COVID-19 vaccination in Canada
This article's lead section may be too long. (December 2022) |
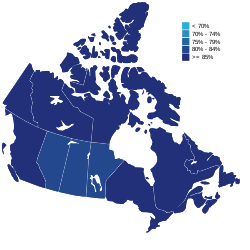 Percentage of the population vaccinated with at least one dose as of September 30, 2022 | |
| Date | December 14, 2020 – present |
|---|---|
| Location | Canada |
| Also known as | Vaccins contre la COVID-19 (French) |
| Cause | COVID-19 pandemic in Canada |
| Organized by | Health Canada Public Health Agency of Canada Provincial and Territorial governments Municipal government in Canada |
| Participants | 32,663,177 people with at least one dose administered of Pfizer, Moderna, AstraZeneca, Johnson & Johnson or Novavax 31,392,262 fully vaccinated people (to which the first and second doses of vaccine were administered) 19,250,351 people with a booster dose [1] |
| Outcome | 85.41% of the Canadian population has received at least one dose of a vaccine [1] 82.08% of the Canadian population is fully vaccinated 49.80% of the Canadian population has received at least one booster dose |
| Website | Government of Canada |
| Part of a series on the |
| COVID-19 pandemic |
|---|
 |
|
|
|
COVID-19 vaccination in Canada is an ongoing, intergovernmental effort coordinated between the bodies responsible in the Government of Canada to acquire and distribute vaccines to individual provincial and territorial governments who in turn administer authorized COVID-19 vaccines during the COVID-19 pandemic in Canada. Provinces have worked with local municipal governments, hospital systems, family doctors and independently owned pharmacies to aid in part, or in full with vaccination rollout.[2] The vaccination effort in full is the largest such immunization effort in the nation's history. The vaccination effort began December 14, 2020, and is currently ongoing.[3]
Health Canada is responsible for approval and regulation of vaccines (and other pharmaceuticals), while the Public Health Agency of Canada (PHAC) is responsible for public health, emergency preparedness and response, and infectious and chronic disease control and prevention. Vaccines are authorized by Health Canada, purchased by the Government of Canada and distributed by PHAC to individual provinces and territories in tranches based on various factors such as population size and prioritized peoples. The National Advisory Committee on Immunization (NACI) has also issued recommendations on how vaccines should be distributed, in what intervals and to which populations. NACI has also been involved in recommendations on the use or disuse of vaccines to certain ages or populations.
The National Research Council Canada (NRC) has made investments in the domestic development of vaccine candidates, including candidates by the University of Saskatchewan and Variation Biotechnologies. In May 2020, the NRC announced a planned agreement to conduct clinical trials of a vaccine candidate by Chinese company CanSino Biologics, and plans to manufacture it at its facilities in Montreal once authorized. However, the deal collapsed due to strained Canada–China relations, and the federal government later announced commitments to purchase vaccines being produced by AstraZeneca, Moderna, Pfizer and Janssen.
In early 2021, both Pfizer–BioNTech and Moderna did not ship the agreed upon quantities of secured vaccines to Canada and other countries, due to manufacturing challenges.[4] This caused a vaccine shortage and significant slowdown in vaccine rollout. By mid-February 2021, significant increases in manufacturing and delivery of vaccines in conjunction with a recommendation by NACI to extend second dose administration to a maximum of 16 weeks resulted in a larger ramp-up in vaccine delivery across the nation and by July 2021, Canada's vaccine supply had grown to allow a return to shortened dose intervals.[5]
Following Health Canada's emergency authorization of the Pfizer–BioNTech vaccine on December 9, 2020, mass vaccination efforts began across the country on December 14, 2020. The agency later authorized the Moderna vaccine on December 23, 2020. On February 26, 2021, Health Canada authorized the Oxford–AstraZeneca vaccine for use. Following concerns of blood clotting events the Oxford-AstraZeneca vaccine was largely discontinued for use, and those who had already received a first dose were encouraged to receive an mRNA vaccine as their second dose.[6] The Janssen vaccine was authorized on March 5, 2021; however, Canada did not receive a delivery of the Janssen vaccine until April 28, 2021;[7] which was then destroyed due to contamination issues at its factory of origin.[8][9] Use of Janssen was put on hold until November 2021, when the government acquired doses for use with vaccine-hesitant populations.[10]
Canada became the first country to authorize a COVID-19 vaccine for people younger than 16 after approving Pfizer's vaccine for children aged 12 to 15 on May 5, 2021.[11] In August 2021, the Moderna vaccine was authorized for use in children aged 12 and up.[12] On September 16, 2021, Health Canada granted full approval to the Moderna and Pfizer vaccines and in November 2021, Health Canada approved the Pfizer-BioNTech and Moderna vaccines for use as booster (or third doses).[13][14] On November 19, 2021, Health Canada approved the Pfizer-BioNTech vaccine (with a lower dosage) for children aged five to eleven.[15] On February 17, 2022, Health Canada approved the Novavax vaccine, which is the first approved COVID-19 protein subunit vaccine.[16]
Background and timeline[edit]
Preparations[edit]
In March 2020, the federal government announced a CA$275 million investment for "coronavirus research and medical countermeasures"[17] and on April 23, 2020, over CA$1 billion in additional financial support was announced. This funding for "national medical research strategy to fight COVID-19" included "vaccine development, the production of treatments, and tracking of the virus."[17]
