Refrigerant

A refrigerant is a working fluid used in the refrigeration cycle of air conditioning systems and heat pumps where in most cases they undergo a repeated phase transition from a liquid to a gas and back again. Refrigerants are heavily regulated due to their toxicity, flammability and the contribution of CFC and HCFC refrigerants to ozone depletion and that of HFC refrigerants to climate change.
Refrigerants are used in a direct expansion (DX) system to transfer energy from one environment to another, typically from inside a building to outside (or vice versa) commonly known as an "air conditioner" or "heat pump". Refrigerants can carry 10 times more energy per kg than water, and 50 times more than air.
In some countries, refrigerants are controlled substances due to high pressures (700–1,000 kPa (100–150 psi)), extreme temperatures (−50 °C [−58 °F] to over 100 °C [212 °F]), flammability (A1 class non-flammable, A2/A2L class flammable and A3 class extremely flammable/explosive) and toxicity (B1-low, B2-medium & B3-high), as classified by ISO 817 & ASHRAE 34.
Refrigerants must only be handled by qualified/certified engineers to the relevant classes of refrigerant (in the UK, C&G 2079 for A1-class, and C&G 6187-2 for A2/A2L & A3 class refrigerants).
History[edit]
The first air conditioners and refrigerators employed toxic or flammable gases, such as ammonia, sulfur dioxide, methyl chloride, or propane, that could result in fatal accidents when they leaked.[1]
In 1928 Thomas Midgley Jr. created the first non-flammable, non-toxic chlorofluorocarbon gas, Freon (R-12). The name is a trademark name owned by DuPont (now Chemours) for any chlorofluorocarbon (CFC), hydrochlorofluorocarbon (HCFC), or hydrofluorocarbon (HFC) refrigerant. Following the discovery of better synthesis methods, CFCs such as R-11,[2] R-12,[3] R-123[2] and R-502[4] dominated the market.
Phasing out of CFCs[edit]
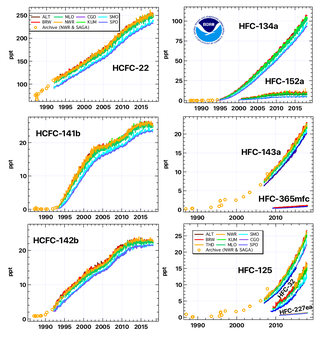
In the early 1980s, scientists discovered that CFCs were causing major damage to the ozone layer that protects the earth from ultraviolet radiation, and to the ozone holes over polar regions. This led to the signing of the Montreal Protocol in 1987 which aimed to phase out CFCs and HCFC[5] but did not address the contributions that HFCs made to climate change. The adoption of HCFCs such as R-22,[6][7][8] and R-123[2] was accelerated and so were used in most U.S. homes in air conditioners and in chillers[9] from the 1980s as they have a dramatically lower Ozone Depletion Potential (ODP) than CFCs, but their ODP was still not zero which led to their eventual phase-out.
Hydrofluorocarbons (HFCs) such as R-134a,[10][11] R-143a, R-407A,[12] R-407C,[13] R-404A,[4] R-410A[14] (a 50/50 blend of R-125/R-32) and R-507[15][16] were promoted as replacements for CFCs and HCFCs in the 1990s and 2000s. HFCs were not ozone-depleting but did have global warming potentials (GWPs) thousands of times greater than CO2 with atmospheric lifetimes that can extend for decades. This in turn, starting from the 2010s, led to the adoption in new equipment of Hydrocarbon and HFO (hydrofluoroolefin) refrigerants R-32,[17] R-290,[18] R-600a,[18] R-454B,[19] R-1234yf,[20][21] R-514A,[22] R-744 (CO2),[23] R-1234ze(E)[24] and R-1233zd(E),[25] which have both an ODP of zero and a lower GWP. Hydrocarbons and CO2 are sometimes called natural refrigerants because they can be found in nature.
