Triiodothyronine
 | |
 | |
 | |
| Names | |
|---|---|
| IUPAC name
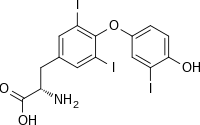
(2S)-2-amino-3-[4-(4-hydroxy-3-iodophenoxy)-3,5-diiodophenyl]propanoic acid
| |
| Other names
triiodothyronine
T3 3,3′,5-triiodo-L-thyronine | |
| Identifiers | |
3D model (JSmol)
|
|
| 2710227 | |
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| DrugBank | |
| ECHA InfoCard | 100.027.272 |
| EC Number |
|
| KEGG | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C15H12I3NO4 | |
| Molar mass | 650.977 g·mol−1 |
| Hazards | |
| GHS labelling: | |

| |
| Warning | |
| H315, H319, H335 | |
| P261, P264, P271, P280, P302+P352, P304+P340, P305+P351+P338, P312, P321, P332+P313, P337+P313, P362, P403+P233, P405, P501 | |
| NFPA 704 (fire diamond) | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Triiodothyronine, also known as T3, is a thyroid hormone. It affects almost every physiological process in the body, including growth and development, metabolism, body temperature, and heart rate.[1]
Production of T3 and its prohormone thyroxine (T4) is activated by thyroid-stimulating hormone (TSH), which is released from the anterior pituitary gland. This pathway is part of a closed-loop feedback process: Elevated concentrations of T3, and T4 in the blood plasma inhibit the production of TSH in the anterior pituitary gland. As concentrations of these hormones decrease, the anterior pituitary gland increases production of TSH, and by these processes, a feedback control system stabilizes the level of thyroid hormones in the bloodstream.
At the cellular level, T3 is the body's more active and potent thyroid hormone.[2] T3 helps deliver oxygen and energy to all of the body's cells, its effects on target tissues being roughly four times more potent than those of T4.[2] Of the thyroid hormone that is produced, just about 20% is T3, whereas 80% is produced as T4. Roughly 85% of the circulating T3 is later formed in the liver and anterior pituitary by removal of the iodine atom from the carbon atom number five of the outer ring of T4. In any case, the concentration of T3 in the human blood plasma is about one-fortieth that of T4. The half-life of T3 is about 2.5 days.[3] The half-life of T4 is about 6.5 days.[4] T3 levels start to rise 45 minutes after administration and peak at about 2.5 hours. Although manufacturer of Cytomel states half-life to be 2.5 days the half-life variability is great and can vary depending on the thyroid status of the patient. Newer studies have found the pharmakokinetics of T3 to be complex and the half-life to vary between 10 – 22 hours.[5]
Production[edit]
Synthesis from T4[edit]

T3 is the more metabolically active hormone produced from T4. T4 is deiodinated by three deiodinase enzymes to produce the more-active triiodothyronine:
- Type I present in liver, kidney, thyroid, and (to a lesser extent) pituitary; it accounts for 80% of the deiodination of T4.
- Type II present in CNS, pituitary, brown adipose tissue, and heart vessel, which is predominantly intracellular. In the pituitary, it mediates negative feedback on thyroid-stimulating hormone.
- Type III present in placenta, CNS, and hemangioma. This deiodinase converts T4 into reverse T3, which, unlike T3, is inactive.
T4 is synthesised in the thyroid follicular cells as follows.
- The sodium-iodide symporter transports two sodium ions across the basement membrane of the follicular cells along with an iodine ion. This is a secondary active transporter that utilises the concentration gradient of Na+ to move I− against its concentration gradient.
- I− is moved across the apical membrane into the colloid of the follicle.
- Thyroperoxidase oxidises I− to form the I radical.
- The thyroperoxidase iodinates the tyrosyl residues of the thyroglobulin within the colloid. The thyroglobulin was synthesised in the ER of the follicular cell and secreted into the colloid.
- Thyroid-stimulating hormone (TSH) released from the anterior pituitary gland binds the TSH receptor (a Gs protein-coupled receptor) on the basolateral membrane of the cell and stimulates the endocytosis of the colloid.
- The endocytosed vesicles fuse with the lysosomes of the follicular cell. The lysosomal enzymes cleave the T4 from the iodinated thyroglobulin.
- These vesicles are then exocytosed, releasing the thyroid hormones.

Direct synthesis[edit]
The thyroid gland also produces small amounts of T3 directly. In the follicular lumen, tyrosine residues become iodinated. This reaction requires hydrogen peroxide. Iodine bonds carbon 3 or carbon 5 of tyrosine residues of thyroglobulin in a process called organification of iodine. The iodination of specific tyrosines yields monoiodotyrosine (MIT) and diiodotyrosine (DIT). One MIT and one DIT are enzymatically coupled to form T3. The enzyme is thyroid peroxidase.
