Trofinetide
 | |
| Clinical data | |
|---|---|
| Trade names | Daybue |
| Other names | NNZ-2566 |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a623019 |
| License data |
|
| Routes of administration | By mouth |
| ATC code |
|
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 84% |
| Metabolism | Insignificant |
| Elimination half-life | ~ 1.5 h |
| Excretion | Urine |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| Chemical and physical data | |
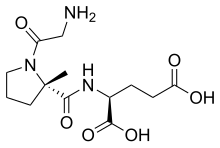
| Formula | C13H21N3O6 |
| Molar mass | 315.326 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Trofinetide, sold under the brand name Daybue, is a medication used for the treatment of Rett syndrome.[1] It is taken by mouth.[1]
The most common adverse reactions include diarrhea and vomiting.[2]
Trofinetide was approved for medical use in the United States in March 2023.[1][2][3] The US Food and Drug Administration (FDA) considers it to be a first-in-class medication.[4]
Medical uses[edit]
Trofinetide is indicated for the treatment of Rett syndrome in people two years of age and older.[1][5]
Rett syndrome is a rare, genetic neurological and developmental disorder that affects the way the brain develops.[2] People with Rett syndrome experience a progressive loss of motor skills and language.[2] Most babies with Rett syndrome seem to develop as expected for the first six months of life.[2] These babies then lose skills they previously had attained at approximately six to 18 months of age — such as the ability to crawl, walk, communicate, or use their hands.[2] The hallmark of Rett syndrome is near constant repetitive hand movements, such as rubbing or clapping.[2] Rett syndrome leads to severe impairments affecting nearly every aspect of life, including the ability to speak, walk, eat, and breathe.[2]
History[edit]
It was developed by Neuren Pharmaceuticals that acts as an analogue of the neuropeptide (1-3) IGF-1, which is a simple tripeptide with sequence Gly-Pro-Glu obtained by enzymatic cleavage of the growth factor IGF-1 within the brain. Trofinetide has anti-inflammatory properties and was originally developed as a potential treatment for stroke,[6][7] but has subsequently been developed for other applications and is now approved by the FDA as an oral solution. It has successfully completed Phase III clinical trial against Rett syndrome.[8] Trofinetide has also had a successful Phase II trial against Fragile X syndrome.[9][10][11] The drug is manufactured by Acadia Pharmaceuticals.[citation needed]
The US Food and Drug Administration (FDA) evaluated the efficacy and safety of trofinetide based on a randomized, double-blind, placebo-controlled, 12-week study (Study 1; NCT04181723) of participants with Rett syndrome five to 20 years of age.[2] Participants were randomized to receive trofinetide (N=93) or matching placebo (N=94) for 12 weeks.[2] The dose of trofinetide was based on participant weight to achieve similar exposure in all participants.[2]
The FDA granted the application for trofinetide priority review, orphan drug, and fast track designations.[2]
References[edit]
- ^ a b c d e "Daybue- trofinetide solution". DailyMed. 29 March 2023. Archived from the original on 2 July 2023. Retrieved 20 November 2023.
- ^ a b c d e f g h i j k l "FDA approves first treatment for Rett Syndrome". U.S. Food and Drug Administration (FDA). 13 March 2023. Archived from the original on 13 March 2023. Retrieved 13 March 2023.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.
- ^ Keam SJ (June 2023). "Trofinetide: First Approval". Drugs. 83 (9): 819–824. doi:10.1007/s40265-023-01883-8. PMID 37191913. S2CID 258715933.
- ^ New Drug Therapy Approvals 2023 (PDF). U.S. Food and Drug Administration (FDA) (Report). January 2024. Archived from the original on 10 January 2024. Retrieved 9 January 2024.
- ^ "Drug Approval Package: Daybue". U.S. Food and Drug Administration (FDA). 6 April 2023. Archived from the original on 20 November 2023. Retrieved 20 November 2023.
- ^ Bickerdike MJ, Thomas GB, Batchelor DC, Sirimanne ES, Leong W, Lin H, et al. (March 2009). "NNZ-2566: a Gly-Pro-Glu analogue with neuroprotective efficacy in a rat model of acute focal stroke". Journal of the Neurological Sciences. 278 (1–2): 85–90. doi:10.1016/j.jns.2008.12.003. PMID 19157421. S2CID 7789415.
- ^ Cartagena CM, Phillips KL, Williams GL, Konopko M, Tortella FC, Dave JR, et al. (September 2013). "Mechanism of action for NNZ-2566 anti-inflammatory effects following PBBI involves upregulation of immunomodulator ATF3". Neuromolecular Medicine. 15 (3): 504–14. doi:10.1007/s12017-013-8236-z. PMID 23765588. S2CID 12522580. Archived from the original on 15 April 2023. Retrieved 10 December 2019.
- ^ "Positive top-line results from pivotal Phase 3 trial in Rett syndrome" (PDF). Rettsyndrome.org. 7 December 2021. Archived (PDF) from the original on 18 August 2022. Retrieved 21 July 2022.
- ^ Deacon RM, Glass L, Snape M, Hurley MJ, Altimiras FJ, Biekofsky RR, et al. (March 2015). "NNZ-2566, a novel analog of (1-3) IGF-1, as a potential therapeutic agent for fragile X syndrome". Neuromolecular Medicine. 17 (1): 71–82. doi:10.1007/s12017-015-8341-2. PMID 25613838. S2CID 11964380.
- ^ "Study Details - Rett Syndrome Study". Rettstudy.com. Archived from the original on 4 October 2016. Retrieved 21 July 2022.
- ^ "Neuren's Tofinetide Successful in Phase 2 Clinical Trial in Fragile X". Fraxa.org. 7 December 2015. Archived from the original on 24 June 2022. Retrieved 21 July 2022.
External links[edit]
- Clinical trial number NCT04181723 for "Study of Trofinetide for the Treatment of Girls and Women With Rett Syndrome (LAVENDER)" at ClinicalTrials.gov
