Plasminogen activator inhibitor-2
| SERPINB2 | |||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
 | |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| Identifiers | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Aliases | SERPINB2, HsT1201, PAI, PAI-2, PAI2, PLANH2, serpin family B member 2 | ||||||||||||||||||||||||||||||||||||||||||||||||||
| External IDs | OMIM: 173390 MGI: 97609 HomoloGene: 20571 GeneCards: SERPINB2 | ||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| Wikidata | |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
Plasminogen activator inhibitor-2 (placental PAI, SerpinB2, PAI-2), a serine protease inhibitor of the serpin superfamily, is a coagulation factor that inactivates tissue plasminogen activator and urokinase. It is present in most cells, especially monocytes/macrophages. PAI-2 exists in two forms, a 60-kDa extracellular glycosylated form and a 43-kDa intracellular form.
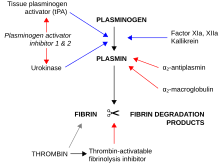
It is present only at detectable quantities in blood during pregnancy, as it is produced by the placenta, and may explain partially the increased rate of thrombosis during pregnancy. The majority of expressed PAI-2 remains unsecreted due to the presence of an inefficient internal signal peptide.
Interactions[edit]
PAI-2 has been reported to bind a series of intracellular and extracellular proteins. Whether PAI-2's physiological function is inhibition of the extracellular protease urokinase and/or whether PAI-2 has intracellular activities remains controversial. At least one of PAI-2's physiological functions may involve regulation of adaptive immunity.[5]
Structure and polymerization[edit]
Like other serpins, PAI-2 has three beta sheets (A, B, C) and nine alpha helices (hA-hI).[6][7] The structure of PAI-2 mutants have been solved, in which the 33-amino acid loop connecting helices C and D is deleted. This CD-loop is particularly flexible and difficult to stabilize, as the loop is known to translocate up to 54 Å during the formation of intramolecular disulfide bonds.[8] In addition to the CD-loop, notable motifs include the reactive center loop (RCL) spanning amino acids 379-383 and an N-terminal hydrophobic signal sequence.

Despite their similar inhibitory targets, PAI-2 is phylogenetically distant from its counterpart plasminogen activator inhibitor-1 (PAI-1). As a member of the ovalbumin-related serpin family, PAI-2 is genetically similar to chicken ovalbumin (Gallus gallus), and is a close mammalian homolog.[9] Both ovalbumin and PAI-2 undergo secretion via uncleaved secretory signal peptides, although PAI-2 secretion is relatively much less efficient.[10]
PAI-2 exists in three polymeric states: monomeric, polymerigenic, and polymer (inactive state). Polymerization occurs by a so-called "loop-sheet" mechanism, in which the RCL of one molecule sequentially inserts into the A-beta-sheet of the next molecule. This process occurs preferentially when PAI-2 is in its polymerigenic form, which is stabilized by a disulfide bond between Cys-79 (located in the CD-loop) and Cys-161.[11] When PAI-2 is in its monomeric form, the CD-loop is vastly out-of-position for this disulfide linkage, and it must translocate a distance of 54 Å to become sufficiently close to Cys-161. Nevertheless, since the CD-loop is quite flexible, the monomeric and polymerigenic forms are fully interconvertible, and one state can be favored over the other by altering the redox environment of the protein.[8] Polymerization of PAI-2 occurs spontaneously under physiological conditions, for instance in the cytosol of placental cells.[12] Cytosolic PAI-2 tends to be monomeric, while PAI-2 in secretory organelles (which tend to be more oxidizing than the cytosol) is more prone to polymerization.[11] For these combined reasons, it is thought that PAI-2 may sense and respond to environmental redox potential.[8]
Mechanism[edit]
PAI-2 uses a suicide inhibition mechanism (a common mechanism for serpins) to irreversibly inactivate tissue plasminogen activator and urokinase.[6] First, the target serine protease docks to PAI-2 and catalyzes cleavage of the RCL, between residues Arg-380 and Thr-381. At this point, two outcomes are possible: the protease escapes, leaving an inactive PAI-2; or the protease forms a permanent, covalently-bonded complex with PAI-2, in which the protease is significantly distorted.