Nancy Harrison and Cédric Bisson are co-chairs of the Therapeutics Task Force (TTF), which is tasked with prioritizing "financial support for promising COVID-19 treatment projects."[18] The secretariat of the Therapeutics Task Force is housed at the Department of Innovation, Science and Industry (ISED). Joanne Langley and J. Mark Lievonen are the co-chairs of the Vaccine Task Force (VTF) advising the federal government on "vaccine development, related bio-manufacturing and international partnerships".[18] The secretariat of the VTF is supported by the National Research Council of Canada (NRC).[18] The NRC was tasked with identifying potential vaccine candidates, and helping to expedite clinical trials and approval of them in Canada.[19]
Failed CanSino agreement[edit]
The NRC signed agreements with several companies that had been developing vaccine candidates, including Variation Biotechnologies (which has a facility in Ottawa), and the University of Saskatchewan Vaccine and Infectious Disease Organization–International Vaccine Centre (VIDO–InterVac). Canada did not have facilities capable of producing COVID-19 vaccines at the outbreak of the pandemic;[20][Notes 1][21][Notes 2][Notes 3][22] the NRC earmarked $45 million to upgrade its laboratories in Montreal in anticipation of vaccine production.[23]
On May 6, 2020, the NRC reached an agreement with Chinese pharmaceutical company CanSino Biologics to conduct clinical trials for its COVID-19 vaccine candidate Ad5-nCoV at Dalhousie University in Nova Scotia.[24] The NRC cited its "strong collaborative history" with CanSino, and past work on adenovirus vaccines, as part of its decision.[19] CanSino's candidate was the first to enter phase II trials, and was considered by the VTF to be one of the top candidates.[23]
The NRC would have received "a non-exclusive right to use, produce, and reproduce the vaccine for emergency pandemic use", free-of-charge. The agreement was announced by Prime Minister Justin Trudeau on May 12 during a press briefing, and trials were expected to begin within the next two weeks.[24] The trials were formally approved on May 15; the NRC stated that it hoped to produce between 70,000 and 100,000 doses of the CanSino vaccine per-month.[23]
The CanSino agreement was impacted by Canada's strained relations with China; the shipments of vaccine doses were blocked by Chinese customs, and the State Council of the People's Republic of China refused to authorize the shipment.[24][19] In July 2020, after reports emerged over the delays, the NRC stated that CanSino "[remained] very committed to the Canadian clinical trials."[23]
Agreements with Moderna, Pfizer, and AstraZeneca[edit]
With the CanSino deal falling through, and the VTF revising its recommendations based on clinical trial data from other countries, Minister of Public Services and Procurement Anita Anand announced on August 5, 2020, that the federal government had committed to purchasing doses of the Moderna and Pfizer–BioNTech vaccines.[23][25] In anticipation of a COVID-19 vaccine, the Canadian government purchased more than 75 million hypodermic needles and syringes.[26]
After the NRC found that its current lab would not meet good manufacturing practices Prime Minister Trudeau announced on August 31 a federal investment of $126 million to "design, construct, commission and qualify a new biomanufacturing facility"—the Biologics Manufacturing Centre, which was projected to be completed by the end of July 2021.[27] It would be built next to the NRC's current Royalmount site in Montreal and have a production capacity of approximately 2 million does per-month".[27]
In September 2020, the federal government committed to purchasing 20 million doses of the Oxford–AstraZeneca vaccine. It also announced that it would make a $440 million investment in the COVAX initiative, to help fund the equitable procurement of COVID-19 vaccines worldwide.[28][29]
In November 2020, Conservative Party leader Erin O'Toole criticized the federal government for focusing too heavily on the CanSino agreement, arguing that he "would not have put all our eggs in the basket of China."[30]
On November 20, 2020, Pfizer and BioNTech submitted their vaccine for emergency authorization to Health Canada.[31]
Initial approvals and rollout[edit]

On December 9, 2020, Health Canada authorized emergency use of the Pfizer–BioNTech COVID-19 vaccine under the Interim Order Respecting the Importation, Sale and Advertising of Drugs for Use in Relation to COVID-19.[32] The Public Health Agency of Canada supervises the rollout and administration of the vaccine.[33] By mid-December 2020, Pfizer had agreements to supply 20 million doses to Canada.[34] On December 10, 2020, Prime Minister Justin Trudeau announced the implementation of Canada's first nationwide vaccine injury compensation program in preparation for the rollout of the first approved vaccine.[35] Healthcare institutions began administering the first 30,000 doses of the Pfizer–BioNTech COVID-19 vaccine in Canada on December 14. A total of 249,000 doses was expected to be delivered by the end of 2020.[36]
Because of the cold storage logistics, the initial doses were delivered to 14 distribution sites in the provinces, with none being sent to the Canadian territories since they could not properly store the vaccine. The 14 original distribution sites are located in St. John's, Halifax, Charlottetown, Miramichi, Quebec City, Montreal, Ottawa, Toronto, Winnipeg, Regina, Edmonton, Calgary, and two in the Greater Vancouver area.[37] The Canadian government expects the vaccine to be administered to high-priority groups, designated by each province, until the end of March.[36] Most provinces were first prioritizing some subset of healthcare workers, except for Quebec, which is prioritizing residents of long-term care homes, as well as British Columbia, New Brunswick, and Prince Edward Island, which are prioritizing both. Most provinces also have plans to expand priority status to additional groups—such as the elderly, or adults in Indigenous communities—before expanding to the general public.[38]
