Fishes of the Pacific". Transactions of the American Microscopical Society. 80 (3): 235–266. doi:10.2307/3223640. ISSN 0003-0023. JSTOR 3223640.
- ^ Martinez, R., & Barrantes, I. (1977). Helmintos paràsitos de Cheilodactylus variegatus Valenciennes, 1833, ‘‘pintadilla’’. I. Paramicrocotyle danielcarrioni n. sp. (Monogenea: Microcotylidae). Biota (Lima), 11, 139–146
- ^ a b Yamaguti, S. (1940). Studies on the helminth fauna of Japan. Part 31. Trematodes of fishes, VII. Japanese Journal of Zoology 9: 35–108, 2 pls.
- ^ a b Van Beneden, P. J., & Hesse, C. E. (1863). Recherches sur les Bdellodes (Hirudinées) et les trématodes marins: Mémoire présenté à l'Académie Royale de Belgique. Mémoires de l'Académie Royale des Sciences, des Lettres et des Beaux-Arts de Belgique, 34, 1-150 + Plates.
- ^ Euzet, Louis; Marc, Annie (1963). "Microcotyle donavini Van Beneden et Hesse 1863 espèce type du genre Microcotyle Van Beneden et Hesse 1863". Annales de Parasitologie Humaine et Comparée. 38 (6): 875–886. doi:10.1051/parasite/1963386875. ISSN 0003-4150. PMID 14152058.

- ^ MacCallum, G. A., & MacCallum, W. G. (1913). Four species of Microcotyle, M. pyragraphorus, macroura, eueides and acanthophallus. Zoologische Jahrbücher Abteilung für Systematik, 34, 223-244.
- ^ a b Mamaev, Yu.L. (1989) [On species composition and morphological features of the Microcotyle genus (Microcotylidae, Monogenoidea)]. In: Lebedev, B.I. (Ed.) [Investigation in parasitology, collection of papers]. Vladivostok: DVNTs SSSR, 167 pp. (In Russian)
- ^ Linton, E. (1940). Trematodes from fishes mainly from the Woods Hole region, Massachusetts. Proceedings of the United States National Museum, 88, 1-172.
- ^ a b Yamaguti, S. (1958). Studies on the helminth fauna of Japan. Part 53. Trematodes of fishes, XII. Publications of the Seto Marine Biological Laboratory, 7, 53-88.
- ^ Bravo-Hollis, M., & Kohn, A. (1990). Una especie nueva del género Microcotyle (Monogenea: Microcotylinae) de las branquias de Eucinostomus argenteus (Baird and Girard, 1854) de Rio de Janeiro, Brasil. Anales del Instituto de Biología. UNAM. Serie zoología
- ^ Zhang J.Y., Yang T.B. & Liu L. (2001). Monogeneans of Chinese marine fishes. Beijing: Agriculture Press, 400 pp.
- ^ a b Sandars, D. F. (1944). A contribution to the knowledge of the Microcotylidae of Western Australia. Transactions of the Royal Society of South Australia, 68(1), 67-81. PDF in BHL

- ^ a b c Goto, S. (1899). Notes on some exotic species of ectoparasitic trematode. The journal of the College of Science, Imperial University of Tokyo, Japan = Tokyo Teikoku Daigaku kiyo. Rika. Tokyo, Japan: The University, 1898-1925. Volume 12, pp. 263-295 + Plates (foldout) Whole volume, PDF in BHL