The environmental organization Greenpeace provided funding to a former East German refrigerator company to research alternative ozone- and climate-safe refrigerants in 1992. The company developed hydrocarbon mixes such as isopentane and isobutane,[citation needed] propane and isobutane, or pure isobutane,[26] called "Greenfreeze", but as a condition of the contract with Greenpeace could not patent the technology, which led to their widespread adoption by other firms.[27][28][29] Policy and political influence by corporate executives resisted change however,[30][31] citing the flammability and explosive properties of the refrigerants,[32] and DuPont together with other companies blocked them in the U.S. with the U.S. EPA.[33][34]
Beginning on 14 November 1994, the U.S. Environmental Protection Agency restricted the sale, possession and use of refrigerants to only licensed technicians, per rules under sections 608 and 609 of the Clean Air Act.[35] In 1995, Germany made CFC refrigerators illegal.[36]
In 1996 Eurammon, a European non-profit initiative for natural refrigerants, was established and comprises European companies, institutions, and industry experts.[37][38][39]
In 1997, FCs and HFCs were included in the Kyoto Protocol to the Framework Convention on Climate Change.
In 2000 in the UK, the Ozone Regulations[40] came into force which banned the use of ozone-depleting HCFC refrigerants such as R22 in new systems. The Regulation banned the use of R22 as a "top-up" fluid for maintenance from 2010 for virgin fluid and from 2015 for recycled fluid.[citation needed]
Addressing greenhouse gases[edit]
With growing interest in natural refrigerants as alternatives to synthetic refrigerants such as CFCs, HCFCs and HFCs, in 2004, Greenpeace worked with multinational corporations like Coca-Cola and Unilever, and later Pepsico and others, to create a corporate coalition called Refrigerants Naturally!.[36][41] Four years later, Ben & Jerry's of Unilever and General Electric began to take steps to support production and use in the U.S.[42] It is estimated that almost 75 percent of the refrigeration and air conditioning sector has the potential to be converted to natural refrigerants.[43]
In 2006, the EU adopted a Regulation on fluorinated greenhouse gases (FCs and HFCs) to encourage to transition to natural refrigerants (such as hydrocarbons). It was reported in 2010 that some refrigerants are being used as recreational drugs, leading to an extremely dangerous phenomenon known as inhalant abuse.[44]
From 2011 the European Union started to phase out refrigerants with a global warming potential (GWP) of more than 150 in automotive air conditioning (GWP = 100-year warming potential of one kilogram of a gas relative to one kilogram of CO2) such as the refrigerant HFC-134a (known as R-134a in North America) which has a GWP of 1526.[45] In the same year the EPA decided in favour of the ozone- and climate-safe refrigerant for U.S. manufacture.[27][46][47]
A 2018 study by the nonprofit organization "Drawdown" put proper refrigerant management and disposal at the very top of the list of climate impact solutions, with an impact equivalent to eliminating over 17 years of US carbon dioxide emissions.[48]
In 2019 it was estimated that CFCs, HCFCs, and HFCs were responsible for about 10% of direct radiative forcing from all long-lived anthropogenic greenhouse gases.[49] and in the same year the UNEP published new voluntary guidelines,[50] however many countries have not yet ratified the Kigali Amendment.
From early 2020 HFCs (including R-404A, R-134a and R-410A) are being superseded: Residential air-conditioning systems and heat pumps are increasingly using R-32. This still has a GWP of more than 600. Progressive devices use refrigerants with almost no climate impact, namely R-290 (propane), R-600a (isobutane) or R-1234yf (less flammable, in cars). In commercial refrigeration also CO2 (R-744) can be used.
Requirements and desirable properties[edit]
A refrigerant needs to have: a boiling point that is somewhat below the target temperature (although boiling point can be adjusted by adjusting the pressure appropriately), a high heat of vaporization, a moderate density in liquid form, a relatively high density in gaseous form (which can also be adjusted by setting pressure appropriately), and a high critical temperature. Working pressures should ideally be containable by copper tubing, a commonly available material. Extremely high pressures should be avoided.[citation needed]
The ideal refrigerant would be: non-corrosive, non-toxic, non-flammable, with no ozone depletion and global warming potential. It should preferably be natural with well-studied and low environmental impact. Newer refrigerants address the issue of the damage that CFCs caused to the ozone layer and the contribution that HCFCs make to climate change, but some do raise issues relating to toxicity and/or flammability.[51]
Common refrigerants[edit]
Refrigerants with very low climate impact[edit]
With increasing regulations, refrigerants with a very low global warming potential are expected to play a dominant role in the 21st century,[52] in particular, R-290 and R-1234yf. Starting from almost no market share in 2018,[53] low GWPO devices are gaining market share in 2022.