In the same week as initial vaccinations for the Pfizer–BioNTech COVID-19 vaccine, Health Canada authorized the Moderna vaccine on December 23, and it was announced that Canada would receive 168,000 doses of the Moderna vaccine before the end of 2020.[39][40] Unlike the Pfizer–BioNTech vaccine, the Moderna vaccine does not require extreme cold temperature storage[40] which meant that as of December 28, Northwest Territories and Yukon had received their first shipments of 7,200 each of the Moderna vaccine.[41] Vaccinations in each territory are not scheduled to occur until mid-January.[42]
On February 2, 2021, Trudeau announced a deal with Novavax to produce COVID-19 vaccines at the Biologics Manufacturing Centre, making it the first to be produced domestically.[43] At that time, the Novavax COVID-19 vaccine was awaiting approval by Health Canada.[44] This is the first deal signed by Canada that allows a domestic manufacturing of a foreign vaccine. The contract with Novavax is for 52 million doses of the vaccine.[43] Following VTF and TTF recommendations, the federal government announced investment in two biomanufacturing companies—Vancouver-based Precision NanoSystems Incorporated (PNI) and Markham, Ontario-based Edesa Biotech Inc. (Edesa).[45] PNI, biotechnology company, will receive up to $25.1 million to build a "$50.2 million biomanufacturing centre to produce vaccines and therapeutics for the prevention and treatment of diseases such as infectious diseases, rare diseases, cancer and other areas of unmet need". Edesa will receive up to $14 million to Edesa Biotech to "advance work on a monoclonal antibody therapy for acute respiratory distress syndrome, which is the leading cause of COVID-19 deaths.[45]
By February 2021, the NRC and the ISED support for domestically-produced vaccines and therapeutics for COVID-19 included $37 million in stage 1 funding for six vaccine candidates, and seven therapeutics candidates.[46]
In February 2021, a call for proposals to develop Canada's Vaccine Injury Support Program (VISP) program was launched. The call closed on February 24. The Public Health Agency of Canada established a committee to review proposals the following month, swiftly and unanimously choosing Raymond Chabot Grant Thornton to act as third party administrator.[47]
Manufacturing delays[edit]

In mid to late January, details emerged about manufacturing delays by both authorized vaccines made by Pfizer and Moderna affecting the country's vaccine rollout in late January through the month of February.[48][49] On February 3, the European Commission approved delivery of COVID-19 vaccines to Canada, in spite of production constraints in Europe. Canada was one of many countries that applied for delivery and does not have its own manufacturing capacity.[50] By February 4, Major General Fortin said while that Pfizer explained its dramatically lower vaccine shipments—with a decrease of 80% in all Pfizer shipments—as caused by "plant upgrades at a facility in Belgium", Moderna has offered no explanation for similar delays.[4] By early 2021, the speed of Canada's deployment of COVID-19 vaccines had become the topic of widespread public discussion, along with the related question of why the vaccines were not being produced in Canada.[51][52]
Fortin said that 180,000 Moderna doses had arrived in Canada on the morning of February 4 and Canada is "still expecting 2 million Moderna doses by the end of March.[4] On February 4, Fortin said that about 70,000 Pfizer-BioNTech doses will arrive in the next week.[53]
On February 3, COVAX published the country-by-country vaccine distribution forecast to COVAX participants—Canada will receive 1,903,200 doses of the Oxford–AstraZeneca vaccine by the end of the first half of 2021.[54][55]
On February 12, the minister of Public Services and Procurement Canada (PSPC) announced that Canada had "negotiated an accelerated delivery schedule for Pfizer-BioNTech’s COVID-19 vaccine."[56] By the end of September, Canada will receive the 40 million Pfizer-BioNTech doses.[56]
Return to regular shipments[edit]

Major General Fortin announced on February 18, that the period in which Canada experienced limited supplies, is now over and there will be an "abundance of supplies" in the spring and summer months. This will result in a "significant scaling-up of immunization plans in provinces."[57] Pfizer's new "locked in" delivery schedule includes 475,000 doses in February and 444,600 per week in March, according to the PHAC's vaccine distribution tracker.[58][57] A February 18, 2021 article in New York Times said that Canadians were concerned about the rollout of the vaccination program.[59] According to the same Times columnist, who had interviewed those involved in "vaccine development, epidemiology, infection control and medical supply chains", they all said it was not surprising that Canada's rollout was not happening as planned. While they could understand why Canadians were frustrated, they said, this is the "nature of new vaccines".[52]
Approval of Oxford-AstraZeneca and Johnson & Johnson[edit]
On February 26, Health Canada authorized the Oxford–AstraZeneca COVID-19 vaccine for use.[60] The agency authorized the use of two versions of the same vaccine, one to originate from the United Kingdom, and the other produced in India by the Serum Institute of India (trade name "Covishield").[60] The first shipment of 500,000 doses of Oxford-AstraZeneca arrived on March 3, 2021. The doses were marked with an April 2, 2021 expiry date, necessitating speedy administration.[61][62]
