User:Support.and.Defend/chain rule
| Part of a series of articles about |
| Calculus |
|---|
In calculus, the chain rule is a method to compute the derivative of the composition of two or more functions.
If a variable, y, depends on a second variable, u, which in turn depends on a third variable, x, that is y = y(u(x)) , then the rate of change of y with respect to x can be computed as the rate of change of y with respect to u multiplied by the rate of change of u with respect to x. Schematically,
The method is called the "chain rule" because it can be applied sequentially to as many functions as are nested inside one another. For example, if f is a function of g which is in turn a function of h, which is in turn a function of x, that is
,
the derivative of f with respect to x is given by
and so on.
The chain rule has broad applications in Physics, Chemistry, and Engineering, as well as being used to study related rates in many disciplines. For example, if a skydiver is falling at 60 miles per hour and the barometric pressure increases at 3 psi per mile fallen, then the skydiver experiences an increasing pressure at a rate of 60 mi/hr * 3 psi/mi = 180 psi per hour. The chain rule can also be generalized to multiple variables in cases where the nested functions depend on more than one variable.
Examples[edit]
Example I[edit]
Suppose that a mountain climber ascends at a rate of 0.5 kilometers per hour. The temperature is lower at higher elevations; suppose the rate by which it decreases is 6 °C per kilometer. To calculate the decrease in air temperature per hour that the climber experiences, one multiplies 6 °C per kilometer by 0.5 kilometer per hour, to obtain 3 °C per hour. This calculation is a typical chain rule application.
Example II[edit]
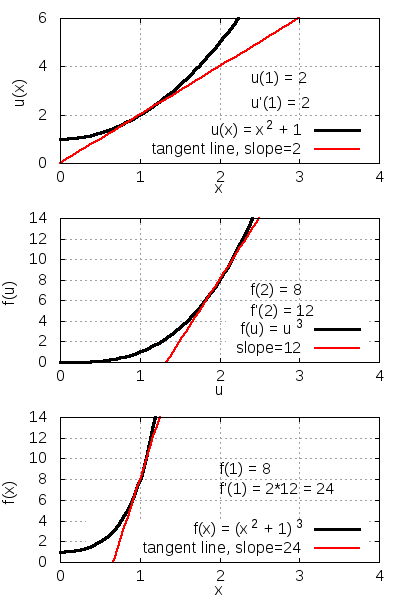
Consider the function f(x) = (x2 + 1)3. It follows from the chain rule that
| Function to differentiate | |
| Define u(x) as inside function | |
| Express f(x) in terms of u(x) | |
| Express chain rule applicable here | |
| Substitute in f(u) and u(x) | |
| Compute derivatives with power rule | |
| Substitute u(x) back in terms of x | |
| Simplify. |
Example III[edit]
In order to differentiate the trigonometric function
one can write:
| Function to differentiate | |
| Define u(x) as inside function | |
| Express f(x) in terms of u(x) | |
| Express chain rule applicable here | |
| Substitute in f(u) and u(x) | |
| Evaluate derivatives | |
| Substitute u in terms of x. |
Example IV Three Nested Functions[edit]
The method is called the "chain rule" because it can be applied sequentially to as many functions as are nested inside one another. For example, if , sequential application of the chain rule yields the derivative as follows:
| Original (outermost) function | |
| Define h(x) as innermost function | |
| g(h) = sin(h) as middle function | |
| Express chain rule applicable here | |
| Differentiate f(g) | |
| Differentiate g(h) | |
| Differentiate h(x) | |
| Substitute into chain rule. |
Chain rule in Physics[edit]
Because one physical quantity often depends on another, which, in turn depends on others, the chain rule has broad applications in physics. This section presents examples of the chain rule in kinematics and simple harmonic motion, the chain rule is also useful in electromagnetic induction.
Physics Example I: Relative Kinematics of Two Vehicles[edit]