The small amount of T3 could be important because different tissues have different sensitivities to T4 due to differences in deiodinase ubiquitination in different tissues.[7] This once again raises the question if T3 should be included in thyroid hormone replacement therapy (THRT).
Mechanism of action[edit]
T3 and T4 bind to nuclear receptors (thyroid hormone receptors).[8] T3 and T4, although being lipophilic, are not able to passively diffuse through the phospholipid bilayers of target cells,[9] instead relying on transmembrane iodothyronine transporters. The lipophilicity of T3 and T4 requires their binding to the protein carrier thyroid-binding protein (TBG) (thyroxine-binding globulins, thyroxine binding prealbumins, and albumins) for transport in the blood. The thyroid receptors bind to response elements in gene promoters, thus enabling them to activate or inhibit transcription. The sensitivity of a tissue to T3 is modulated through the thyroid receptors.
Transportation[edit]

T3 and T4 are carried in the blood, bound to plasma proteins. This has the effect of increasing the half-life of the hormone and decreasing the rate at which it is taken up by peripheral tissues. There are three main proteins that the two hormones are bound to. Thyroxine-binding globulin (TBG) is a glycoprotein that has a higher affinity for T4 than for T3. Transthyretin is also a glycoprotein, but only carries T4, with hardly any affinity at all for T3. Finally, both hormones bind with a low affinity to serum albumin, but, due to the large availability of albumin, it has a high capacity.
The saturation of binding spots on thyronine-binding globulin (TBG) by endogenous T3 can be estimated by the triiodothyronine resin uptake test. The test is performed by taking a blood sample, to which an excess of radioactive exogenous T3 is added, followed by a resin that also binds T3. A fraction of the radioactive T3 binds to sites on TBG not already occupied by endogenous thyroid hormone, and the remainder binds to the resin. The amount of labeled hormones bound to the resin is then subtracted from the total that was added, with the remainder thus being the amount that was bound to the unoccupied binding sites on TBG.[11]
Effects[edit]
T3 increases the basal metabolic rate and, thus, increases the body's oxygen and energy consumption. The basal metabolic rate is the minimal caloric requirement needed to sustain life in a resting individual. T3 acts on the majority of tissues within the body, with a few exceptions including the spleen. It increases the synthesis and activity of the Na+/K+-ATPase (which normally constitutes a substantial fraction of total cellular ATP expenditure) without disrupting transmembrane ion balance.[12] In general, it increases the turnover of different endogenous macromolecules by increasing their synthesis and degradation.
Skeletal growth[edit]
Thyroid hormones are essential for normal growth and skeletal maturation.[13] They potentiate the effect of growth hormone and somatomedins to promote bone growth, epiphysial closure and bone maturation.[12][13]
Protein[edit]
T3 stimulates the production of RNA polymerase I and II and, therefore, increases the rate of protein synthesis. It also increases the rate of protein degradation, and, in excess, the rate of protein degradation exceeds the rate of protein synthesis. In such situations, the body may go into negative ion balance.[further explanation needed]
Lipids[edit]
T3 stimulates the breakdown of cholesterol and increases the number of LDL receptors, thereby increasing the rate of lipolysis.
Heart[edit]
T3 increases the heart rate and force of contraction, thus increasing cardiac output, by increasing β-adrenergic receptor levels in myocardium.[14] This results in increased systolic blood pressure and decreased diastolic blood pressure. The latter two effects act to produce the typical bounding pulse seen in hyperthyroidism. [citation needed] It also upregulates the thick filament protein myosin, which helps to increase contractility. A helpful clinical measure to assess contractility is the time between the QRS complex and the second heart sound. This is often decreased in hyperthyroidism.
Development[edit]
T3 has profound effect upon the developing embryo and infants. It affects the lungs and influences the postnatal growth of the central nervous system. It stimulates the production of myelin, the production of neurotransmitters, and the growth of axons. It is also important in the linear growth of bones.
Neurotransmitters[edit]
T3 may increase serotonin in the brain, in particular in the cerebral cortex, and down-regulate 5HT-2 receptors, based on studies in which T3 reversed learned helplessness in rats and physiological studies of the rat brain.[15]
Physiological function[edit]
Thyroid hormones act to increase protein turnover. This might serve an adaptive function in regard to long-term calorie restriction with adequate protein.[16][17] When calories are in short supply, reduction in protein turnover may ameliorate the effects of the shortage.