Biological Functions[edit]
Although extracellular (glycosylated) PAI-2 functions to regulate fibrinolysis, it remains unclear whether this inhibitory role is the main function of PAI-2. PAI-2 is predominantly intracellular. The secretory signal peptide of PAI-2 is relatively inefficient, perhaps by evolutionary design, as various mutations to the signal sequence can significantly enhance secretion efficiency.[10] PAI-2 is undetectable in adult plasma, and is typically only detectable during pregnancy, in myelomonocytic leukemias, or in gingival crevicular fluid; moreover, PAI-2 is a slower inhibitor than its counterpart PAI-1 by orders of magnitude (based on second order rate constants).[13] On the other hand, detailed intracellular roles for PAI-2 have not yet been conclusively established.
PAI-2 is upregulated during both pregnancy and immune responses. During pregnancy, PAI-2 is particularly present in the decidua and amniotic fluid, where it may protect membranes from digestion and aid in remodeling fetal and uterine tissues.[14] PAI-2 assists PAI-1 in regulating fibrinolysis and may help prevent overexpression of PAI-1, which increases risk of thrombosis.[14][15] Over the course of a pregnancy, PAI-2 plasma concentration rises from nearly-undetectable levels to 250 ng/mL (mostly in glycosylated form).[13]
Among immune cells, macrophages are the main producers of PAI-2, as both B-cells and T-cells do not produce significant amounts.[16] PAI-2 plays a role in inflammatory responses and infections, potentially in downregulating T cells that secrete IgG2c and interferon type II.[16]
Due to its position on chromosome 18 close to the bcl-2 protooncogene and several other serpins, PAI-2's role in apoptosis has been investigated, but current evidence remains inconclusive.[13][17] A recent study suggests PAI-2 may be a direct downstream target and activator of p53, and may directly stabilize p21; in addition, PAI-2 expression is increased in senescent fibroblasts and may arrest growth of young fibroplasts.[18]
Potential roles in cancer[edit]
The role of PAI-2 in cancer growth and metastasis is complex, as PAI-2 may have tumor-promoting and tumor-inhibiting effects. Notably, it is high expression of PAI-2 by tumor cells, not the host organism, which influences cancer growth.[19] Cancer cells may facilitate export of PAI-2 via microparticles.[19]
PAI-2 provides protection for cancer cells against plasmin-induced cell death, which can exert a lethal effect on tumors. This protection is particularly salient in brain metastases, which tend to express high levels of PAI-2 and neuroserpin, and whose growth may be partially inhibited by knockout of PAI-2.[20] Due to its high expression in tumor cells, PAI-2 has been used to track and study the spread of angiotropic melanoma cells.[21]
Although PAI-2 expression can promote metastasis to the brain, in other cases high PAI-2 expression significantly decreases metastasis to the lungs and other organs.[19][22] The particular effects of PAI-2 on metastasis may depend on cancer type and location in the body.
See also[edit]
References[edit]
- ^ a b c GRCh38: Ensembl release 89: ENSG00000197632 – Ensembl, May 2017
- ^ a b c GRCm38: Ensembl release 89: ENSMUSG00000062345 – Ensembl, May 2017
- ^ "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ "Mouse PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ Schroder WA, Major L, Suhrbier A (2011). "The role of SerpinB2 in immunity". Critical Reviews in Immunology. 31 (1): 15–30. doi:10.1615/critrevimmunol.v31.i1.20. PMID 21395508.
- ^ a b Law RH, Zhang Q, McGowan S, Buckle AM, Silverman GA, Wong W, Rosado CJ, Langendorf CG, Pike RN, Bird PI, Whisstock JC (2006). "An overview of the serpin superfamily". Genome Biology. 7 (5): 216. doi:10.1186/gb-2006-7-5-216. PMC 1779521. PMID 16737556.
- ^ Di Giusto DA, Sutherland AP, Jankova L, Harrop SJ, Curmi PM, King GC (November 2005). "Plasminogen activator inhibitor-2 is highly tolerant to P8 residue substitution--implications for serpin mechanistic model and prediction of nsSNP activities". Journal of Molecular Biology. 353 (5): 1069–80. doi:10.1016/j.jmb.2005.09.008. PMID 16214170.
- ^ a b c Lobov S, Wilczynska M, Bergström F, Johansson LB, Ny T (December 2004). "Structural bases of the redox-dependent conformational switch in the serpin PAI-2". Journal of Molecular Biology. 344 (5): 1359–68. doi:10.1016/j.jmb.2004.10.010. PMID 15561148.
- ^ Ye RD, Ahern SM, Le Beau MM, Lebo RV, Sadler JE (April 1989). "Structure of the gene for human plasminogen activator inhibitor-2. The nearest mammalian homologue of chicken ovalbumin". The Journal of Biological Chemistry. 264 (10): 5495–502. doi:10.1016/S0021-9258(18)83572-4. PMID 2494165.