| Code | Chemical | Name | GWP 20yr[54] | GWP 100yr[54] | Status | Commentary |
|---|---|---|---|---|---|---|
| R-290 | C3H8 | Propane | 3.3[55] | Increasing use | Low cost, widely available and efficient. They also have zero ozone depletion potential. Despite their flammability, they are increasingly used in domestic refrigerators and heat pumps. In 2010, about one-third of all household refrigerators and freezers manufactured globally used isobutane or an isobutane/propane blend, and this was expected to increase to 75% by 2020.[56] | |
| R-600a | HC(CH3)3 | Isobutane | 3.3 | Widely used | See R-290. | |
| R-717 | NH3 | Ammonia | 0 | 0[57] | Widely used | Commonly used before the popularisation of CFCs, it is again being considered but does suffer from the disadvantage of toxicity, and it requires corrosion-resistant components, which restricts its domestic and small-scale use. Anhydrous ammonia is widely used in industrial refrigeration applications and hockey rinks because of its high energy efficiency and low cost. |
| R-1234yf HFO-1234yf | C3H2F4 | 2,3,3,3-Tetrafluoropropene | <1 | Less performance but also less flammable than R-290.[52] GM announced that it would start using "hydro-fluoro olefin", HFO-1234yf, in all of its brands by 2013.[58] | ||
| R-744 | CO2 | Carbon dioxide | 1 | 1 | In use | Was used as a refrigerant prior to the discovery of CFCs (this was also the case for propane)[1] and now having a renaissance due to it being non-ozone depleting, non-toxic and non-flammable. It may become the working fluid of choice to replace current HFCs in cars, supermarkets, and heat pumps such as EcoCute. Coca-Cola has fielded CO2-based beverage coolers and the U.S. Army is considering CO2 refrigeration.[59][60] Due to the need to operate at pressures of up to 130 bars (1,900 psi; 13,000 kPa), CO2 systems require highly resistant components, however these have already been developed for mass production in many sectors. |
Most used[edit]
| Code | Chemical | Name | Global warming potential 20yr[54] | GWP 100yr[54] | Status | Commentary |
|---|---|---|---|---|---|---|
| R-32 HFC-32 | CH2F2 | Difluoromethane | 2430 | 677 | Widely used | Promoted as climate-friendly substitute for R-134a and R-410A, but still with high climate impact. Has excellent heat transfer and pressure drop performance, both in condensation and vaporisation.[61] It has an atmospheric lifetime of nearly 5 years.[62] Currently used in residential and commercial air-conditioners and heat pumps. |
| R-134a HFC-134a | CH2FCF3 | 1,1,1,2-Tetrafluoroethane | 3790 | 1550 | Widely used | Most used in 2020 for hydronic heat pumps in Europe and the United States in spite of high GWP.[53] Commonly used in automotive air conditioners prior to phase out which began in 2012. |
| R-410A | 50% R-32 / 50% R-125 (pentafluoroethane) | Between 2430 (R-32) and 6350 (R-125) | > 677 | Widely Used | Most used in split heat pumps / AC by 2018. Almost 100% share in the USA.[53] |
Banned / Phased out[edit]
| Code | Chemical | Name | Global warming potential 20yr[54] | GWP 100yr[54] | Status | Commentary |
|---|---|---|---|---|---|---|
| R-11 CFC-11 | CCl3F | Trichlorofluoromethane | 6900 | 4660 | Banned | Production was banned in developed countries by Montreal Protocol in 1996 |
| R-12 CFC-12 | CCl2F2 | Dichlorodifluoromethane | 10800 | 10200 | Banned | Also known as Freon, a widely used chlorofluorocarbon halomethane (CFC). Production was banned in developed countries by Montreal Protocol in 1996, and in developing countries (article 5 countries) in 2010.[63] |
| R-22 HCFC-22 | CHClF2 | Chlorodifluoromethane | 5280 | 1760 | Being phased out | A widely used hydrochlorofluorocarbon (HCFC) and powerful greenhouse gas with a GWP equal to 1810. Worldwide production of R-22 in 2008 was about 800 Gg per year, up from about 450 Gg per year in 1998. R-438A (MO-99) is a R-22 replacement.[64] |