Measurement[edit]
Triiodothyronine can be measured as free triiodothyronine, which is an indicator of triiodothyronine activity in the body. It can also be measured as total triiodothyronine, which also depends on the triiodothyronine that is bound to thyroxine-binding globulin.[18]
Uses[edit]
Treatment of depressive disorders[edit]
The addition of triiodothyronine to existing treatments such as SSRIs is one of the most widely studied augmentation strategies for refractory depression,[19] however success may depend on the dosage of T3. A long-term case series study by Kelly and Lieberman of 17 patients with major refractory unipolar depression found that 14 patients showed sustained improvement of symptoms over an average timespan of two years, in some cases with higher doses of T3 than the traditional 50 µg required to achieve therapeutic effect, with an average of 80 µg and a dosage span of 24 months; dose range: 25–150 µg.[19] The same authors published a retrospective study of 125 patients with the two most common categories of bipolar disorders II and NOS whose treatment had previously been resistant to an average of 14 other medications. They found that 84% experienced improvement and 33% experienced full remission over a period of an average of 20.3 months (standard deviation of 9.7). None of the patients experienced hypomania while on T3.[20]
Use as a fat loss supplement[edit]
3,5-Diiodo-L-thyronine and 3,3′-diiodo-L-thyronine are used as ingredients in certain over-the-counter fat-loss supplements, designed for bodybuilding. Several studies have shown that these compounds increase the metabolization of fatty acids and the burning of adipose fat tissue in rats.[21][22]
Alternative medicine[edit]
Triiodothyronine has been used to treat Wilson's syndrome, an alternative medical diagnosis not recognized as a medical condition by mainstream medicine. This diagnosis involves various non-specific symptoms that are attributed to the thyroid, despite normal thyroid function tests. The American Thyroid Association has raised concern that the prescribed treatment with triiodothyronine is potentially harmful.[23]
History[edit]
In 1950 Dr Jack Gross, a Canadian endocrinologist, came to the British National Institute for Medical Research to work with Rosalind Pitt-Rivers as a postdoctoral fellow. Gross had previous experience working at McGill University under Professor Charles Leblond, where they used radioactive iodine to study the physiology of thyroid hormone and applied chromatography to analyze radioiodinated proteins in human blood after radioiodine therapy. Gross and Leblond found an unknown radioactive compound in the blood of rats given radioactive iodine. The compound migrated close to thyroxine in chromatography and they initially named it 'unknown 1' . Around that time a group led by Jean Roche in Paris described a deiodinating activity in the sheep thyroid gland, raising the possibility that 'unknown 1' is the less iodinated analogue of T4, triiodothyronine.[24] In march of 1952 Gross & Pitt-Rivers published a paper in The Lancet titled "The identification of 3: 5: 3'-L-triiodothyronine in human plasma".[25]
While Gross & Pitt-Rivers are normally credited with discovering T3, this compound was actually first isolated by the biochemists Hird & Trikojus at the University of Melbourne in 1948.[26] It has been suggested that their published paper was little-known and therefore easily ignored.[27] It has also been stated that Pitt-Rivers had read this paper but failed to mention it.[28]
See also[edit]
References[edit]
- ^ Bowen R (2010-07-24). "Physiologic Effects of Thyroid Hormones". Colorado State University. Retrieved 2013-09-29.
- ^ a b "How Your Thyroid Works – "A delicate Feedback Mechanism"". endocrineweb. 2012-01-30. Retrieved 2013-09-29.
- ^ "Cytomel (Liothyronine Sodium) Drug Information". RxList. 2011-01-03. Retrieved 2013-09-29.
- ^ Irizarry L (23 April 2014). "Thyroid Hormone Toxicity". Medscape. WedMD LLC. Retrieved 2 May 2014.
- ^ Jonklaas J, Burman KD, Wang H, Latham KR (February 2015). "Single Dose T3 Administration: Kinetics and Effects on Biochemical and Physiologic Parameters". Therapeutic Drug Monitoring. 37 (1): 110–118. doi:10.1097/FTD.0000000000000113. PMC 5167556. PMID 24977379.
- ^ Boron WF (2005). Medical Physiology: A Cellular And Molecular Approach. Philadelphia, PA: Elsevier / Saunders. p. 1300. ISBN 1-4160-2328-3. LCCN 2004051158.
- ^ Werneck de Castro JP, Fonseca TL, Ueta CB, McAninch EA, Abdalla S, Wittmann G, et al. (February 2015). "Differences in hypothalamic type 2 deiodinase ubiquitination explain localized sensitivity to thyroxine". The Journal of Clinical Investigation. 125 (2): 769–781. doi:10.1172/JCI77588. PMC 4319436. PMID 25555216.
- ^ Lazar MA, Chin WW (December 1990). "Nuclear thyroid hormone receptors". The Journal of Clinical Investigation. 86 (6): 1777–1782. doi:10.1172/JCI114906. PMC 329808. PMID 2254444.
- ^ Dietrich JW, Brisseau K, Boehm BO (August 2008). "[Absorption, transport and bio-availability of iodothyronines]" [Absorption, transport and bio-availability of iodothyronines]. Deutsche Medizinische Wochenschrift (in German). 133 (31–32): 1644–1648. doi:10.1055/s-0028-1082780. PMID 18651367.