- ^ a b Gupta, S. P. & Krishna (1980) On some monogenetic trematodes from marine fishes of Puri, Orissa. Indian Journal of Helminthology 31, 81–110. Reference in Bionames
- ^ Rana, H., Bilqees, F. M., & Nasira, K. (2011). Microcotyle jonii n. sp. of (Monogenea: Microcotylidae) in the fish Lutjanus jonii from Karachi coast. Proceedings of Parasitology, (52), 69-81.
- ^ Ayadi, Zouhour El Mouna; Tazerouti, Fadila (2023). "Microcotyle justinei n. sp. (Monogenea: Microcotylidae) from the gills of the Cardinal Fish Apogon imberbis (Teleostei: Apogonidae) off the Algerian Coast of the Western Mediterranean". Acta Parasitologica. 68 (4): 842–852. doi:10.1007/s11686-023-00722-w. ISSN 1230-2821.
- ^ Ariola, V. (1899). Di alcuni trematodi di pesci marini. Bollettino dei Musei e laboratori di zoologia e anatomia comparata della R. Università di Genova, 4(81), 1-12.
- ^ a b Ishii N., Sawada T. 1938: Studies on the ectoparasitic trematodes. In: Livro Jubilar Prof. Travassos, Rio de Janeiro, Brasil, III, pp. 231–243.
- ^ Villalba, C., & Fernàndez, J. (1986). Dos nuevas especies de tremàtodos paràsitos de peces marinos en Chile. Parasitologia al Dia, 10, 45–51.
- ^ Caballero, C. E., Bravo-Hollis, M., Grocott, R.G. Helmintos de la República de Panamá. VII. Descripción de algunos tremátodos de peces marinos. Anales del Instituto de Biologia, 1953, vol. 24, p. 97-136.
- ^ Machkewskyi, Volodymyr K.; Dmitrieva, Evgenija V.; Al-Jufaili, Sara; Al-Mazrooei, Nashwa A. M. (2013). "Microcotyle omanae n. sp. (Monogenea: Microcotylidae), a parasite of Cheimerius nufar (Valenciennes) (Sparidae) from the Arabian Sea". Systematic Parasitology. 86 (2): 153–163. doi:10.1007/s11230-013-9444-5. ISSN 0165-5752. PMID 24048747. S2CID 15324209.
- ^ a b Pearse, A. S. (1949). "Observations on Flatworms and Nemerteans Collected at Beaufort, N. C". Proceedings of the United States National Museum. 100 (3255): 25–38, figures 6–9. doi:10.5479/si.00963801.100-3255.25. ISSN 0096-3801.

- ^ a b Crane, J. W. 1972. Systematics and new species of marine monogenea from California. Wasmann Journal of Biology 30:109-166
- ^ Machida, M. & Azaki, J., 1977, Monogenetic and digenetic trematodes of the sweeper, Pempheris xanthoptera. Bulletin of the National Science Museum, Tokyo, s. A, Zoology, v. 3 (1), 1-7 PDF

- ^ Pogorel'tseva, T. P. (1964). New and little known species of monogenetic trematodes of fish in the Black Sea. Problemy Parazitologii (Trudy ukr. respubl. nauch. Obshch. Parazit.), 3, 30-42.
- ^ Amato, J. F. R., & Cezar, A. D. (1994). A new species of Microcotyle van Beneden & Hesse, 1863, parasitic of ‘‘namorado”, Pseudopercis numida Ribeiro, 1903 and O. semifasciata (Cuvier, 1829), from the coast of the State of Rio de Janeiro. Brazil. Rev. Bras. Parasitol. Vet, 3, 41-44.
- ^ Hadi, R., & Bilqees, F. M. (2010). Microcotyle rubrum n. sp (Monogenea: Microcotylidae) from Otolithus ruber of Karachi coast, Pakistan. Proceedings of Parasitology, (50), 115-126.
- ^ Caballero y Caballero, E. & Bravo-Hollis, M. (1972) Monogenea (van Beneden, 1858) Carus, 1863 de peces marinos del litoral Mexicano del Golfo de Mexico y del Mar Caribe. Revista de Biología Tropical, 20, 151-165.
- ^ Woolcock, Violet (1938). "Monogenetic Trematodes from some Australian Fishes". Parasitology. 28 (1): 79–91. doi:10.1017/S0031182000022277. ISSN 0031-1820. S2CID 251058449.
- ^ Bouguerche, Chahinez; Gey, Delphine; Justine, Jean-Lou; Tazerouti, Fadila (2019). "Microcotyle visa n. sp. (Monogenea: Microcotylidae), a gill parasite of Pagrus caeruleostictus (Valenciennes) (Teleostei: Sparidae) off the Algerian coast, Western Mediterranean" (PDF). Systematic Parasitology. 96 (2): 131–147. doi:10.1007/s11230-019-09842-2. ISSN 0165-5752. PMID 30701382. S2CID 59526278. (subscription required)
