User:Tyronnep/sandbox
Warning This page contains syntax errors ("cite%20note") caused by a VisualEditor bug. Do not copy/move content from this page until the errors have been repaired. See {{Warning VisualEditor bug}} for more information. |
Alfred Werner | |
|---|---|
 | |
| Born | 12 December 1866 |
| Died | 15 November 1919 (aged 52) |
| Nationality | Swiss |
| Alma mater | University of Zurich ETH Zurich |
| Known for | configuration of transition metal complexes |
| Awards | Nobel Prize for Chemistry (1913) |
| Scientific career | |
| Fields | Inorganic chemistry |
| Institutions | University of Zurich |
| Doctoral advisor | Arthur Rudolf Hantzsch, Marcellin Berthelot[citation needed] |
Alfred Werner (12 December 1866 – 15 November 1919) was a Swiss chemist who was a student at ETH Zurich and a professor at the University of Zurich. He won the Nobel Prize in Chemistry in 1913 for proposing the octahedral configuration of transition metal complexes.[1] He was the first inorganic chemist to win the Nobel prize, and the only one prior to 1973.[1] Werner has been regarded as the founding father of modern inorganic stereochemistry and the founder and father of coordination theory.[2][3]
Biography[edit]
Werner was born in 1866 in Mulhouse, Alsace (which was then part of France, but which was annexed by Germany in 1871).[1] His father, J.A. Werner, a foundry worker and locksmith, and his mother was named, Salomé Jeanette Tesché and she came from a wealth family.[4] He was raised as Roman Catholic.[2] While in school he showed interest in chemistry and at the age of 18 he conduct his own independent chemical research.[5] For a year he did military service in Karlsruhe and during this time he also attended lectures of Engler at the Technical High School at Zürich.[1]
He obtained a Diploma in Technical Chemistry in 1889 and started to become influenced by Professor Arthur Rudolf Hantzsch.[1][5] He went to Switzerland to study chemistry at the Swiss Federal Institute (Polytechnikum) in Zurich where he obtained his doctorate in 1890, at the same institution he presented his thesis on the spatial arrangements of the atoms in molecules containing nitrogen.[6]His studies later on brought him to Paris, where he worked under Professor Berthelot at the College de France, he returned to the Swiss Federal Institute to teach (1892), in 1893 he moved to the University of Zurich where he became a professor a year later at 29 years old, which is the same year he became a Swiss citizen.[7] He married Emma Giesker of Zurich, with whom he had a son, Alfred, and a daughter, Charlotte.[1]
While a professor at the University of Zurich he gave lectures on Organic chemistry, and later on in 1902, Inorganic chemistry.[2] After postdoctoral study Werner became associated with the theory of coordination, published a paper on the theory of affinity and valence, and stated a theory of variable valence.[3] For the next 20 years after becoming a professor, Werner and his collaborators studied molecular compounds and their configurations.[1] He also published 150 papers by himself, which eventually led to the discovery of optically-active isomers.[1] In 1913, he received a Noble Prize for Chemistry for his work "in recognition of his work on the linkage of atoms in molecules by which he has thrown new light on earlier investigations and opened up new fields of research especially in inorganic chemistry."[8] During this time Werner was suffering from arteriosclerosis. By 1915 he was compelled to give up his general lectures on chemistry and in 1919 he had to give up his professorship.[6]
Research[edit]
Coordination chemistry[edit]

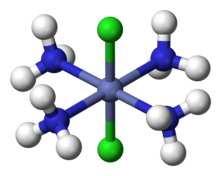
In 1893, Werner was the first to propose correct structures for coordination compounds containing complex ions, in which a central transition metal atom is surrounded by neutral or anionic ligands. Although he was not the first to work on coordination complexes. Werner theory was sparked from Sophus Mads Jorgensen's theory of coordination complexes. His work was originally derived from Christian Wilhelm Blomstrand. Blomstrand developed what has be known as the complex ion chain theory.[9] It was two important changes to Jorgensen's theory that mad it most widely accepted and still used today.
For example, it was known that cobalt forms a "complex" hexamminecobalt(III) chloride, with formula CoCl3•6NH3, but the nature of the association indicated by the dot was mysterious. Werner proposed the structure [Co(NH3)6]Cl3, with the Co3+ ion surrounded by six NH3 at the vertices of an octahedron. The three Cl− are dissociated as free ions, which Werner confirmed by measuring the conductivity of the compound in aqueous solution, and also by chloride anion analysis using precipitation with silver nitrate. Later, magnetic susceptibility analysis was also used to confirm Werner's proposal for the chemical nature of CoCl3•6NH3.
For complexes with more than one type of ligand, Werner succeeded in explaining the number of isomers observed. For example, he explained the existence of two tetrammine isomers, "Co(NH3)4Cl3", one green and one purple. Werner proposed that these are two geometric isomers of formula [Co(NH3)4Cl2]Cl, with one Cl− ion dissociated as confirmed by conductivity measurements. The Co atom is surrounded by four NH3 and two Cl ligands at the vertices of an octahedron. The green isomer is "trans" with the two Cl ligands at opposite vertices, and the purple is "cis" with the two Cl at adjacent vertices.