- ^ References used in image are found in image article in Commons:Commons:File:Thyroid_system.png#References.
- ^ triiodothyronine resin uptake test from Farlex Medical Dictionary, citing: Mosby's Medical Dictionary, 8th edition. 2009, Elsevier.
- ^ a b Costanzo LS (2018). Physiology (6th ed.). Philadelphia, PA: Elsevier. p. 425. ISBN 978-0-323-47881-6. OCLC 966608835.
- ^ a b Barrett KE, Barman SM, Brooks HL, Yuan JX, Ganong WF (2019). Ganong's review of medical physiology (26th ed.). New York: McGraw-Hill Education. pp. 364–365. ISBN 978-1-260-12240-4. OCLC 1076268769.
- ^ "Thyroid physiology and tests of function". Anaesthetist.com.
- ^ Martin P, Brochet D, Soubrie P, Simon P (September 1985). "Triiodothyronine-induced reversal of learned helplessness in rats". Biological Psychiatry. 20 (9): 1023–1025. doi:10.1016/0006-3223(85)90202-1. PMID 2992618. S2CID 43784239.
- ^ Fontana L, Klein S, Holloszy JO, Premachandra BN (August 2006). "Effect of long-term calorie restriction with adequate protein and micronutrients on thyroid hormones". The Journal of Clinical Endocrinology and Metabolism. 91 (8): 3232–3235. doi:10.1210/jc.2006-0328. PMID 16720655.
- ^ Roth GS, Handy AM, Mattison JA, Tilmont EM, Ingram DK, Lane MA (July 2002). "Effects of dietary caloric restriction and aging on thyroid hormones of rhesus monkeys". Hormone and Metabolic Research = Hormon- und Stoffwechselforschung = Hormones et Metabolisme. 34 (7): 378–382. doi:10.1055/s-2002-33469. PMID 12189585. S2CID 20574006.
- ^ Military Obstetrics & Gynecology – Thyroid Function Tests In turn citing: Operational Medicine 2001, Health Care in Military Settings, NAVMED P-5139, May 1, 2001, Bureau of Medicine and Surgery, Department of the Navy, 2300 E Street NW, Washington, D.C., 20372-5300
- ^ a b Kelly TF, Lieberman DZ (May 2009). "Long term augmentation with T3 in refractory major depression". Journal of Affective Disorders. 115 (1–2): 230–233. doi:10.1016/j.jad.2008.09.022. PMID 19108898.
- ^ Kelly T, Lieberman DZ (August 2009). "The use of triiodothyronine as an augmentation agent in treatment-resistant bipolar II and bipolar disorder NOS". Journal of Affective Disorders. 116 (3): 222–226. doi:10.1016/j.jad.2008.12.010. PMID 19215985.
- ^ Lombardi A, Lanni A, Moreno M, Brand MD, Goglia F (February 1998). "Effect of 3,5-di-iodo-L-thyronine on the mitochondrial energy-transduction apparatus". The Biochemical Journal. 330 (Pt 1): 521–526. doi:10.1042/bj3300521. PMC 1219168. PMID 9461551.
- ^ Lanni A, Moreno M, Lombardi A, de Lange P, Silvestri E, Ragni M, et al. (September 2005). "3,5-diiodo-L-thyronine powerfully reduces adiposity in rats by increasing the burning of fats". FASEB Journal. 19 (11): 1552–1554. doi:10.1096/fj.05-3977fje. PMID 16014396. S2CID 1624615.
- ^ "ATA Statement on "Wilson's Syndrome"". American Thyroid Association. 24 May 2005.
- ^ Smith TS (2018-08-05). "HISTORY: Rosalind Pitt-Rivers, the co-discoverer of T3 hormone". Thyroid Patients Canada. Retrieved 2022-09-13.
- ^ Gross J, Pitt-Rivers R (March 1952). "The identification of 3:5:3'-L-triiodothyronine in human plasma". Lancet. 1 (6705): 439–441. doi:10.1016/s0140-6736(52)91952-1. PMID 14898765.
- ^ Hird F, Trikojus VM (June 1948). "Paper partition chromatography with thyroxine and analogues". The Australian Journal of Science. 10 (6): 185–187. ISSN 0365-3668. PMID 18875255.
- ^ Hulbert AJ (2001). The comparative physiology of vertebrate metabolism :studies of its evolution, control and development (Thesis thesis). UNSW Sydney. doi:10.26190/unsworks/14477. hdl:1959.4/69845.
- ^ Humphreys LR. "Trikojus, Victor Martin (Trik) (1902–1985)". Australian Dictionary of Biography. National Centre of Biography, Australian National University. ISSN 1833-7538. Retrieved 2023-07-27.
External links[edit]
- Triiodothyronine bound to proteins in the PDB
- T3 at Lab Tests Online