| R-123 HCFC-123 | CHCl2CF3 | 2,2-Dichloro-1,1,1-trifluoroethane | 292 | 79 | US phase-out | Used in large tonnage centrifugal chiller applications. All U.S. production and import of virgin HCFCs will be phased out by 2030, with limited exceptions.[65] R-123 refrigerant was used to retrofit some chiller that used R-11 refrigerant Trichlorofluoromethane. The production of R-11 was banned in developed countries by Montreal Protocol in 1996.[66] |
Other[edit]
| Code | Chemical | Name | Global warming potential 20yr[54] | GWP 100yr[54] | Commentary |
|---|---|---|---|---|---|
| R-152a HFC-152a | CH3CHF2 | 1,1-Difluoroethane | 506 | 138 | As a compressed air duster |
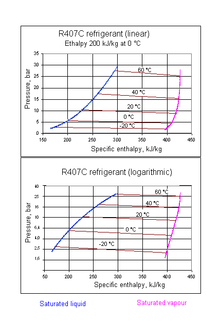
| R-407C | Mixture of difluoromethane and pentafluoroethane and 1,1,1,2-tetrafluoroethane | A mixture of R-32, R-125, and R-134a | |||
| R-454B | Difluoromethane and 2,3,3,3-Tetrafluoropropene | HFOs blend of refrigerants Difluoromethane (R-32) and 2,3,3,3-Tetrafluoropropene (R-1234yf).[67][68][69][70] | |||
| R-513A | An HFO/HFC blend (56% R-1234yf/44%R-134a) | May replace R-134a as an interim alternative[71] | |||
| R-514A | HFO-1336mzz-Z/trans-1,2- dichloroethylene (t-DCE) | An hydrofluoroolefin (HFO)-based refrigerant to replace R-123 in low pressure centrifugal chillers for commercial and industrial applications.[72][73] |
Refrigerant reclamation and disposal[edit]
Coolant and refrigerants are found throughout the industrialized world, in homes, offices, and factories, in devices such as refrigerators, air conditioners, central air conditioning systems (HVAC), freezers, and dehumidifiers. When these units are serviced, there is a risk that refrigerant gas will be vented into the atmosphere either accidentally or intentionally, hence the creation of technician training and certification programs in order to ensure that the material is conserved and managed safely. Mistreatment of these gases has been shown to deplete the ozone layer and is suspected to contribute to global warming.[74]
With the exception of isobutane and propane (R600a, R441A and R290), ammonia and CO2 under Section 608 of the United States' Clean Air Act it is illegal to knowingly release any refrigerants into the atmosphere.[75][76]
Refrigerant reclamation is the act of processing used refrigerant gas which has previously been used in some type of refrigeration loop such that it meets specifications for new refrigerant gas. In the United States, the Clean Air Act of 1990 requires that used refrigerant be processed by a certified reclaimer, which must be licensed by the United States Environmental Protection Agency (EPA), and the material must be recovered and delivered to the reclaimer by EPA-certified technicians.[77]
Classification of refrigerants[edit]
Refrigerants may be divided into three classes according to their manner of absorption or extraction of heat from the substances to be refrigerated:[citation needed]
- Class 1: This class includes refrigerants that cool by phase change (typically boiling), using the refrigerant's latent heat.
- Class 2: These refrigerants cool by temperature change or 'sensible heat', the quantity of heat being the specific heat capacity x the temperature change. They are air, calcium chloride brine, sodium chloride brine, alcohol, and similar nonfreezing solutions. The purpose of Class 2 refrigerants is to receive a reduction of temperature from Class 1 refrigerants and convey this lower temperature to the area to be cooled.
- Class 3: This group consists of solutions that contain absorbed vapors of liquefiable agents or refrigerating media. These solutions function by nature of their ability to carry liquefiable vapors, which produce a cooling effect by the absorption of their heat of solution. They can also be classified into many categories.
