Transplant glomerulopathy
| Transplant glomerulopathy | |
|---|---|
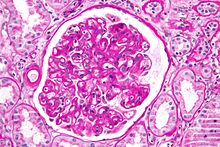 | |
| Micrograph showing a glomerulus with changes characteristic of a transplant glomerulopathy. PAS stain. | |
| Specialty | Nephrology |
Transplant glomerulopathy (TG) is a morphologic lesion of renal allografts that is histologically identified by glomerular basement membrane (GBM) duplication and/or multilayering.[1] Proteinuria, hypertension, and deteriorating graft function are the hallmarks of TG.[2]
Acute rejection, pre-transplant antibody levels, and de novo HLA antibodies are all linked to TG. There are further risks associated with HLA class II and/or donor-specific antibodies.[2]
Five years after transplant, TG is present in 5–10% of renal allografts; in rare cases, protocol biopsies may reveal TG as a subclinical finding. With chronic alloantibody-mediated injury, the lesion is particularly associated with a poor prognosis for the outcome of the graft.[3]
Signs and symptoms[edit]
Transplant glomerulopathy (TG) may present as clinically silent and only be detected on biopsy.[4]
Low-level proteinuria and/or a slight decline in glomerular filtration rate are examples of early clinical manifestations.[5] However, as the lesion worsens, hypertension, a drop in glomerular filtration rate, and an increase in proteinuria, often into the nephrotic range, may occur.[6]
Complications[edit]
Numerous studies have linked reduced allograft survival to transplant glomerulopathy.[7][8][9]
Causes[edit]
TG is thought to be a morphologic expression of chronic antibody-mediated rejection (ABMR) in most cases and is well-documented as being correlated with a buildup of donor-specific antibodies (DSAs), particularly against HLA class II antigens. But TG is not exclusive to chronic ABMR; in one-third to one-fourth of cases, thrombotic microangiopathy (TMA), hepatitis C, and possibly T cell-mediated rejection (TCMR) appear to be the alternative etiologies.[10]
Mechanism[edit]
Transplant glomerulopathy (TG) is a slowly developing, long-lasting damage to the glomerular capillary walls that is thought to result from long-term glomerular endothelial activation and injury. The glomerular basement membrane is also repaired and remodeled in conjunction with this condition.[11] Reduced podocyte density and glomerular epithelial cell stress are also factors in transplant-related glomerular injury, which includes increased urine podocyte excretion in TG and significant podocyte detachment, both of which are linked to progressive glomerulosclerosis.[12]
Out of all the suggested mechanisms, the one that has been documented the most is the correlation between donor-specific antibody against the human leukocyte antigen (anti-HLA-DSA), antibody-mediated mediated rejection (ABMR), and endothelial damage that results in the development of TG.[13]
Clinical investigations have also demonstrated the connection between TG and humoral alloimmunity.[13]TG is first thought to be the characteristic lesion of chronic ABMR. Compared to other transplants, HLA-incompatible transplants that need to be desensitized have a higher prevalence of TG.[14] Patients with TG have consistently been found to have an increased prevalence of donor-specific anti-HLA antibodies (DSA) and an increased incidence of antecedent or concurrent ABMR.[15][16] Apart from preformed DSA, de novo DSA development has also been linked to TG, indicating that pathogenetic relevance to TG also extends to either new or recurrent endothelial damage resulting from de novo DSA.[17]
The most compelling evidence for the theory that non-HLA antibodies could cause ABMR has come from studies on HLA identical renal allograft recipients who developed anti-donor endothelial reactive antibodies.[18] According to data, non-HLA auto- and alloantibodies may contribute to chronic graft loss either by themselves through cytotoxicity or in conjunction with anti-HLA antibodies.[19]
Clinical biopsy studies showing TG lesions in the absence of discernible acute or chronic ABMR have raised the possibility that non-humoral mechanisms may be responsible for the TG pathogenesis in these cases.[20]
It has long been believed that humoral immunity is the cause of microvascular inflammation (MVI), which occurs in conjunction with TG and ABMR.[21]
Diagnosis[edit]
Transplant glomerulopathy (TG) is best recognized histologically by reduplications or "double contours" of the glomerular basement membrane in peripheral glomerular capillary loops; basement membrane stains (methanamine silver and periodic acid-Schiff) work best for this kind of identification. Since it is a focal lesion in the early stages and only affects a small percentage of glomeruli, sufficient glomerular sampling is required. The lesion becomes more diffuse over time, accompanied by secondary focal segmental and global glomerulosclerosis and hyalinosis, variable mesangial matrix expansion (without the formation of mesangial nodules or significant mesangial cell proliferation), and associated glomerular hypertrophy. Severe nodular hyaline arteriolosclerosis, arterial fibrous intimal thickening, and progressive interstitial fibrosis and tubular atrophy are typically linked to advanced TG.[3]
According to the Banff '97 criteria, double contours had to be present in at least 10% of the capillary loops in the most affected glomerulus in order to diagnose transplant glomerulopathy.[22]
Immunofluorescence (IF) results for TG show no diagnostic staining for IgG, IgA, or C1q, as well as mild-to-moderate glomerular mesangial along with capillary wall deposition of IgM and C3.[3]
Differential diagnosis[edit]
Differential diagnosis (DDX) of transplant glomerulopathy includes thrombotic microangiopathies, recurrent membranoproliferative glomerulonephritis, and ischemia.[23]
Treatment[edit]
The general management of patients with TG involves renin-angiotensin-aldosterone (RAAS) blockade, blood pressure and diabetes control, weight loss, and other strategies to lower intra-glomerular pressure.[13]
See also[edit]
References[edit]
- ^ Haas, Mark (2015). "Transplant Glomerulopathy". Journal of the American Society of Nephrology. 26 (6). Ovid Technologies (Wolters Kluwer Health): 1235–1237. doi:10.1681/asn.2014090945. ISSN 1046-6673. PMC 4446884. PMID 25388221.