- ^ a b Belin D, Guzman LM, Bost S, Konakova M, Silva F, Beckwith J (January 2004). "Functional activity of eukaryotic signal sequences in Escherichia coli: the ovalbumin family of serine protease inhibitors". Journal of Molecular Biology. 335 (2): 437–53. doi:10.1016/j.jmb.2003.10.076. PMID 14672654.
- ^ a b Wilczynska M, Lobov S, Ohlsson PI, Ny T (April 2003). "A redox-sensitive loop regulates plasminogen activator inhibitor type 2 (PAI-2) polymerization". The EMBO Journal. 22 (8): 1753–61. doi:10.1093/emboj/cdg178. PMC 154470. PMID 12682008.
- ^ Mikus P, Ny T (April 1996). "Intracellular polymerization of the serpin plasminogen activator inhibitor type 2". The Journal of Biological Chemistry. 271 (17): 10048–53. doi:10.1074/jbc.271.17.10048. PMID 8626560.
- ^ a b c Kruithof EK, Baker MS, Bunn CL (December 1995). "Biological and clinical aspects of plasminogen activator inhibitor type 2". Blood. 86 (11): 4007–24. doi:10.1182/blood.v86.11.4007.bloodjournal86114007. PMID 7492756.
- ^ a b Astedt B, Lindoff C, Lecander I (1998). "Significance of the plasminogen activator inhibitor of placental type (PAI-2) in pregnancy". Seminars in Thrombosis and Hemostasis. 24 (5): 431–5. doi:10.1055/s-2007-996035. PMID 9834009. S2CID 39347062.
- ^ Thompson PN, Cho E, Blumenstock FA, Shah DM, Saba TM (October 1992). "Rebound elevation of fibronectin after tissue injury and ischemia: role of fibronectin synthesis". The American Journal of Physiology. 263 (4 Pt 1): G437–45. doi:10.1152/ajpgi.1992.263.4.G437. PMID 1415704.
- ^ a b Schroder WA, Le TT, Major L, Street S, Gardner J, Lambley E, Markey K, MacDonald KP, Fish RJ, Thomas R, Suhrbier A (March 2010). "A physiological function of inflammation-associated SerpinB2 is regulation of adaptive immunity". Journal of Immunology. 184 (5): 2663–70. doi:10.4049/jimmunol.0902187. PMID 20130210.
- ^ Lee JA, Cochran BJ, Lobov S, Ranson M (June 2011). "Forty years later and the role of plasminogen activator inhibitor type 2/SERPINB2 is still an enigma". Seminars in Thrombosis and Hemostasis. 37 (4): 395–407. doi:10.1055/s-0031-1276589. PMID 21805446. S2CID 260316614.
- ^ Hsieh HH, Chen YC, Jhan JR, Lin JJ (October 2017). "The serine protease inhibitor serpinB2 binds and stabilizes p21 in senescent cells". Journal of Cell Science. 130 (19): 3272–3281. doi:10.1242/jcs.204974. PMID 28794016.
- ^ a b c Schroder WA, Major LD, Le TT, Gardner J, Sweet MJ, Janciauskiene S, Suhrbier A (June 2014). "Tumor cell-expressed SerpinB2 is present on microparticles and inhibits metastasis". Cancer Medicine. 3 (3): 500–13. doi:10.1002/cam4.229. PMC 4101741. PMID 24644264.
- ^ Valiente M, Obenauf AC, Jin X, Chen Q, Zhang XH, Lee DJ, Chaft JE, Kris MG, Huse JT, Brogi E, Massagué J (February 2014). "Serpins promote cancer cell survival and vascular co-option in brain metastasis". Cell. 156 (5): 1002–16. doi:10.1016/j.cell.2014.01.040. PMC 3988473. PMID 24581498.
- ^ Bentolila LA, Prakash R, Mihic-Probst D, Wadehra M, Kleinman HK, Carmichael TS, Péault B, Barnhill RL, Lugassy C (April 2016). "Imaging of Angiotropism/Vascular Co-Option in a Murine Model of Brain Melanoma: Implications for Melanoma Progression along Extravascular Pathways". Scientific Reports. 6: 23834. Bibcode:2016NatSR...623834B. doi:10.1038/srep23834. PMC 4822155. PMID 27048955.