The Janssen COVID-19 vaccine was authorized on March 5, 2021, to become the fourth vaccine to receive Health Canada approval.[63] The vaccine was the first single-dose vaccine to be authorized in Canada.[64] Delivery times for the vaccine remain unknown.[65]
After an agreement was reached with Pfizer to move up some deliveries, it was projected a total of 8 million vaccine doses (from three suppliers) will be available in Canada by the end of March 2021.[66] To maximize distribution of first doses, NACI issued a recommendation that second doses be administered up to four months after the first dose.[67]
Minister Anand confirmed on March 26, 2021, that 1.5 million Oxford-AstraZeneca shots would arrive from the United States on March 30, 2021,[68] and that 10 million shots of the Johnson & Johnson vaccine would arrive between April and September 2021.[69]
Vaccination ramp-up and increase in deliveries[edit]

In mid-April 2021, the Government of Canada reached a deal with Pfizer to receive 8 million more vaccines on top of what was already ordered, with more than 2 million vaccines coming into the country per week beginning in the month of May.[70] Moderna however continued to delay deliveries and slashed an order for the end of April from 1.2 million vaccines to 650,000.[71]
Due to public frustration with variable vaccine booking systems across the country, a small volunteer group known as Vaccine Hunters Canada created a website, Twitter and Facebook page to help "eligible Canadians find vaccines." The group posts relevant information on appointments available, eligibility requirements and other tips to the public that might otherwise not be common knowledge.[72][73][74][75] On April 28, 2021, the City of Toronto officially partnered with the group to provide them end of day information in regards to vaccine availability and appointments that are currently unused, as well as information for pop-up vaccine clinics.[76][77][78]
The first shipment of the Johnson & Johnson vaccine arrived in Canada on April 28, 2021. The country received 304,800 doses of the single-shot vaccine in its first shipment.[79] However, Health Canada held the shipment for a safety review, citing production issues at the facilities of Emergent BioSolutions.[80][81]

In May 2021, NACI stated a recommendation that mRNA-based vaccines were preferred over viral vector vaccines such as the AstraZeneca and Johnson & Johnson vaccines due to the rare risk of post-vaccination embolic and thrombotic events, with vice-chairman Shelly Deeks stating that "individuals need to have an informed choice to be vaccinated with the first vaccine that’s available, or to wait for an mRNA vaccine." NACI received criticism for the recommendation, with critics arguing that it contradicted the view that patients should receive the first vaccine they are offered, and that the statement was mixed messaging over the safety of the vaccines that could lead to increased hesitancy.[82][83][84]
On May 5, 2021, Canadian health officials authorized Pfizer's vaccine for adolescents 12 to 15 years old, becoming the first country in the world to do so.[11]
On May 11 and 12, multiple provinces, including Alberta, Manitoba, Nova Scotia, Ontario, and Saskatchewan, announced that they would suspend their use of the AstraZeneca vaccine for first doses, citing supply shortages, with some provinces also citing the blood clot risk.[85][86][87]
Deliveries from manufacturers continued to accelerate in mid-May. In the week of May 16, Canada was expected to receive a record 4.5 million doses of the two mRNA vaccines from Pfizer and Moderna.[88]
In British Columbia, the Vancouver Coastal Health authority apologized on May 31 after accidentally administering the Moderna vaccine to 12 adolescents who were to receive the Pfizer vaccine per the approval.[89]
50% vaccination milestone and second doses[edit]


By May 22, 2021, at least half of the overall population had received at least one vaccine dose, with Canada having administered 20.328 million vaccine doses overall.[90] The country has passed other large countries in coverage like the U.S. vaccination program.[91]
As new arrivals of the Oxford-AstraZeneca vaccine came into the country in late May, provinces chose whether to administer second doses, for example as Ontario is going forward with,[92] or providing people who took a first dose to choose a different second vaccine, as in British Columbia.[93]
The country's percentage of single doses had been disproportionately high in comparison to second doses, due to NACI's earlier recommendations for intervals of up to four months. This led to calls from medical experts for provinces to prioritize second doses, citing the possibility having only one dose would make populations vulnerable to SARS-CoV-2 variants.[94] By late-May, amid an increased supply, several provinces had begun to shorten their intervals for second doses.[95] NACI followed suit on May 28, issuing new guidance that second doses should be administered "as soon as possible", especially to high-risk individuals.[96][97]
For the week following the Victoria Day weekend, Canada expected a smaller vaccine shipment than normal, as Pfizer–BioNTech sent more vaccine the previous week.[98]
The Vaccine Injury Support Program (VISP) was launched on June 1, 2021, at which point it began accepting claims. Program statistics were first reported publicly on December 1, 2021.[99]
The government had previously indicated that Canada would have 55 million doses delivered by the end of July 2021, but this mostly included Pfizer distributions. Moderna had now committed to delivering 11 million doses in July 2021, with 5 million of those doses potentially arriving in June. Combined with an expected 9 million Pfizer doses to be delivered, this would be enough to give Canada 68 million doses by the end of July.[100]