- ^ a b Fotheringham, James; Angel, Carole A.; McKane, William (July 10, 2009). "Transplant Glomerulopathy: Morphology, Associations and Mechanism". Nephron Clinical Practice. 113 (1). S. Karger AG: c1–c7. doi:10.1159/000228069. ISSN 1660-2110. PMID 19590229.
- ^ a b c Gibson, Ian W. (March 30, 2021). "Transplant Glomerulopathy: Importance of Ultrastructural Examination". Glomerular Diseases. 1 (2). S. Karger AG: 68–81. doi:10.1159/000513522. ISSN 2673-3633. PMC 9677739. PMID 36751426.
- ^ Gloor, J.M.; Sethi, S.; Stegall, M.D.; Park, W.D.; Moore, S.B.; DeGoey, S.; Griffin, M.D.; Larson, T.S.; Cosio, F.G. (2007). "Transplant Glomerulopathy: Subclinical Incidence and Association with Alloantibody". American Journal of Transplantation. 7 (9). Elsevier BV: 2124–2132. doi:10.1111/j.1600-6143.2007.01895.x. ISSN 1600-6135. PMID 17608832.
- ^ Filippone, Edward J; McCue, Peter A; Farber, John L (2018). "Transplant glomerulopathy". Modern Pathology. 31 (2). Elsevier BV: 235–252. doi:10.1038/modpathol.2017.123. ISSN 0893-3952. PMID 29027535.
- ^ Banfi, Giovanni; Villa, Margarita; Cresseri, Donata; Ponticelli, Claudio (November 27, 2005). "The Clinical Impact of Chronic Transplant Glomerulopathy in Cyclosporine Era". Transplantation. 80 (10). Ovid Technologies (Wolters Kluwer Health): 1392–1397. doi:10.1097/01.tp.0000181167.88133.d2. ISSN 0041-1337. PMID 16340780.
- ^ Kieran, Niamh; Wang, Xiaotong; Perkins, James; Davis, Connie; Kendrick, Elizabeth; Bakthavatsalam, Ramaswamy; Dunbar, Nancy; Warner, Paul; Nelson, Karen; Smith, Kelly D.; Nicosia, Roberto F.; Alpers, Charles E.; Leca, Nicolae; Kowalewska, Jolanta (2009). "Combination of Peritubular C4d and Transplant Glomerulopathy Predicts Late Renal Allograft Failure". Journal of the American Society of Nephrology. 20 (10). Ovid Technologies (Wolters Kluwer Health): 2260–2268. doi:10.1681/asn.2009020199. ISSN 1046-6673. PMC 2754096. PMID 19729438.
- ^ Issa, Naim; Cosio, Fernando G.; Gloor, James M.; Sethi, Sanjeev; Dean, Patrick G.; Moore, S Breanndan; DeGoey, Steve; Stegall, Mark D. (September 15, 2008). "Transplant Glomerulopathy: Risk and Prognosis Related to Anti-Human Leukocyte Antigen Class II Antibody Levels". Transplantation. 86 (5). Ovid Technologies (Wolters Kluwer Health): 681–685. doi:10.1097/tp.0b013e3181837626. ISSN 0041-1337. PMID 18791449.
