Willgerodt rearrangement
| Willgerodt rearrangement | |
|---|---|
| Named after | Conrad Willgerodt |
| Reaction type | Rearrangement reaction |
| Identifiers | |
| RSC ontology ID | RXNO:0000185 |
The Willgerodt rearrangement or Willgerodt reaction is an organic reaction converting an aryl alkyl ketone, alkyne, or alkene to the corresponding amide by reaction with ammonium polysulfide, named after Conrad Willgerodt.[1][2][3][4] The formation of the corresponding carboxylic acid is a side reaction resulting from hydrolysis of the amide. When the alkyl group is an aliphatic chain (n typically 0 to 5), multiple reactions take place with the amide group always ending up at the terminal end. The net effect is thus migration of the carbonyl group to the end of the chain and oxidation.
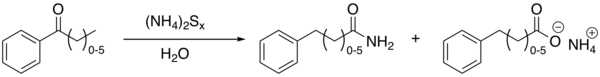
An example with modified reagents (sulfur, concentrated ammonium hydroxide and pyridine) is the conversion of acetophenone to 2-phenylacetamide and phenylacetic acid[5]

Willgerodt–Kindler reaction[edit]
| Willgerodt–Kindler reaction | |
|---|---|
| Named after | Conrad Willgerodt Karl Kindler |
| Reaction type | Rearrangement reaction |
| Identifiers | |
| Organic Chemistry Portal | willgerodt-kindler-reaction |
| RSC ontology ID | RXNO:0000186 |
The related Willgerodt–Kindler reaction[6] takes place with elemental sulfur and an amine like morpholine. The initial product is a thioamide for example that of acetophenone[7] which can again be hydrolyzed to the amide. The reaction is named after Karl Kindler

A possible reaction mechanism for the Kindler variation is depicted below:[8]

The first stage of the reaction is basic imine formation by the ketone group and the amine group of morpholine to give an enamine. This reacts as a nucleophile with electrophilic sulfur, similar to an Stork enamine alkylation reaction.[verification needed] The actual rearrangement reaction takes place when the amine group attacks the thiocarbonyl in a nucleophilic addition temporarily forming an aziridine and the thioacetamide by tautomerization.
References[edit]
- ^ Willgerodt, Conrad (1887). "Ueber die Einwirkung von gelbem Schwefelammonium auf Ketone und Chinone". Berichte der Deutschen Chemischen Gesellschaft. 20 (2): 2467–2470. doi:10.1002/cber.18870200278.
- ^ Willgerodt, Conrad (1888). "Ueberführung von Ketonen und Aldehyden in Säuren und Säureamide mittelst gelben Schwefelammoniums". Berichte der Deutschen Chemischen Gesellschaft. 21 (1): 534–536. doi:10.1002/cber.18870200278.
- ^ Carmack, Marvin; F. DeTar, DeLos (1946). "The Willgerodt and Kindler Reactions. III. Amides from Acetylenes and Olefins; Studies Relating to the Reaction Mechanisms". Journal of the American Chemical Society. 68 (10): 2029–2033. doi:10.1021/ja01214a048.
- ^ Carmack, Marvin; Spielman, M. A. (1946). "The Willgerodt Reaction". Organic Reactions. 3: 83–107. doi:10.1002/0471264180.or003.02. ISBN 9780471005285.
- ^ The Willgerodt Reaction. II. A Study of Reaction Conditions with Acetophenone and Other KetonesDeLos F. DeTar and Marvin Carmack J. Am. Chem. Soc. 1946, 68(10), 2025 - 2029. (doi:10.1021/ja01214a047)
- ^ Karl Kindler (1923). "Studien über den Mechanismus chemischer Reaktionen. Erste Abhandlung. Reduktion von Amiden und Oxydation von Aminen". Liebigs Annalen. 431 (1): 187–230. doi:10.1002/jlac.19234310111.
- ^ Organic Syntheses, Coll. Vol. 9, p.99 (1998); Vol. 74, p.257 (1997). (Article)
- ^ Name Reactions and Reagents in Organic Synthesis Bradford P. Mundy, Michael G. Ellerd, Frank G. Jr. Favaloro 2005 ISBN 0-471-22854-0