Vaccine mandates and full approval[edit]
On August 13, 2021, Minister of Intergovernmental Affairs Dominic LeBlanc and Transport Minister Omar Alghabra announced that the federal government plans to mandate the vaccination of all federal public servants, employees in federally-regulated transport industries, and passengers of commercial air travel, interprovincial rail service, and large marine vessels with overnight accommodations (e.g. cruise ships). The government will also "expect" the vaccination of all employees in industries regulated by the Canada Labour Code.[101][102] At the time of the announcement, the Government of Canada estimated that there are approximately 19,000 employers and 1,235,000 employees (8% of all workers in Canada) subject to the vaccine mandate.[103]
Soon after the announcement by the Government of Canada, some provinces began mandating vaccination for their public servants, starting with the Ontario government, who announced on August 17 that public health services and school boards will be required to have a "vaccination policy" in place that would require an employee to either be fully vaccinated or undergo regular testing.[104][105] The same day, the Quebec government announced that healthcare workers in both the public and private sector must be fully vaccinated by October 1, 2021.[106] New Brunswick was the next province to follow, mandating vaccination or regular testing for provincial public servants.[107][108]
On August 27, 2021, the Moderna vaccine was authorized for emergency use in children aged 12 and up.[109]
On September 16, 2021, with the expiration of the aforementioned Interim Order, Health Canada officially granted full approval to the Moderna and Pfizer vaccines for individuals aged 12 and older.[110]
Approval for younger children and boosters[edit]
On October 18, 2021, Pfizer-BioNTech submitted the vaccine for approval to Health Canada for children aged five to eleven. The submission called for smaller doses of the same vaccine used for adults.[111] A few days later, Canada ordered 2.9 million child-dose Pfizer vaccines.[112]
In late October, NACI recommended the use of booster shots (third doses) of vaccines for certain immunocompromised peoples, people over the age of 70, people who had received a full series of the Oxford-AstraZeneca vaccine or Janssen vaccine and those in healthcare following 6 months of full vaccination. Many provinces then followed suit.[113]
On November 19, Health Canada approved the Pfizer-BioNTech vaccine for children aged five to eleven.[114] Health Canada recommended children receive the smaller doses, with an interval of at least 8 weeks between vaccinations.[115][116] Doses began to arrive several days later.[117]
Amid the discovery of the Omicron variant, NACI reviewed information on recommendations for booster shots for most adults.[118] Prime Minister Trudeau said that if boosters were offered to all, Canada would have ample supply.[119] Some provinces began offering boosters to those outside of NACI recommendations in advance.[120]
On January 28, 2022, truckers from across Canada drove to Ottawa to protest against mandatory vaccination for truckers crossing the border from the U.S. to Canada.[121]
On April 5, 2022, NACI began recommending that fourth doses be given to vulnerable populations, namely seniors living in long-term care homes and people aged 80 and up.[122]
On July 14, 2022, Health Canada approved the Moderna vaccine for children aged between six months and five years, rendering an additional 1.7 million people eligible for vaccination. Children in this age group will receive approximately a quarter of the dose given to adults.[123][124]
New formulations[edit]
On September 1, 2022, Moderna's new bivalent vaccine, which targeted both the original strain of SARS-CoV-2 and the Omicron BA.1 variant, received authorization from Health Canada.[125] A week later, the first shipments arrived in Quebec,[126] followed by Ontario and other provinces shortly after.[127] The bivalent vaccine was not initially set to replace the original Moderna vaccine, but instead to coexist with it, with the federal government maintaining stocks of both.[128]
On October 7, 2022, Pfizer's new bivalent vaccine, which targeted the original strain of SARS-CoV-2 as well as the Omicron BA.4 and BA.5 subvariants, was approved by Health Canada for individuals aged 12 years and up. According to Health Canada, 97% of COVID-19 cases in Canada were either of the BA.4 or BA.5 subvariant as of mid-September 2022. As such, NACI issued a recommendation for people to use one of the approved bivalent vaccines as their upcoming booster shot.[129] The Pfizer bivalent vaccine was later approved for children aged 5 to 11 years old on December 9.[130]
Health Canada received a submission from Moderna on June 29, 2023 for yet another reformulated COVID-19 vaccine based on the Omicron XBB.1.5 subvariant of SARS-CoV-2.[131] The updated version was tested in a small human trial of 101 participants who had previously received four doses of previous formulations of the Moderna COVID-19 vaccine. Its safety profile was found to be "consistent with previously authorized vaccines."[132][133] It was authorized for use in people six months and older on September 12, 2023.[134][135]
Vaccination by province and territory[edit]
Vaccine rollout[edit]
In December 2020, NACI issued initial recommendations for a multi-stage rollout of vaccines that would prioritize specific populations, beginning with people 70 and older, healthcare workers, employees and residents of long-term care facilities, and Indigenous people. The rollout then progressed towards other vulnerable groups, including first responders, essential workers, and people with comorbidities, before opening up to the wider general public.