- ^ Lesage, Julie; Noël, Réal; Lapointe, Isabelle; Côté, Isabelle; Wagner, Eric; Désy, Olivier; Caumartin, Yves; Agharazii, Mohsen; Batal, Ibrahim; Houde, Isabelle; De Serres, Sacha A. (2015). "Donor-Specific Antibodies, C4d and Their Relationship With the Prognosis of Transplant Glomerulopathy". Transplantation. 99 (1). Ovid Technologies (Wolters Kluwer Health): 69–76. doi:10.1097/tp.0000000000000310. ISSN 0041-1337. PMID 25073036.
- ^ Haas, Mark (2015). "Transplant Glomerulopathy". Journal of the American Society of Nephrology. 26 (6). Ovid Technologies (Wolters Kluwer Health): 1235–1237. doi:10.1681/asn.2014090945. ISSN 1046-6673. PMC 4446884. PMID 25388221.
- ^ Sis, B.; Campbell, P.M.; Mueller, T.; Hunter, C.; Cockfield, S.M.; Cruz, J.; Meng, C.; Wishart, D.; Solez, K.; Halloran, P.F. (2007). "Transplant Glomerulopathy, Late Antibody-Mediated Rejection and the ABCD Tetrad in Kidney Allograft Biopsies for Cause". American Journal of Transplantation. 7 (7). Elsevier BV: 1743–1752. doi:10.1111/j.1600-6143.2007.01836.x. ISSN 1600-6135. PMID 17564636.
- ^ Yang, Yan; Hodgin, Jeffrey B.; Afshinnia, Farsad; Wang, Su Q.; Wickman, Larysa; Chowdhury, Mahboob; Nishizono, Ryuzoh; Kikuchi, Masao; Huang, Yihung; Samaniego, Milagros; Wiggins, Roger C. (2015). "The Two Kidney to One Kidney Transition and Transplant Glomerulopathy". Journal of the American Society of Nephrology. 26 (6). Ovid Technologies (Wolters Kluwer Health): 1450–1465. doi:10.1681/asn.2014030287. ISSN 1046-6673. PMC 4446871. PMID 25388223.
- ^ a b c Gokhale, Avantee; Chancay, Jorge; Shapiro, Ron; Randhawa, Parmjeet; Menon, Madhav C. (February 6, 2021). "Chronic transplant glomerulopathy: New insights into pathogenesis". Clinical Transplantation. 35 (3). Wiley: e14214. doi:10.1111/ctr.14214. ISSN 0902-0063. PMC 8281330. PMID 33389755.
- ^ Sharif, Adnan; Kraus, Edward S.; Zachary, Andrea A.; Lonze, Bonnie E.; Nazarian, Susanna M.; Segev, Dorry L.; Alachkar, Nada; Arend, Lois J.; Bagnasco, Serena M.; Racusen, Lorraine C.; Montgomery, Robert A. (March 15, 2014). "Histologic Phenotype on 1-Year Posttransplantation Biopsy and Allograft Survival in HLA-Incompatible Kidney Transplants". Transplantation. 97 (5). Ovid Technologies (Wolters Kluwer Health): 541–547. doi:10.1097/01.tp.0000442513.27641.7e. ISSN 0041-1337.
- ^ Gloor, J.M.; Cosio, F.G.; Rea, D.J.; Wadei, H.M.; Winters, J.L.; Moore, S.B.; DeGoey, S.R.; Lager, D.J.; Grande, J.P.; Stegall, M.D. (2006). "Histologic Findings One Year After Positive Crossmatch or ABO Blood Group Incompatible Living Donor Kidney Transplantation". American Journal of Transplantation. 6 (8). Elsevier BV: 1841–1847. doi:10.1111/j.1600-6143.2006.01416.x. ISSN 1600-6135. PMID 16780546.
- ^ Loupy, A.; Suberbielle-Boissel, C.; Hill, G.S.; Lefaucheur, C.; Anglicheau, D.; Zuber, J.; Martinez, F.; Thervet, E.; Méjean, A.; Charron, D.; Duong van Huyen, J.P.; Bruneval, P.; Legendre, C.; Nochy, D. (2009). "Outcome of Subclinical Antibody-Mediated Rejection in Kidney Transplant Recipients with Preformed Donor-Specific Antibodies". American Journal of Transplantation. 9 (11). Elsevier BV: 2561–2570. doi:10.1111/j.1600-6143.2009.02813.x. ISSN 1600-6135. PMID 19775320.
- ^ Wiebe, C.; Gibson, I.W.; Blydt-Hansen, T.D.; Karpinski, M.; Ho, J.; Storsley, L.J.; Goldberg, A.; Birk, P.E.; Rush, D.N.; Nickerson, P.W. (2012). "Evolution and Clinical Pathologic Correlations of De Novo Donor-Specific HLA Antibody Post Kidney Transplant". American Journal of Transplantation. 12 (5). Elsevier BV: 1157–1167. doi:10.1111/j.1600-6143.2012.04013.x. ISSN 1600-6135. PMID 22429309.
