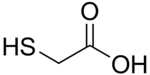
Thioglycolic acid
| |

| |
| Names | |
|---|---|
| Preferred IUPAC name
Sulfanylacetic acid | |
| Other names
2-Sulfanylacetic acid
2-Mercaptoacetic acid Acetyl mercaptan Mercaptoacetate Mercaptoacetic acid Thioglycolic acid Thiovanic acid[2] | |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.000.616 |
| KEGG | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C2H4O2S | |
| Molar mass | 92.11 g·mol−1 |
| Appearance | colorless, clear liquid[3] |
| Odor | strong, disagreeable[3] |
| Density | 1.32 g/cm3 |
| Melting point | −16 °C (3 °F; 257 K) |
| Boiling point | 96 °C (205 °F; 369 K) at 5 mmHg |
| miscible[3] | |
| Vapor pressure | 10 mmHg (17.8 °C)[3] |
| −50.0·10−6 cm3/mol | |
| Hazards | |
| GHS labelling: | |
 
| |
| H301+H311+H331, H314 | |
| Flash point | > 110 °C; 230 °F; 383 K[3] |
| Explosive limits | 5.9%[3] |
| NIOSH (US health exposure limits): | |
PEL (Permissible)
|
none[3] |
REL (Recommended)
|
TWA 1 ppm (4 mg/m3) [skin][3] |
IDLH (Immediate danger)
|
N.D.[3] |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Thioglycolic acid (TGA) is the organic compound HSCH2CO2H. TGA is often called mercaptoacetic acid (MAA). It contains both a thiol (mercaptan) and carboxylic acid functional groups. It is a colorless liquid with a strongly unpleasant odor.[4][5] TGA is miscible with polar organic solvents.[6][7]
Uses[edit]
TGA is used as a chemical depilatory and is still used as such, especially in salt forms, including calcium thioglycolate and sodium thioglycolate. TGA is the precursor to ammonium thioglycolate, which is used for permanents. TGA and its derivatives break the disulfide bonds in the cortex of hair. One reforms these broken bonds in giving hair a "perm". Alternatively and more commonly, the process leads to depilation, as is done commonly in leather processing. It is also used as an acidity indicator, manufacturing of thioglycolates, and in bacteriology for preparation of thioglycolate media.[7] Thioglycolysis reactions are used on condensed tannins to study their structure.[8][9][10][11]
TGA has also been used to soften nails, either to reshape pincer nails into the correct position[12] or to help topical antifungals penetrate the nail.[13]
Organotin derivatives of thioglycolic acid isooctyl esters are widely used as stabilizers for PVC. These species have the formula R2Sn(SCH2CO2C8H17)2.[7]
Sodium thioglycolate is a component of thioglycolate broth, a special bacterial growth media. It is also used in so-called "fallout remover"[14] or "wheel cleaner" to remove iron oxide residue from wheels.[15] Ferrous iron combines with thioglycolate to form red-violet[16] ferric thioglycolate.[17][18]
Production[edit]
Thioglycolic acid is prepared by reaction of sodium or potassium chloroacetate with alkali metal hydrosulfide in aqueous medium.[19] It can be also prepared via the Bunte salt obtained by reaction of sodium thiosulfate with chloroacetic acid:[7][20]
- ClCH2CO2H + Na2S2O3 → Na[O3S2CH2CO2H] + NaCl
- Na[O3S2CH2CO2H] + H2O → HSCH2CO2H + NaHSO4
Reactions[edit]
Thioglycolic acid with a pKa of 3.83 [7] is an acid about 10 times stronger than acetic acid (pKa 4.76):
- HSCH2CO2H → HSCH2CO2− + H+
The second ionization has a pKa of 9.3:
- HSCH2CO2− → −SCH2CO2− + H+
Thioglycolic acid is a reducing agent, especially at higher pH. It oxidizes to the corresponding disulfide (2-[(carboxymethyl)disulfanyl]acetic acid or dithiodiglycolic acid):
- 2 HSCH2CO2H + "O" → [SCH2CO2H]2 + H2O
With metal ions[edit]
Thioglycolic acid, usually as its dianion, forms complexes with metal ions. Such complexes have been used for the detection of iron, molybdenum, silver, and tin. Thioglycolic acid reacts with diethyl acetylmalonate to form acetylmercaptoacetic acid and diethyl malonate, the reducing agent in the conversion of Fe(III) to Fe(II).[21]
History[edit]
Scientist David R. Goddard, in the early 1930s, identified TGA as a useful reagent for reducing the disulfide bonds in proteins, including keratin (hair protein), while studying why protease enzymes could not easily digest hair, nails, feathers, and such. He realized that while the disulfide bonds, which stabilize proteins by cross-linking, were broken, the structures containing these proteins could be reshaped easily, and that they would retain this shape after the disulfide bonds were allowed to re-form.[22] TGA was developed in the 1940s for use as a chemical depilatory.
Safety and detection[edit]
The LD50 (oral, rat) is 261 mg/kg,[7] LC50 inhalation for rat is 21 mg/m3 for 4 h, and LD50 dermal for rabbit is 848 mg/kg.[23] Mercaptoacetic acid in hair waving and depilatory products containing other mercapto acids can be identified by using thin-layer chromatography and gas chromatography.[24][25] MAA also has been identified by using potentiometric titration with silver nitrate solution.[26]
See also[edit]
References[edit]
- ^ Merck Index, 11th Edition, 9265
- ^ NIOSH Pocket Guide to Chemical Hazards. CDC.