Werner also prepared complexes with optical isomers, and in 1914 he reported the first synthetic chiral compound lacking carbon, known as hexol with formula [Co(Co(NH3)4(OH)2)3]Br6. Copied from Alfred Werner.This theory allows one to understand the difference between a coordinated ligand and a charge balancing ion in a compound, for example the chloride ion in the cobaltammine chlorides and to explain many of the previously inexplicable isomers.[9]
His work on coordination chemistry has held create significance in other discoveries and an impact on today's society as while. Gilbert N. Lewis published a monograph Valance in 192, he emphasized the historical credit due to Alfred Werner. In the monograph it showed that Irving Langmuir's octet did not show importance in many compounds containing elements from the higher rows of the periodic table. From the coordination theory Werner and his students were able to identify dozens of previously unknown compounds and synthesize dozens more.[2] Today coordination chemistry is applied to bioinorganic chemistry, bioorganometallic chemistry, homogeneous catalysis, identification of the presence of metals in a sample (but replaced by atomic absorption spectroscopy and inductively coupled plasma mass spectrometry), electronic theory of valency, and virtual enterprise.
Nature of valence[edit]
Before Werner, chemists defined the valence of an element as the number of its bonds without distinguishing different types of bond. It was Werner's data on complexes of ammonia with PtCl4 that led him to postulate that metal ions have two different kinds of valence, primary and secondary. However, in complexes such as [Co(NH3)6]Cl3 for example, Werner considered that the Co-Cl bonds correspond to a "primary" valence of 3 at long distance, while the Co-NH3 bonds which correspond to a "secondary" or weaker valence of 6 at shorter distance. This secondary valence of 6 he referred to as the coordination number which he defined as the number of molecules (here of NH3) directly linked to the central metal atom. In other complexes he found coordination numbers of 4 or 8. Copied from Alfred Werner.
On these views, and other similar views, in 1904 Richard Abegg formulated what is now known as Abegg's rule which states that the difference between the maximum positive and negative valence of an element is frequently eight. This rule was used later in 1916 when Gilbert N. Lewis formulated the “octet rule” in his cubical atom theory.
In modern terminology Werner's primary valence corresponds to the oxidation state, and his secondary valence is called coordination number. Coordination number is the total number of neighbors of a central atom in a molecule or ion, as defined by Werner.[10][11] The Co-Cl bonds (in the above example) are now classed as ionic, and each Co-N bond is a coordinate covalent bond between the Lewis acid Co3+ and the Lewis base NH3.Copied from Alfred Werner.
Works[edit]
- Lehrbuch der Stereochemie . Fischer, Jena 1904 Digital edition by the University and State Library Düsseldorf
- Neuere anschauungen auf dem gebiete der anoroganischen chemie {New ideas on inorganic chemistry} . Werner, Alfred, Hedley, Edgar 1911 Digital edition by University System of MD & partners
- Beitragezur Theorie der Affinitat und Valenz {Contributions to the Theory of Affinity and Valence}. Werner, Alfred 1891.
- Lehrbuch der Stereochemie’ [Textbook of Stereochemistry]. Werner, Alfred 1904 Digital edition by the University System of MD & partners
References[edit]
- ^ a b c d e f g h "Alfred Werner Biography". Nobel Prize. Retrieved 19 July 2019.
- ^ a b c "Alfred Werner". www.nndb.com. Retrieved 2019-06-22.
- ^ a b Worldofchemicals.com, Data Research Analyst (2012-12-11). "Alfred Werner - Founder of coordination chemistry". worldofchemicals.com. Retrieved 2019-06-22.
- ^ "Biography of Alfred Werner". www.chemistryexplained.com. Retrieved 2019-06-29.
- ^ a b "120 Years of Top Inorganic Chemistry — Alfred Werner :: News :: ChemistryViews". www.chemistryviews.org. Retrieved 2019-06-29.
- ^ a b "The Nobel Prize in Chemistry 1913". NobelPrize.org. Retrieved 2019-06-22.
- ^ "Alfred Werner | Swiss chemist". Encyclopedia Britannica. Retrieved 2019-06-29.
- ^ "The Nobel Prize in Chemistry 1913". NobelPrize.org. Retrieved 2019-06-22.
- ^ a b "Coordination compound - Characteristics of coordination compounds". Encyclopedia Britannica. Retrieved 2019-06-29.
- ^ IUPAC, Compendium of Chemical Terminology, 2nd ed. (the "Gold Book") (1997). Online corrected version: (2006–) "coordination number". doi:10.1351/goldbook.C01331
- ^ De, A.K. (2003). A Text Book of Inorganic Chemistry. New Age International Publishers. p. 88. ISBN 978-8122413847.
External links[edit]
- Biography at Nobelprize.org
- The Nobel Prize in Chemistry 1913 - short article about his work on the linkage of atoms in molecules by which he has thrown new light on earlier investigations and opened up new fields of research especially in inorganic chemistry.
{{Authority control}} {{DEFAULTSORT:Werner, Alfred}} [[Category:1866 births]] [[Category:1919 deaths]] [[Category:People from Mulhouse]] [[Category:Inorganic chemists]] [[Category:Nobel laureates in Chemistry]] [[Category:Swiss Nobel laureates]] [[Category:ETH Zurich alumni]] [[Category:University of Zurich faculty]] [[Category:Swiss chemists]] [[Category:People involved with the periodic table]]