- ^ AHERN, ANNE T.; ARTRUC, SCARLET B.; DELLAPELLE, PATRICIA; COSIMI, A. BENEDICT; RUSSELL, PAUL S.; COLVIN, ROBERT B.; FULLER, THOMAS C. (1982). "Hyperacute Rejection of Hla-Ab-Identical Renal Allografts Associated with B Lymphocyte and Endothelial Reactive Antibodies". Transplantation. 33 (1). Ovid Technologies (Wolters Kluwer Health): 103–106. doi:10.1097/00007890-198201000-00023. ISSN 0041-1337. PMID 7039014.
- ^ Opelz, Gerhard (2005). "Non-HLA transplantation immunity revealed by lymphocytotoxic antibodies". The Lancet. 365 (9470). Elsevier BV: 1570–1576. doi:10.1016/s0140-6736(05)66458-6. ISSN 0140-6736. PMID 15866311.
- ^ Akalin, Enver; Dinavahi, Rajani; Dikman, Steven; de Boccardo, Graciela; Friedlander, Rex; Schroppel, Bernd; Sehgal, Vinita; Bromberg, Jonathan S.; Heeger, Peter; Murphy, Barbara (2007). "Transplant Glomerulopathy May Occur in the Absence of Donor-Specific Antibody and C4d Staining". Clinical Journal of the American Society of Nephrology. 2 (6). Ovid Technologies (Wolters Kluwer Health): 1261–1267. doi:10.2215/cjn.02420607. ISSN 1555-9041. PMID 17942771.
- ^ Kohei, Naoki; Tanaka, Toshiaki; Tanabe, Kazunari; Masumori, Naoya; Dvorina, Nina; Valujskikh, Anna; Baldwin, William M.; Fairchild, Robert L. (2016). "Natural killer cells play a critical role in mediating inflammation and graft failure during antibody-mediated rejection of kidney allografts". Kidney International. 89 (6). Elsevier BV: 1293–1306. doi:10.1016/j.kint.2016.02.030. ISSN 0085-2538. PMC 4868788.
- ^ Racusen, Lorraine C.; Solez, Kim; Colvin, Robert B.; Bonsib, Stephen M.; Castro, Maria C.; Cavallo, Tito; Croker, Byron P.; Demetris, A. Jake; Drachenberg, Cynthia B.; Fogo, Agnes B.; Furness, Peter; Gaber, Lillian W.; Gibson, Ian W.; Glotz, Dennis; Goldberg, Julio C.; Grande, Joseph; Halloran, Philip F.; Hansen, H.E.; Hartley, Barry; Hayry, Pekka J.; Hill, Claire M.; Hoffman, Ernesto O.; Hunsicker, Lawrence G.; Lindblad, Anne S.; Marcussen, Niels; Mihatsch, Michael J.; Nadasdy, Tibor; Nickerson, Peter; Olsen, T. Steen; Papadimitriou, John C.; Randhawa, Parmjeet S.; Rayner, David C.; Roberts, Ian; Rose, Stephen; Rush, David; Salinas-Madrigal, Luis; Salomon, Daniel R.; Sund, Stale; Taskinen, Eero; Trpkov, Kiril; Yamaguchi, Yutaka (1999). "The Banff 97 working classification of renal allograft pathology". Kidney International. 55 (2). Elsevier BV: 713–723. doi:10.1046/j.1523-1755.1999.00299.x. ISSN 0085-2538.
- ^ Cosio, F.G.; Gloor, J.M.; Sethi, S.; Stegall, M.D. (2008). "Transplant Glomerulopathy". American Journal of Transplantation. 8 (3). Elsevier BV: 492–496. doi:10.1111/j.1600-6143.2007.02104.x. ISSN 1600-6135. PMID 18294145.
Further reading[edit]
- Patri, Pallavi; Seshan, Surya V.; Matignon, Marie; Desvaux, Dominique; Lee, John R.; Lee, Jun; Dadhania, Darshana M.; Serur, David; Grimbert, Philippe; Hartono, Choli; Muthukumar, Thangamani (2016). "Development and validation of a prognostic index for allograft outcome in kidney recipients with transplant glomerulopathy". Kidney International. 89 (2). Elsevier BV: 450–458. doi:10.1038/ki.2015.288. ISSN 0085-2538. PMC 4814368. PMID 26422505.
- Haas, Mark (2011). "Transplant glomerulopathy: it's not always about chronic rejection". Kidney International. 80 (8). Elsevier BV: 801–803. doi:10.1038/ki.2011.192. ISSN 0085-2538.