| COVID-19 vaccinations in Canada by province and territory, September 30, 2022 () | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Province | Population | People who have at least one dose | People fully vaccinated | Total population with at least one dose (%) | Total population fully vaccinated (%) | Ref | ||||
| 5,174,724 | 4,559,145 | 4,399,082 | 87.43% | 84.36% | [115] | |||||
| 4,444,277 | 3,630,480 | 3,425,775 | 81.72% | 77.11% | [115] | |||||
| 1,179,906 | 976,094 | 916,695 | 82.73% | 77.70% | [115] | |||||
| 1,382,904 | 1,145,269 | 1,101,542 | 82.77% | 79.61% | [115] | |||||
| 14,789,778 | 12,790,406 | 12,302,531 | 86.27% | 82.98% | [115] | |||||
| 8,585,523 | 7,907,623 | 7,206,045 | 91.90% | 83.75% | [115] | |||||
| 783,721 | 700,212 | 661,271 | 88.72% | 83.79% | [115] | |||||
| 160,536 | 156,154 | 144,466 | 95.03% | 87.92% | [115] | |||||
| 982,326 | 889,828 | 842,600 | 89.70% | 84.94% | [115] | |||||
| 520,286 | 498,432 | 476,756 | 95.75% | 91.59% | [136] | |||||
| 42,596 | 36,691 | 35,380 | 85.36% | 82.31% | [115] | |||||
| 44,991 | 41,094 | 39,663 | 90.31% | 87.16% | [115] | |||||
| 39,536 | 33,543 | 29,021 | 85.13% | 73.65% | [115] | |||||
| 38,131,139 | 33,364,971 | 31,580,827 | 87.24% | 82.57% | [115] | |||||
Additionally, 198,370 Moderna, 5,850 Pfizer-BioNTech and 460 Oxford-AstraZeneca vaccine doses have been kept for "Federal Application" for use with the Public Health Agency of Canada (PHAC), Canadian Armed Forces and the Correctional Service of Canada.[137]
Vaccine mandates, policies and measures[edit]


On a federal scale, the Government of Canada mandated all federal workers be vaccinated by October 29, 2021, or face termination.[138] Travellers over the age of 12 on all federally regulated modes of transportation (such as domestic and international flights departing from most airports in Canada, those riding on Via Rail and Rocky Mountaineer trains as well as cruise ships) were required to show proof of full vaccination against COVID-19 beginning October 30, 2021. Although up until November 30, 2021, the government offered a one-month grace period to offer those who were not yet fully vaccinated against COVID-19 to provide a recently negative test prior to boarding.[138][139] Since the implementation of the vaccine mandate, wrongful dismissal lawsuits from fired unvaccinated workers have been piling up.[140] Legal scholars also questioned the constitutionality of vaccine mandates, not only on the basis of individual exemptions, but also the extent of the impact that firing so many workers would have on the Canadian population at-large. Alternatively, Si Wen Shen from the Université de Montréal Faculty of Law proposed the idea of provincial governments invoking Section 33 (notwithstanding clause) of the Canadian Charter of Rights and Freedoms in order to circumvent any legal challenges on constitutional grounds.[141]
Many private companies nationwide like Air Canada[142] and major accounting firms have mandated vaccination policies internally.[143] Most education institutions like colleges and universities nationwide have announced mandates for employees and students[144] and for-profit long-term care home companies such as Chartwell Retirement Residences, Revera and Sienna Senior Living have all announced mandates for vaccination for employees.[145]
The following is a table representing individual provincial measures, policies and mandates for vaccination requirements by province and territory:
| Province or territory | Healthcare workers | Provincial workers | Other private workers | Vaccine passport | References |
|---|---|---|---|---|---|
| Yes | Yes | Partial | Terminated[a] | [146][147][148] | |
| Terminated | Terminated | Partial | Terminated[b] | [150][151][152][153][154][155] | |
| No | Terminated | Partial | Terminated[c] | [157][158][159][160][161] | |
| Partial[d] | Partial | Partial | Terminated[e] | [162][163][164][165] | |
| Partial[f] | Partial | Partial | Terminated[g] | [167][168][169][170][171] | |
| Partial[h] | No | No | Terminated[i] | [172][173][174] | |
| Yes | Partial | Partial | Terminated[j] | [175][176][177][178][179] | |
| Partial[k] | No | Partial | Terminated[l] | [181][182][183] | |
| Yes | Yes | Partial | Terminated[m] | .[184][185][186][187] | |
| Yes | Yes | Partial | Terminated[n] | [188][189][190][191][192][193][194] | |
| Yes | Yes | Partial | Terminated[o] | [195][196][197] | |
| No | Partial | No | Terminated[p] | [199][198][200] | |
| January 10, 2022[q] | No | No | No | [201] |
- ^ BC's Restrictions Exemption Program operated from September 13, 2021 to April 8, 2022.
