Pegvaliase
 | |
| Clinical data | |
|---|---|
| Pronunciation | peg val' i ase |
| Trade names | Palynziq |
| Other names | Pegvaliase-pqpz; PEG-PAL; RAvPAL-PEG |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a618057 |
| License data |
|
| Pregnancy category | |
| Routes of administration | Subcutaneous |
| ATC code | |
| Legal status | |
| Legal status | |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| Chemical and physical data | |
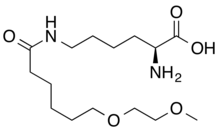
| Formula | C15H30N2O5 |
| Molar mass | 318.414 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Pegvaliase, sold under the brand name Palynziq, is a medication used for the treatment of the genetic disease phenylketonuria.[5][6][7] It is a phenylalanine (Phe)‑metabolizing enzyme.[5] Chemically, it is a pegylated derivative of the enzyme phenylalanine ammonia-lyase that metabolizes phenylalanine to reduce its blood levels.[5]
The most common adverse events include injection site reactions, joint pain, hypersensitivity reactions, headache, generalized skin reactions lasting at least 14 days, pruritus (itchy skin), nausea, dizziness, abdominal pain, throat pain, fatigue, vomiting, cough and, diarrhea.[6]
It was approved by the US Food and Drug Administration (FDA) for use in the United States in 2018.[6] The FDA considers it to be a first-in-class medication.[8]
Medical uses[edit]
Pegvaliase is indicated to reduce blood Phe concentrations in adults with phenylketonuria who have uncontrolled blood Phe concentrations greater than 600 micromol/L on existing management.[5]
Adverse effects[edit]
The FDA label for pegvaliase includes a boxed warning for anaphylaxis.
History[edit]
The safety and efficacy of pegvaliase were studied in two clinical trials in adult participants with PKU with blood phenylalanine concentrations greater than 600 µmol/L on existing management.[6] Most PKU participants in the pegvaliase trials were on an unrestricted diet prior to and during the trials.[6] The first trial was a randomized, open-label trial in participants treated with increasing doses of pegvaliase administered as a subcutaneous injection up to a target dose of either 20 mg once daily or 40 mg once daily.[6] The second trial was an 8-week, placebo-controlled, randomized withdrawal trial in participants who were previously treated with pegvaliase.[6] Participants treated with pegvaliase achieved statistically significant reductions in blood phenylalanine concentrations from their pre-treatment baseline blood Phe concentrations.[6]
The FDA granted approval of Palynziq to BioMarin Pharmaceutical.[6]
References[edit]
- ^ a b "Palynziq". Therapeutic Goods Administration (TGA). 23 July 2021. Archived from the original on 5 September 2021. Retrieved 5 September 2021.
- ^ "Updates to the Prescribing Medicines in Pregnancy database". Therapeutic Goods Administration (TGA). 12 May 2022. Archived from the original on 3 April 2022. Retrieved 13 May 2022.
- ^ "FDA-sourced list of all drugs with black box warnings (Use Download Full Results and View Query links.)". nctr-crs.fda.gov. FDA. Retrieved 22 October 2023.
- ^ "Palynziq Product information". Health Canada. 25 April 2012. Archived from the original on 29 June 2022. Retrieved 29 June 2022.
- ^ a b c d e "Palynziq- pegvaliase-pqpz injection, solution". DailyMed. 21 December 2022. Retrieved 12 December 2023.
- ^ a b c d e f g h i "FDA approves a new treatment for PKU, a rare and serious genetic disease" (Press release). U.S. Food and Drug Administration (FDA). 24 May 2018. Archived from the original on 25 July 2021. Retrieved 12 October 2020.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.
- ^ Mahan KC, Gandhi MA, Anand S (April 2019). "Pegvaliase: a novel treatment option for adults with phenylketonuria". Current Medical Research and Opinion. 35 (4): 647–651. doi:10.1080/03007995.2018.1528215. PMID 30247930. S2CID 52813510.
- ^ New Drug Therapy Approvals 2018 (PDF). U.S. Food and Drug Administration (FDA) (Report). January 2019. Archived from the original on 17 September 2020. Retrieved 16 September 2020.