R numbering system[edit]
The R- numbering system was developed by DuPont (which owned the Freon trademark), and systematically identifies the molecular structure of refrigerants made with a single halogenated hydrocarbon. ASHRAE has since set guidelines for the numbering system as follows:[78]
R-X1X2X3X4
- X1 = Number of unsaturated carbon-carbon bonds (omit if zero)
- X2 = Number of carbon atoms minus 1 (omit if zero)
- X3 = Number of hydrogen atoms plus 1
- X4 = Number of fluorine atoms
Series[edit]
- R-xx Methane Series
- R-1xx Ethane Series
- R-2xx Propane Series
- R-4xx Zeotropic blend
- R-5xx Azeotropic blend
- R-6xx Saturated hydrocarbons (except for propane which is R-290)
- R-7xx Inorganic Compounds with a molar mass < 100
- R-7xxx Inorganic Compounds with a molar mass ≥ 100
Ethane Derived Chains[edit]
- Number Only Most symmetrical isomer
- Lower Case Suffix (a,b,c,etc.) indicates increasingly unsymmetrical isomers
Propane Derived Chains[edit]
- Number Only If only one isomer exists; otherwise:
- First lower case suffix (a-f):
- a Suffix Cl2 central carbon substitution
- b Suffix Cl, F central carbon substitution
- c Suffix F2 central carbon substitution
- d Suffix Cl, H central carbon substitution
- e Suffix F, H central carbon substitution
- f Suffix H2 central carbon substitution
- 2nd Lower Case Suffix (a, b, c, etc.) Indicates increasingly unsymmetrical isomers
Propene derivatives[edit]
- First lower case suffix (x, y, z):
- x Suffix Cl substitution on central atom
- y Suffix F substitution on central atom
- z Suffix H substitution on central atom
- Second lower case suffix (a-f):
- a Suffix =CCl2 methylene substitution
- b Suffix =CClF methylene substitution
- c Suffix =CF2 methylene substitution
- d Suffix =CHCl methylene substitution
- e Suffix =CHF methylene substitution
- f Suffix =CH2 methylene substitution
Blends[edit]
- Upper Case Suffix (A,B,C,etc.) Same blend with different compositions of refrigerants
Miscellaneous[edit]
- R-Cxxx Cyclic compound
- R-Exxx Ether group is present
- R-CExxx Cyclic compound with an ether group
- R-4xx/5xx + Upper Case Suffix (A,B,C,etc.) Same blend with different composition of refrigerants
- R-6xx + Lower Case Letter Indicates increasingly unsymmetrical isomers
- 7xx/7xxx + Upper Case Letter Same molar mass, different compound
- R-xxxxB# Bromine is present with the number after B indicating how many bromine atoms
- R-xxxxI# Iodine is present with the number after I indicating how many iodine atoms
- R-xxx(E) Trans Molecule
- R-xxx(Z) Cis Molecule
For example, R-134a has 2 carbon atoms, 2 hydrogen atoms, and 4 fluorine atoms, an empirical formula of tetrafluoroethane. The "a" suffix indicates that the isomer is unbalanced by one atom, giving 1,1,1,2-Tetrafluoroethane. R-134 (without the "a" suffix) would have a molecular structure of 1,1,2,2-Tetrafluoroethane.
The same numbers are used with an R- prefix for generic refrigerants, with a "Propellant" prefix (e.g., "Propellant 12") for the same chemical used as a propellant for an aerosol spray, and with trade names for the compounds, such as "Freon 12". Recently, a practice of using abbreviations HFC- for hydrofluorocarbons, CFC- for chlorofluorocarbons, and HCFC- for hydrochlorofluorocarbons has arisen, because of the regulatory differences among these groups.[citation needed]
Refrigerant safety[edit]
ASHRAE Standard 34, Designation and Safety Classification of Refrigerants, assigns safety classifications to refrigerants based upon toxicity and flammability.
Using safety information provided by producers, ASHRAE assigns a capital letter to indicate toxicity and a number to indicate flammability. The letter "A" is the least toxic and the number 1 is the least flammable.[79]
See also[edit]
References[edit]
- ^ a b Pearson, S. Forbes. "Refrigerants Past, Present and Future" (PDF). R744. Archived from the original (PDF) on 2018-07-13. Retrieved 2021-03-30.
- ^ a b c "Finally, a replacement for R123?". Cooling Post. 17 October 2013.
- ^ https://asrjetsjournal.org/index.php/American_Scientific_Journal/article/download/3297/1244/
- ^ a b Tomczyk, John (1 May 2017). "What's the Latest with R-404A?". achrnews.com.
- ^ "Air Conditioners, Dehumidifiers, and R-410A Refrigerant". Sylvane. 1 July 2011. Retrieved 27 July 2023.
- ^ Protection, United States Congress Senate Committee on Environment and Public Works Subcommittee on Environmental (May 14, 1987). "Clean Air Act Amendments of 1987: Hearings Before the Subcommittee on Environmental Protection of the Committee on Environment and Public Works, United States Senate, One Hundredth Congress, First Session, on S. 300, S. 321, S. 1351, and S. 1384 ..." U.S. Government Printing Office – via Google Books.
