Nef reaction
| Nef reaction | |
|---|---|
| Named after | John Ulric Nef |
| Reaction type | Substitution reaction |
| Identifiers | |
| Organic Chemistry Portal | nef-reaction |
| RSC ontology ID | RXNO:0000157 |
In organic chemistry, the Nef reaction is an organic reaction describing the acid hydrolysis of a salt of a primary or secondary nitroalkane (R−NO2) to an aldehyde (R−CH=O) or a ketone (R2C=O) and nitrous oxide (N2O). The reaction has been the subject of several literature reviews.[1][2][3]

1. Salt of a nitroalkane
3. Ketone
4. Nitrous oxide
The reaction was reported in 1894 by the chemist John Ulric Nef,[4] who treated the sodium salt of nitroethane with sulfuric acid resulting in an 85–89% yield of nitrous oxide and at least 70% yield of acetaldehyde. However, the reaction was pioneered a year earlier in 1893 by Konovalov,[5] who converted the potassium salt of 1-phenylnitroethane with sulfuric acid to acetophenone.
Reaction mechanism[edit]
The reaction mechanism starting from the nitronate salt as the resonance structures 1a and 1b is depicted below:
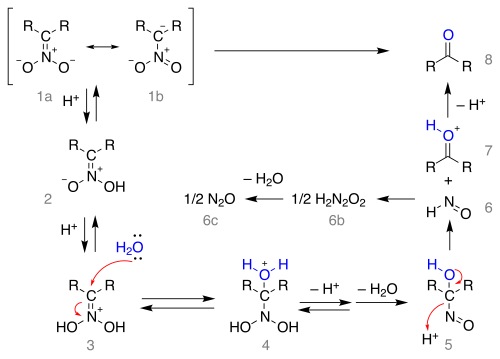
The salt is protonated forming the nitronic acid 2 (in some cases these nitronates have been isolated) and once more to the iminium ion 3. This intermediate is attacked by water in a nucleophilic addition forming 4 which loses a proton and then water to the 1-nitroso-alkanol 5 which is believed to be responsible for the deep-blue color of the reaction mixture in many Nef reactions. This intermediate rearranges to hyponitrous acid 6 (forming nitrous oxide 6c through 6b) and the oxonium ion 7 which loses a proton to form the carbonyl compound.
Note that formation of the nitronate salt from the nitro compound requires an alpha hydrogen atom and therefore the reaction fails with tertiary nitro compounds.
Scope[edit]
Nef-type reactions are frequently encountered in organic synthesis, because they turn the Henry reaction into a convenient method for functionalization at the β and γ locations.[6] Thus, for example, the reaction is combined with the Michael reaction in the synthesis of the γ-keto-carbonyl methyl 3-acetyl-5-oxohexanoate, itself a cyclopentenone intermediate:[7][8]

In carbohydrate chemistry, they are a chain-extension method for aldoses, as in the isotope labeling of C14-D‑mannose and C14-D‑glucose from D‑arabinose and C14‑nitromethane (the first step here is a Henry reaction):

The opposite reaction is the Wohl degradation.
Variants[edit]
Nef's original protocol, using concentrated sulfuric acid, has been described as "violent".[9] Strong-acid hydrolysis without the intermediate salt stage results in the formation of carboxylic acids and hydroxylamine salts,[citation needed] but Lewis acids such as tin(IV) chloride[10] and iron(III) chloride[11] give a clean hydrolysis. Alternatively, strong oxidizing agents, such as oxone,[12] ozone, or permanganates, will cleave the nitronate tautomer at the double bond to form a carbonyl and nitrate. Oxophilic reductants, such as titanium salts, will reduce the nitronate to a hydrolysis-susceptible imine, but less selective reductants give the amine instead.[9]
References[edit]
- ^ Noland, Wayland E. (1955). "The NEF Reaction". Chemical Reviews. 55 (1): 137–155. doi:10.1021/cr50001a003.
- ^ Pinnick, Harold W. (1990). "The Nef Reaction". In Paquette, Leo A. (ed.). Organic Reactions Volume 38 (1st ed.). New York: Wiley. pp. 655–792. doi:10.1002/0471264180.or038.03. ISBN 9780471515944.
- ^ Grierson, David S.; Husson, Henri-Philippe (1991). "4.7 – Polonovski- and Pummerer-type Reactions and the Nef Reaction". In Trost, Barry; Fleming, Ian (eds.). Comprehensive Organic Synthesis: Selectivity, Strategy and Efficiency in Modern Organic Synthesis, Volume 6 (1st ed.). New York: Pergamon. pp. 909–947. doi:10.1016/B978-0-08-052349-1.00175-X. ISBN 9780080359298.
- ^ Nef, John Ulric (1894). "Ueber die Constitution der Salze der Nitroparaffine". Justus Liebigs Annalen der Chemie. 280 (2–3): 263–291. doi:10.1002/jlac.18942800209.
- ^ Konovalov.,: J. Russ. Phys. Chem. Soc. 2 1893, 6(I), 509.
- ^ Warren & Wyatt 2008, p. 161.
- ^ A convenient synthesis of γ-functionalized cyclopentenones Nour Lahmar, Taïcir Ben Ayed, Moncef Bellassoued and Hassen Amri Beilstein Journal of Organic Chemistry 2005, 1:11 doi:10.1186/1860-5397-1-11
- ^ McMurry, John E.; Melton, Jack (1977). "Conversion of Nitro to Carbonyl by Ozonolysis of Nitronates: 2,5-Heptandione". Organic Syntheses. 56: 36. doi:10.15227/orgsyn.056.0036.
- ^ a b Warren, Stuart; Wyatt, Paul (2008). Organic Synthesis: the disconnection approach (2nd ed.). Wiley. pp. 161–164.
- ^ Miyashita, Masaaki; Yanami, Tetsuji; Yoshikoshi, Akira (1981). "Synthesis of 1,4-Diketones from Silyl Enol Ethers and Nitroolefins: 2-(2-Oxopropyl)cyclohexanone". Organic Syntheses. 60: 117. doi:10.15227/orgsyn.060.0117.
- ^ Heinzelman, R. V. (1955). "o-Methoxyphenylacetone". Organic Syntheses. 35: 74. doi:10.15227/orgsyn.035.0074.
- ^ Ceccherelli, Paolo; Curinia, Massimo; Marcotullioa, Maria Carla; Epifanoa, Francesco; Rosatia, Ornelio (1998). "Oxone Promoted Nef Reaction. Simple Conversion of Nitro Group into Carbonyl". Synthetic Communications. 28 (16): 3057–3064. doi:10.1080/00397919808004885.