- ^ Alberta's Restrictions Exemption Program operated from September 20, 2021 to February 8, 2022. It allowed certain businesses to opt-in to using proof of vaccination, therefore not being subject to certain COVID-19 restrictions. Those without a proof of vaccination could present a recent negative test as an alternative.[149]
- ^ Saskatchewan's proof-of-vaccination policy ran from September 28, 2021 to February 14, 2022. It gave residents the option of having proof of a recent negative test as an alternative to proof-of-vaccination.[156]
- ^ Unvaccinated healthcare workers can opt to undergo regular testing 3 times per week[162]
- ^ Manitoba's Immunization Card requirement operated from July 17, 2021 to March 1, 2022
- ^ Some Ontario hospitals have instituted a vaccine mandate despite no provincial mandate and others offer a rapid testing alternative to unvaccinated workers [166]
- ^ Ontario's vaccine passport program operated from September 22, 2021 to March 1, 2022.
- ^ Unvaccinated healthcare workers must be tested three times per week, and lose bonuses[172]
- ^ Quebec's vaccine passport program operated from September 1, 2021 to March 12, 2022.
- ^ New Brunswick's proof of vaccination program operated from September 22, 2021 to February 27, 2022.
- ^ Unvaccinated healthcare workers can be tested up to three times per week[180]
- ^ PEI's Vax Pass Program operated from October 5, 2021 to February 27, 2022.
- ^ Nova Scotia's proof of vaccination program operated from October 4, 2021 to February 27, 2022.
- ^ Newfoundland and Labrador's proof of vaccination program operated from October 22, 2021 to March 13, 2022.
- ^ Yukon's vaccine passport program operated from November 30, 2021 to March 18, 2022.
- ^ NWT operated an voluntary proof of vaccination program from October 22, 2021 to March 1, 2022. It allowed certain businesses to opt-in to using proof of vaccination, therefore not being subject to certain COVID-19 restrictions.[198]
- ^ Nunavut has announced a vaccine mandate for most healthcare workers in the territory by January 10, 2022. Unvaccinated workers may be reallocated to other positions or be frequently tested
National vaccination progress and orders[edit]
Total number of people receiving vaccinations in Canada as of September 11, 2022[1]
Vaccines delivered per pharmaceutical company as of July 14, 2023[202]
There are several COVID-19 vaccines at various stages of development around the world. As of 2 December 2020[update] the Canadian government had invested over $1 billion,[203] including pre-placed orders for seven different vaccines, four of which are now authorized for use by Health Canada. These pre-orders total more than 400 million doses.[204][205][203][206] Six of the seven vaccines require two doses each to be effective, with the exception of the Janssen Pharmaceutica (Johnson & Johnson) vaccine which only requires one dose.[203]
| COVID-19 vaccines on order in Canada () | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Vaccine | Progress | Doses ordered | Approval | Deployment | ||||||
| Pfizer–BioNTech | phase III clinical trials | 51-76 million[207][208][209] | ||||||||
| Moderna | phase III clinical trials | 44 million[211] | ||||||||
| Oxford–AstraZeneca | phase III clinical trials | 23.9 million[213] | ||||||||
| Janssen (Johnson & Johnson) | phase III clinical trials | 10-38 million | ||||||||
| Novavax | phase III clinical trials | up to 76 million | ||||||||
| Medicago | phase III clinical trials[222] | up to 76 million | ||||||||
| Sanofi–GSK | phase III clinical trials | up to 72 million | Pending | Pending | ||||||
Pharmacological data and developments[edit]
As the COVID-19 vaccines are relatively new, certain aspects of delivery and pharmacological care of the products have changed rapidly over the course of the vaccination program.