For example, one can consider the kinematics problem where one vehicle is heading West toward an intersection at 80 miles per hour while another is heading North away from the intersection at 60 miles per hour. One can ask whether the vehicles are getting closer or further apart and at what rate at the moment when the North bound vehicle is 3 miles North of the intersection and the West bound vehicle is 4 miles East of the intersection.
Big idea: use chain rule to compute rate of change of distance between two vehicles.
Plan:
- Choose coordinate system
- Identify variables
- Draw picture
- Big idea: use chain rule to compute rate of change of distance between two vehicles
- Express c in terms of x and y via Pythagorean theorem
- Express dc/dt using chain rule in terms of dx/dt and dy/dt
- Substitute in x, y, dx/dt, dy/dt
- Simplify.
Choose coordinate system: Let the y-axis point North and the x-axis point East.
Identify variables: Define y(t) to be the distance of the vehicle heading North from the origin and x(t) to be the distance of the vehicle heading West from the origin.
Express c in terms of x and y via Pythagorean theorem:
Express dc/dt using chain rule in terms of dx/dt and dy/dt:
| Apply derivative operator to entire function | |
| Sum of squares is inside function | |
| Distribute differentiation operator | |
| Apply chain rule to x(t) and y(t)} | |
| Simplify. |
Substitute in x = 4 mi, y = 3 mi, dx/dt = -80 mi/hr, dy/dt = 60 mi/hr and Simplify
Consequently, the two vehicles are getting closer together at a rate of 28 mi/hr.
Physics Example II: Harmonic Oscillator[edit]
If the displacement of a simple harmonic oscillator from equilibrium is given by x, and it is released from its maximum displacement A at time t = 0, then the position at later times is given by
where ω = 2 π/T is the angular frequency and T is the period of oscillation. The velocity, v, being the first time derivative of the position can be computed with the chain rule:
| Definition of velocity in one dimension | |
| Substitute x(t) | |
| Bring constant A outside of derivative | |
| Differentiate outside function (cosine) | |
| Bring negative sign in front | |
| Evaluate remaining derivative | |
| Simplify. |
The acceleration is then the second time derivative of position, or simply dv/dt.
| Definition of acceleration in one dimension | |
| Substitute v(t) | |
| Bring constant term outside of derivative | |
| Differentiate outside function (sine) | |
| Evaluate remaining derivative | |
| Simplify. |
From Newton's second law, F = ma, where F is the net force and m is the object's mass.
| Newton's second law | |
| Substitute a(t) | |
| Simplify | |
| Substitute original x(t). |
Thus it can be seen that these results are consistent with the observation that the force on a simple harmonic oscillator is a negative constant times the displacement.
Chain rule in Chemistry[edit]
The chain rule has many applications in Chemistry because many equations in Chemistry describe how one physical quantity depends on another, which in turn depends on another. For example, the ideal gas law describes the relationship between pressure, volume, temperature, and number of moles, all of which can also depend on time.
Chemistry Example I: Ideal Gas Law[edit]

Suppose a sample of n moles of an ideal gas is held in an isothermal (constant temperature, T) chamber with initial volume V0. The ideal gas is compressed by a piston so that its volume changes at a constant rate so that V(t) = V0 - kt, where t is the time. The chain rule can be employed to find the time rate of change of the pressure.[1] The ideal gas law can be solved for the pressure, P to give:
where P(t) and V(t) have been written as explicit functions of time and the other symbols are constant. Differentiating both sides yields
where the constant terms, n, R, and T, have been moved to the left of the derivative operator. Applying the chain rule gives
where the power rule has been used to differentiate 1/V, Since V(t) = V0 - kt, dV/dt = -k. Substituting in for V and dV/dt yields dP/dt.
To find P(t), one would integrate dP/dt.
Chemistry Example II: Kinetic Theory of Gases[edit]

A second application of the chain rule in Chemistry is finding the rate of change of the average molecular speed, v, in an ideal gas as the absolute temperature T, increases at a constant rate so that T = T0 + at, where T0 is the initial temperature and t is the time.[2] The kinetic theory of gases relates the root mean square of the molecular speed to the temperature, so that if v(t) and T(t) are functions of time,
where R is the ideal gas constant, and M is the molecular weight.
Differentiating both sides with respect to time yields:
Using the chain rule to express the right side in terms of the with respect to temperature, T, and time, t, respectively gives
Evaluating the derivative with respect to temperature, T, yields
Evaluating the remaining derivative with respect to T, taking the reciprocal of the negative power, and substituting T = T0 + at, produces
Evaluating the derivative with respect to t yields
which simplifies to
This can be integrated to find v(t), the rms molecular speed as a function of time.
Chain rule in Engineering[edit]
Coming soon
See also[edit]
- Inverse chain rule
- Quotient Rule
- Triple product rule
- Leibniz integral rule
- Leibniz rule (generalized product rule)
- Itō's Lemma
References[edit]
- ^ University of British Columbia, UBC Calculus Online Course Notes, Applications of the Chain Rule, http://www.ugrad.math.ubc.ca/coursedoc/math100/notes/derivative/chainap.html Accessed 11/15/2010.
- ^ University of British Columbia, UBC Calculus Online Course Notes, Applications of the Chain Rule, http://www.ugrad.math.ubc.ca/coursedoc/math100/notes/derivative/chainap.html Accessed 11/15/2010.
External links[edit]
Category:Differentiation rules Category:Articles containing proofs







![{\displaystyle f(x)=[u(x)]^{3}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/8adafb762d4c0348840f71fc6903cbad7147ca0d)























![{\displaystyle ={\frac {1}{2}}(x^{2}+y^{2})^{-1/2}\left[{\frac {d}{dt}}(x^{2})+{\frac {d}{dt}}(y^{2})\right]}](https://wikimedia.org/api/rest_v1/media/math/render/svg/750fe398b8f2335a387fec30fecf6af33a89030c)
![{\displaystyle ={\frac {1}{2}}(x^{2}+y^{2})^{-1/2}\left[2x{\frac {dx}{dt}}+2y{\frac {dy}{dt}}\right]}](https://wikimedia.org/api/rest_v1/media/math/render/svg/25a26e9a0048df56b0d150bf839592a6e4acb697)






