- ^ a b c d e f g h i NIOSH Pocket Guide to Chemical Hazards. "#0610". National Institute for Occupational Safety and Health (NIOSH).
- ^ "What exactly is Mercaptan?". Independent.co.uk. 22 January 2013. Archived from the original on 2022-05-07.
- ^ "Thioglycolic acid :Understanding the risk of specific chemicals of interest - PREVOR". 11 September 2014.
- ^ The Merck index, 14th ed.; O’Neil, Maryadele J., Ed.; Merck & Co., Inc.: Whitehouse Station, NJ, 2006; p. 9342.
- ^ a b c d e f Robert Rippel "Mercaptoacetic Acid and Derivatives" in Ullmann's Encyclopedia of Industrial Chemistry, 2012, Wiley-VCH, Weinheim. doi:10.1002/14356007.a16_265.
- ^ Vernhet A, Dubascoux S, Cabane B, Fulcrand H, Dubreucq E, Poncet-Legrand C (2011-09-01). "Characterization of oxidized tannins: comparison of depolymerization methods, asymmetric flow field-flow fractionation and small-angle X-ray scattering". Analytical and Bioanalytical Chemistry. 401 (5): 1559–1569. doi:10.1007/s00216-011-5076-2. ISSN 1618-2650. PMID 21573842. S2CID 4645218.
- ^ Suc L, Rigou P, Mouls L (2021-03-17). "Detection and Identification of Oxidation Markers of the Reaction of Grape Tannins with Volatile Thiols Commonly Found in Wine". Journal of Agricultural and Food Chemistry. 69 (10): 3199–3208. doi:10.1021/acs.jafc.0c07163. ISSN 0021-8561. PMID 33657810. S2CID 232112725.
- ^ Trivelato P, Mayer C, Barakat A, Fulcrand H, Aouf C (2016-08-01). "Douglas bark dry fractionation for polyphenols isolation: From forestry waste to added value products". Industrial Crops and Products. 86: 12–15. doi:10.1016/j.indcrop.2016.03.014. ISSN 0926-6690.
- ^ Ben Aziz M, Mouls L, Fulcrand H, Douieb H, Hajjaj H (2017-05-01). "Phenolic compounds of Moroccan red press wines: Influence of fining agents and micro-oxygenation treatments". LWT. 78: 143–150. doi:10.1016/j.lwt.2016.12.034. ISSN 0023-6438.
- ^ Okada, K., Okada, E. (2012). "Novel treatment using thioglycolic acid for pincer nails". The Journal of Dermatology. 39 (12). Wiley: 996–999. doi:10.1111/j.1346-8138.2012.01670.x. PMID 22963208. S2CID 7413716.
- ^ Gregorí Valdes, B. S., Serro, A. P., Gordo, P. M., Silva, A., Gonçalves, L., Salgado, A., Marto, J., Baltazar, D., Santos, R. G. dos, Bordado, J. M., Ribeiro, H. M. (2017). "New Polyurethane Nail Lacquers for the Delivery of Terbinafine: Formulation and Antifungal Activity Evaluation". Journal of Pharmaceutical Sciences. 106 (6). Elsevier BV: 1570–1577. doi:10.1016/j.xphs.2017.02.017. PMID 28263845.
- ^ "What is a fallout remover". DetailingWiki. 2016-03-17. Retrieved 2016-06-14.
- ^ "SONAX Xtreme Wheel Cleaner" Safety data sheet
- ^ McKetta JJ Jr (1981-01-01). Encyclopedia of Chemical Processing and Design. Vol. 12: Corrosion to Cottonseed. CRC Press. p. 103. ISBN 9780824724627.
- ^ Lyons E (1927-08-01). "Thioglycolic Acid as a Color Test for Iron". Journal of the American Chemical Society. 49 (8): 1916–1920. doi:10.1021/ja01407a010. ISSN 0002-7863.
- ^ "Limit Test of Iron". Web Formulas. Retrieved 2016-06-14.
- ^ Cosmetic, Toiletry, and Fragrance Association (CTFA). Thioglycolic Acid. 1987;(1987a). Submission of unpublished data by CTFA Code No. 3-25-2
- ^ Saeed M. Hameed N. Madan V. Mansoor S. Preparation and Mechanisms studies of Thioglycolic Acid. Pak. J. Sci. Ind. Res. 1992, 35: 131-132
- ^ Lee CW, Phil M. The detection of iron traces on hands by ferrozine sparys: a report on the sensitivity and interference of the method and recommended procedure in forensic science investigation. J Forensic Sci. 1986, 31:920-930.
- ^ National Academies Press:Biographical Memoirs:David Rockwell Goddard:by Ralph O. Erickson
- ^ Sigma-Aldrich MSDS. http://www.sigmaaldrich.com/safety-center.html?cm_sp=Search-_-MSDS-_-MSDS1 (accessed Nov 10, 2013). Product Number – T3758
- ^ Goetz N, Gataud P, Bore P. Determination of mercaptoacetic acid in hair waving and depilatory products. Analyst. 1979,104:1062-1069
- ^ Goetz N, Gataud P, Bore P. Gas-chromatographic determination of mercaptoacetic acid in hair-waving and diplatory products. Cosmet Sci Technol Ser. 1985, 4:65-79.
- ^ Vandeputte M, Dryon L, Van Den Winkel P, Mertens J, Massart DL. Determination of thioglycolic acid using a silver sulfide single crystal electrode. Analysis. 1975,3:500-504.
Further reading[edit]
- Okada K, Okada E. Novel treatment using thioglycolic acid for pincer nails. J. Dermatol. 2012, volume 39, pp. 996-999.