- ^ Mueller BM, Yu YB, Laug WE (January 1995). "Overexpression of plasminogen activator inhibitor 2 in human melanoma cells inhibits spontaneous metastasis in scid/scid mice". Proceedings of the National Academy of Sciences of the United States of America. 92 (1): 205–9. Bibcode:1995PNAS...92..205M. doi:10.1073/pnas.92.1.205. PMC 42846. PMID 7816818.
Further reading[edit]
- Rasmussen HH, van Damme J, Puype M, Gesser B, Celis JE, Vandekerckhove J (December 1992). "Microsequences of 145 proteins recorded in the two-dimensional gel protein database of normal human epidermal keratinocytes". Electrophoresis. 13 (12): 960–9. doi:10.1002/elps.11501301199. PMID 1286667. S2CID 41855774.
- Ellis V, Wun TC, Behrendt N, Rønne E, Danø K (June 1990). "Inhibition of receptor-bound urokinase by plasminogen-activator inhibitors". The Journal of Biological Chemistry. 265 (17): 9904–8. doi:10.1016/S0021-9258(19)38757-5. PMID 2161846.
- Estreicher A, Mühlhauser J, Carpentier JL, Orci L, Vassalli JD (August 1990). "The receptor for urokinase type plasminogen activator polarizes expression of the protease to the leading edge of migrating monocytes and promotes degradation of enzyme inhibitor complexes". The Journal of Cell Biology. 111 (2): 783–92. doi:10.1083/jcb.111.2.783. PMC 2116194. PMID 2166055.
- Samia JA, Alexander SJ, Horton KW, Auron PE, Byers MG, Shows TB, Webb AC (January 1990). "Chromosomal organization and localization of the human urokinase inhibitor gene: perfect structural conservation with ovalbumin". Genomics. 6 (1): 159–67. doi:10.1016/0888-7543(90)90461-3. PMID 2303256.
- Schwartz BS, Monroe MC, Bradshaw JD (June 1989). "Endotoxin-induced production of plasminogen activator inhibitor by human monocytes is autonomous and can be inhibited by lipid X". Blood. 73 (8): 2188–95. doi:10.1182/blood.V73.8.2188.2188. PMID 2471561.
- Ye RD, Ahern SM, Le Beau MM, Lebo RV, Sadler JE (April 1989). "Structure of the gene for human plasminogen activator inhibitor-2. The nearest mammalian homologue of chicken ovalbumin". The Journal of Biological Chemistry. 264 (10): 5495–502. doi:10.1016/S0021-9258(18)83572-4. PMID 2494165.
- Laug WE, Aebersold R, Jong A, Rideout W, Bergman BL, Baker J (June 1989). "Isolation of multiple types of plasminogen activator inhibitors from vascular smooth muscle cells". Thrombosis and Haemostasis. 61 (3): 517–21. doi:10.1055/s-0038-1646626. PMID 2799763. S2CID 2349338.
- Kruithof EK, Cousin E (October 1988). "Plasminogen activator inhibitor 2. Isolation and characterization of the promoter region of the gene". Biochemical and Biophysical Research Communications. 156 (1): 383–8. doi:10.1016/S0006-291X(88)80852-0. PMID 2845977.
- Ye RD, Wun TC, Sadler JE (March 1987). "cDNA cloning and expression in Escherichia coli of a plasminogen activator inhibitor from human placenta". The Journal of Biological Chemistry. 262 (8): 3718–25. doi:10.1016/S0021-9258(18)61414-0. PMID 3029122.
- Antalis TM, Clark MA, Barnes T, Lehrbach PR, Devine PL, Schevzov G, Goss NH, Stephens RW, Tolstoshev P (February 1988). "Cloning and expression of a cDNA coding for a human monocyte-derived plasminogen activator inhibitor". Proceedings of the National Academy of Sciences of the United States of America. 85 (4): 985–9. Bibcode:1988PNAS...85..985A. doi:10.1073/pnas.85.4.985. PMC 279685. PMID 3257578.
- Schleuning WD, Medcalf RL, Hession C, Rothenbühler R, Shaw A, Kruithof EK (December 1987). "Plasminogen activator inhibitor 2: regulation of gene transcription during phorbol ester-mediated differentiation of U-937 human histiocytic lymphoma cells". Molecular and Cellular Biology. 7 (12): 4564–7. doi:10.1128/mcb.7.12.4564. PMC 368144. PMID 3325828.