- ^ Fluorinated Hydrocarbons—Advances in Research and Application (2013 ed.). ScholarlyEditions. June 21, 2013. p. 179. ISBN 9781481675703 – via Google Books.
- ^ Whitman, Bill; Johnson, Bill; Tomczyk, John; Silberstein, Eugene (February 25, 2008). Refrigeration and Air Conditioning Technology. Cengage Learning. p. 171. ISBN 978-1111803223 – via Google Books.
- ^ "Scroll Chillers: Conversion from HCFC-22 to HFC-410A and HFC-407C" (PDF). Archived from the original (PDF) on 2021-07-20. Retrieved 2021-03-29.
- ^ "What's Happening With R-134a? | 2017-06-05 | ACHRNEWS | ACHR News". achrnews.com.
- ^ "Conversion R12/R134a" (PDF). Behr Hella Service GmbH. 1 October 2005. Retrieved 27 July 2023.
- ^ "R-407A Gains SNAP OK". achrnews.com (Press release). 22 June 2009.
- ^ "June 26, 2009: Emerson Approves R-407A, R-407C for Copeland Discus Compressors". achrnews.com.
- ^ "Taking New Refrigerants to the Peak". achrnews.com.
- ^ Koenig, H. (31 December 1995). "R502/R22 - replacement refrigerant R507 in commercial refrigeration; R502/R22 - Ersatzkaeltemittel R507 in der Gewerbekuehlung. Anwendungstechnik - Kaeltemittel".
- ^ Linton, J. W.; Snelson, W. K.; Triebe, A. R.; Hearty, P. F. (31 December 1995). "System performance comparison of R-507 with R-502". OSTI 211821.
- ^ "Daikin reveals details of R32 VRV air conditioner". Cooling Post. 6 February 2020.
- ^ a b "Refrigerant blends to challenge hydrocarbon efficiencies". Cooling Post. 22 December 2019.
- ^ "An HVAC Technician's Guide to R-454B". achrnews.com.
- ^ "The truth about new automotive A/C refrigerant R1234YF". 25 July 2018.
- ^ Kontomaris, Konstantinos (2014). "HFO-1336mzz-Z: High Temperature Chemical Stability and Use as A Working Fluid in Organic Rankine Cycles". International Refrigeration and Air Conditioning Conference.
Paper 1525
- ^ "Trane adopts new low GWP refrigerant R514A". Cooling Post. 15 June 2016.
- ^ "R404A – the alternatives". Cooling Post. 26 February 2014.
- ^ "Carrier expands R1234ze chiller range". Cooling Post. 20 May 2020.
- ^ "Carrier confirms an HFO refrigerant future". Cooling Post. 5 June 2019.
- ^ "Greenfreeze: A revolution in domestic refrigeration". ecomall.com. Retrieved 2022-07-04.
- ^ a b "Happy birthday, Greenfreeze!". Greenpeace. 25 March 2013. Archived from the original on 2020-04-08. Retrieved 8 June 2015.
- ^ "Ozone Secretariat". United Nations Environment Programme. Archived from the original on 12 April 2015.
- ^ Gunkel, Christoph (13 September 2013). "Öko-Coup aus Ostdeutschland". Der Spiegel (in German). Retrieved 4 September 2015.
- ^ Maté, John (2001). "Making a Difference: A Case Study of the Greenpeace Ozone Campaign". Review of European Community & International Environmental Law. 10 (2): 190–198. doi:10.1111/1467-9388.00275.
- ^ Benedick, Richard Elliot Ozone Diplomacy Cambridge, MA: Harvard University 1991.
- ^ Honeywell International, Inc. (2010-07-09). "Comment on EPA Proposed Rule Office of Air and Radiation Proposed Significant New Alternatives Policy (SNAP) Protection of Stratospheric Ozone: Listing of Substitutes for Ozone-Depleting Substances – Hydrocarbon Refrigerants" (PDF).
- ^ "Discurso de Frank Guggenheim no lançamento do Greenfreeze | Brasil". Greenpeace.org. Archived from the original on 24 September 2015. Retrieved 10 June 2015.