- Webb AC, Collins KL, Snyder SE, Alexander SJ, Rosenwasser LJ, Eddy RL, Shows TB, Auron PE (July 1987). "Human monocyte Arg-Serpin cDNA. Sequence, chromosomal assignment, and homology to plasminogen activator-inhibitor". The Journal of Experimental Medicine. 166 (1): 77–94. doi:10.1084/jem.166.1.77. PMC 2188630. PMID 3496414.
- Dickinson JL, Bates EJ, Ferrante A, Antalis TM (November 1995). "Plasminogen activator inhibitor type 2 inhibits tumor necrosis factor alpha-induced apoptosis. Evidence for an alternate biological function". The Journal of Biological Chemistry. 270 (46): 27894–904. doi:10.1074/jbc.270.46.27894. PMID 7499264.
- Mikus P, Urano T, Liljeström P, Ny T (December 1993). "Plasminogen-activator inhibitor type 2 (PAI-2) is a spontaneously polymerising SERPIN. Biochemical characterisation of the recombinant intracellular and extracellular forms". European Journal of Biochemistry. 218 (3): 1071–82. doi:10.1111/j.1432-1033.1993.tb18467.x. PMID 7506655.
- Jensen PJ, Wu Q, Janowitz P, Ando Y, Schechter NM (March 1995). "Plasminogen activator inhibitor type 2: an intracellular keratinocyte differentiation product that is incorporated into the cornified envelope". Experimental Cell Research. 217 (1): 65–71. doi:10.1006/excr.1995.1064. PMID 7867722.
- Akiyama H, Ikeda K, Kondo H, Kato M, McGeer PL (December 1993). "Microglia express the type 2 plasminogen activator inhibitor in the brain of control subjects and patients with Alzheimer's disease". Neuroscience Letters. 164 (1–2): 233–5. doi:10.1016/0304-3940(93)90899-V. PMID 8152607. S2CID 40620114.
- Ragno P, Montuori N, Vassalli JD, Rossi G (June 1993). "Processing of complex between urokinase and its type-2 inhibitor on the cell surface. A possible regulatory mechanism of urokinase activity". FEBS Letters. 323 (3): 279–84. doi:10.1016/0014-5793(93)81357-6. PMID 8388810. S2CID 1822666.
- Bartuski AJ, Kamachi Y, Schick C, Overhauser J, Silverman GA (August 1997). "Cytoplasmic antiproteinase 2 (PI8) and bomapin (PI10) map to the serpin cluster at 18q21.3". Genomics. 43 (3): 321–8. doi:10.1006/geno.1997.4827. PMID 9268635.
- Mahony D, Stringer BW, Dickinson JL, Antalis TM (September 1998). "DNase I hypersensitive sites in the 5' flanking region of the human plasminogen activator inhibitor type 2 (PAI-2) gene are associated with basal and tumor necrosis factor-alpha-induced transcription in monocytes". European Journal of Biochemistry. 256 (3): 550–9. doi:10.1046/j.1432-1327.1998.2560550.x. PMID 9780231.
- Nishida Y, Hayashi Y, Imai Y, Itoh H (February 1998). "Expression and localization of the urokinase-type plasminogen activator receptor (uPAR) in the human placenta". The Kobe Journal of Medical Sciences. 44 (1): 31–43. PMID 9846056.
External links[edit]
- The MEROPS online database for peptidases and their inhibitors: I04.007
- Plasminogen+Activator+Inhibitor+2 at the U.S. National Library of Medicine Medical Subject Headings (MeSH)





![1jrr: HUMAN PLASMINOGEN ACTIVATOR INHIBITOR-2.[LOOP (66-98) DELETIONMUTANT] COMPLEXED WITH PEPTIDE MIMIckING THE REACTIVE CENTER LOOP](http://upload.wikimedia.org/wikipedia/commons/thumb/3/31/PDB_1jrr_EBI.jpg/180px-PDB_1jrr_EBI.jpg)
![2arq: Human plasminogen activator inhibitor-2.[loop (66-98) deletion mutant] complexed with peptide n-acetyl-teaaagdggvmtgr-oh](http://upload.wikimedia.org/wikipedia/commons/thumb/0/07/PDB_2arq_EBI.png/180px-PDB_2arq_EBI.png)
![2arr: Human plasminogen activator inhibitor-2.[loop (66-98) deletion mutant] complexed with peptide n-acetyl-teaaagmggvmtgr-oh](http://upload.wikimedia.org/wikipedia/commons/thumb/1/18/PDB_2arr_EBI.png/180px-PDB_2arr_EBI.png)