- ^ "Der Greenfreeze - endlich in den USA angekommen". Greenpeace.de (in German). 28 December 2011. Retrieved 10 June 2015.
- ^ "Complying With The Section 608 Refrigerant Recycling Rule | Ozone Layer Protection - Regulatory Programs". Epa.gov. 21 April 2015. Retrieved 10 June 2015.
- ^ a b "Greenfreeze: a Revolution in Domestic Refrigeration". ecomall.com. Retrieved 8 June 2015.
- ^ "Company background". Archived from the original on 2020-02-20. Retrieved 2021-03-15.
- ^ Safeguarding the ozone layer and the global climate System: issues related to Hydrofluorocarbons and Perfluorocarbons (Report). IPCC/TEAP. 2005.
- ^ Crowley, Thomas J. (2000). "Causes of Climate Change over the Past 1000 Years". Science. 289 (5477): 270–277. Bibcode:2000Sci...289..270C. doi:10.1126/science.289.5477.270. PMID 10894770.
- ^ "2010 to 2015 government policy: environmental quality". GOV.UK. 8 May 2015. Retrieved 10 June 2015.
- ^ "PepsiCo Brings First Climate-Friendly Vending Machines to the U.S." phx.corporate-ir.net. Retrieved 8 June 2015.
- ^ "Climate-Friendly Greenfreezers Come to the United States". WNBC. 2 October 2008. Retrieved 8 June 2015.
- ^ Data, Reports and (7 August 2020). "Natural Refrigerants Market To Reach USD 2.88 Billion By 2027 | Reports and Data". GlobeNewswire News Room (Press release). Retrieved 17 December 2020.
- ^ Harris, Catharine. "Anti-inhalant Abuse Campaign Targets Building Codes: 'Huffing’ of Air Conditioning Refrigerant a Dangerous Risk." The Nation's Health. American Public Health Association, 2010. Web. 5 December 2010. https://www.thenationshealth.org/content/39/4/20
- ^ IPCC AR6 WG1 Ch7 2021
- ^ "GreenFreeze". Greenpeace.
- ^ "Significant New Alternatives Program: Substitutes in Household Refrigerators and Freezers". Epa.gov. 13 November 2014. Retrieved 4 June 2018.
- ^ Berwald, Juli (29 April 2019). "One overlooked way to fight climate change? Dispose of old CFCs". National Geographic - Environment. Archived from the original on April 29, 2019. Retrieved 30 April 2019.
- ^ Butler J. and Montzka S. (2020). "The NOAA Annual Greenhouse Gas Index (AGGI)". NOAA Global Monitoring Laboratory/Earth System Research Laboratories.
- ^ Environment, U. N. (31 October 2019). "New guidelines for air conditioners and refrigerators set to tackle climate change". UN Environment. Retrieved 30 March 2020.
- ^ Rosenthal, Elisabeth; Lehren, Andrew (20 June 2011). "Relief in Every Window, but Global Worry Too". The New York Times. Retrieved 21 June 2012.
- ^ a b Yadav et al 2022
- ^ a b c BSRIA 2020
- ^ a b c d e f g h IPCC AR5 WG1 Ch8 2013, pp. 714, 731–737
- ^ "European Commission on retrofit refrigerants for stationary applications" (PDF). Archived from the original on August 5, 2009. Retrieved 2010-10-29.
{{cite web}}: CS1 maint: unfit URL (link) - ^ "Protection of Stratospheric Ozone: Hydrocarbon Refrigerants" (PDF). Environment Protection Agency. Retrieved 5 August 2018.
- ^ ARB 2022
- ^ GM to Introduce HFO-1234yf AC Refrigerant in 2013 US Models
- ^ "The Coca-Cola Company Announces Adoption of HFC-Free Insulation in Refrigeration Units to Combat Global Warming". The Coca-Cola Company. 5 June 2006. Archived from the original on 1 November 2013. Retrieved 11 October 2007.
- ^ "Modine reinforces its CO2 research efforts". R744.com. 28 June 2007. Archived from the original on 10 February 2008.
- ^ Longo, Giovanni A.; Mancin, Simone; Righetti, Giulia; Zilio, Claudio (2015). "HFC32 vaporisation inside a Brazed Plate Heat Exchanger (BPHE): Experimental measurements and IR thermography analysis". International Journal of Refrigeration. 57: 77–86. doi:10.1016/j.ijrefrig.2015.04.017.
- ^ May 2010 TEAP XXI/9 Task Force Report
- ^ "1:Update on Ozone-Depleting Substances (ODSs) and Other Gases of Interest to the Montreal Protocol". Scientific assessment of ozone depletion: 2018 (PDF) (Global Ozone Research and Monitoring Project–Report No. 58 ed.). Geneva, Switzerland: World Meteorological Organization. 2018. p. 1.10. ISBN 978-1-7329317-1-8. Retrieved 22 November 2020.
- ^ [1] Chemours M099 as R22 Replacement
- ^ [2] Management of HCFC-123 through the Phaseout and Beyond | EPA | Published August 2020 | Retrieved Dec. 18, 2021
- ^ [3] Refrigerant R11 (R-11), Freon 11 (Freon R-11) Properties & Replacement
- ^ [4] R-454B XL41 refrigerant fact & info sheet
- ^ [5] R-454B emerges as a replacement for R-410A | ACHR News (Air Conditioning, Heating, Refrigeration News)
- ^ [6] Ccarrier introduces [R-454B] Puron Advance™ as the next generation refrigerant for ducted residential, light commercial products in North America | Indianapolis - 19 December 2018
- ^ [7] Johnson Controls selects R-454B as future refrigerant for new HVAC equipment | 27 May 2021
- ^ [8] A conversation on refrigerants | ASHRAE Journal, March 2021 | page 30, column 1, paragraph 2
- ^ [9] Opteon™ XP30 (R-514A) refrigerant
- ^ [10] Trane adopts new low GWP refrigerant R514A | 15 June 2016
- ^ "Emissions of Greenhouse Gases in the United States 1998 - Executive Summary". 18 August 2000. Archived from the original on 18 August 2000.
- ^ "Frequently Asked Questions on Section 608". Environment Protection Agency. Retrieved 20 December 2013.
- ^ "US hydrocarbons". Retrieved 5 August 2018.
- ^ "42 U.S. Code § 7671g - National recycling and emission reduction program". LII / Legal Information Institute.
- ^ ASHRAE; UNEP (Nov 2022). "Designation and Safety Classification of Refrigerants" (PDF). ASHRAE. Retrieved 1 July 2023.
- ^ "Update on New Refrigerants Designations and Safety Classifications" (PDF). American Society of Heating, Refrigerating and Air-Conditioning Engineers (ASHRAE). April 2020. Archived from the original (PDF) on February 13, 2023. Retrieved October 22, 2022.
Sources[edit]
IPCC reports[edit]
- IPCC (2013). Stocker, T. F.; Qin, D.; Plattner, G.-K.; Tignor, M.; et al. (eds.). Climate Change 2013: The Physical Science Basis (PDF). Contribution of Working Group I to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change. Cambridge, United Kingdom and New York, NY, USA: Cambridge University Press. ISBN 978-1-107-05799-9. (pb: 978-1-107-66182-0). Fifth Assessment Report - Climate Change 2013
- Myhre, G.; Shindell, D.; Bréon, F.-M.; Collins, W.; et al. (2013). "Chapter 8: Anthropogenic and Natural Radiative Forcing" (PDF). Climate Change 2013: The Physical Science Basis. Contribution of Working Group I to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change. pp. 659–740.
- IPCC (2021). Masson-Delmotte, V.; Zhai, P.; Pirani, A.; Connors, S. L.; et al. (eds.). Climate Change 2021: The Physical Science Basis (PDF). Contribution of Working Group I to the Sixth Assessment Report of the Intergovernmental Panel on Climate Change. Cambridge University Press (In Press).
- Forster, Piers; Storelvmo, Trude (2021). "Chapter 7: The Earth's Energy Budget, Climate Feedbacks, and Climate Sensitivity" (PDF). IPCC AR6 WG1 2021.
Other[edit]
- "High GWP refrigerants". California Air Resources Board. Retrieved 13 February 2022.
- "BSRIA's view on refrigerant trends in AC and Heat Pump segments". 2020. Retrieved 2022-02-14.
- Yadav, Saurabh; Liu, Jie; Kim, Sung Chul (2022). "A comprehensive study on 21st-century refrigerants - R290 and R1234yf: A review". International Journal of Heat and Mass Transfer. 122: 121947. doi:10.1016/j.ijheatmasstransfer.2021.121947. S2CID 240534198.