Second dose administration[edit]
Early in the vaccination effort, debate on when to administer the second dose of the approved vaccines appeared medically and politically.[225] Each manufacturer had recommended respectively 21 days apart for Pfizer-BioNTech;[226] 28 days apart for Moderna;[227] and 4–12 weeks apart for Oxford-AstraZeneca.[228] New data suggested the manufacturer's recommendation could be lengthened, in theory allowing for more people to be vaccinated as supply efforts ramped up. Stanley Plotkin and Neal Halsey wrote an article published by Oxford Clinical Infectious Diseases that urged single dose interim use in order to extend vaccination to as many people as possible until vaccine availability improved.[229] Several other articles and media provided evidence for delaying 2nd doses in the same line of reasoning.[230][231][232] The province of Quebec began to withhold second dose administration,[233][234] and eventually adopted a 90-day strategy.[235] The National Advisory Committee on Immunization (NACI) had initially only recommended a maximum of 42 days in between doses.[236] The province of British Columbia had also followed suit and began plans to extend second dose administration to a four-month delay.[237]
On March 4, 2021, the National Advisory Committee on Immunization (NACI) recommended that a maximum of four months or 112 days could be achieved effectively between second doses.[238] Other provinces began to adjust their administration programme based on the federal-level recommendation.[237]
Quebec no longer recommends a second dose for those previously infected with COVID-19, as the immunity is considered strong enough.[239] People still wanting to receive a second dose will be allowed to get it.
Beginning May 24, 2021, Saskatchewan became one of the first provinces to offer second dose administration on a shortened interval timeline, allowing people to book second dose appointments based on their age (beginning 80+) or first dose administration date.[240] Meanwhile, Ontario allowed people who received their first dose of the Oxford-AstraZeneca vaccine between March 10 and 19, 2021 to book second dose appointments of the same vaccine.[241]
Pfizer-BioNTech vial capacity[edit]
In January 2021, the province of Saskatchewan discovered it could draw extra doses of the Pfizer vaccine out of vials labelled as having 5 doses.[242]
As Pfizer prepared for industrial retooling to match international demands for its vaccine, causing a supply slowdown in Canada in January and February 2021, the company announced that their vials indeed contained 6 doses of vaccine, rather than 5.[243] Health Canada accepted the relabelling of the product, however the revelation required the acquisition of millions of low dead space syringes in order to extract the last dose from the vial.[244]
Pfizer-BioNTech storage requirements[edit]
Initially the Pfizer-BioNTech vaccine was required to be stored at ultra-cold temperatures (between -80 and -60 °C) in perpetuity, until application (in which time after dilution, the vaccine must be used within 6 hours).[245] On February 25, Health Canada recommended that the vaccine could be stored at regular freezer temperatures (between -25 and -15 °C) for up to two weeks.[246]
Oxford-AstraZeneca age range[edit]
On March 1, 2021, two days after the first delivery of the vaccine, the NACI announced that it would recommend that the Oxford-AstraZeneca vaccine not be given to patients older than 65 due to “the insufficiency of evidence of efficacy in this age group at this time.”[247] This recommendation was based on clinical trial data that only went as far as December 7, 2020.[248] Quebec did not follow this recommendation, but its own Immunization Committee recommended that the Pfizer and Moderna vaccines be given to higher-priority groups in the event of limited vaccine availability (with the AstraZeneca vaccine given to lower-priority groups).[249] On March 16, NACI revised its recommendations based on newly obtained efficacy data from other countries.[248]
On March 29, 2021, NACI recommended that the Oxford-AstraZeneca vaccine not be given to patients younger than 55, citing reports of a rare, increased risk of blood clots with low levels of blood platelets (post-vaccination embolic and thrombotic events). Most provinces adhered to this recommendation.[250][251][252] In April 2021, due to the third wave of infections, and increased hesitancy towards the vaccine due to these side effects, multiple provinces (including Alberta and Ontario) went against NACI guidance and began to distribute the vaccine to people as young as 40.[253][254][255] On April 23, NACI changed its guidance to recommend the vaccine be offered to patients as young as 30 years old if benefits outweigh the risks, and the patient "does not wish to wait for an mRNA vaccine".[256]
Cessation of Oxford-AstraZeneca in certain provinces[edit]
Due to increased reports of post-vaccination embolic and thrombotic events, on May 11, 2021, the provinces of Alberta and Ontario decided to cease offering first dose appointments of the Oxford-AstraZeneca vaccine.[257][258] On May 12, Nova Scotia and Manitoba also announced similar restrictions,[259] with Quebec following the next day.[260]
Mixing of vaccines[edit]
Health Canada and NACI began looking into the possibility of mixing different vaccines in about April 2021.[261] As the future of AstraZeneca vaccines in Canada became increasingly uncertain, due to both shipment delays and the risk of rare blood clots, and preliminary data on the subject became increasingly available,[262][263][264] provincial governments' interest in the proposal grew. In early May, Quebec became the first province to approve of the mixing of an adenovirus vaccine (AstraZeneca) with an mRNA vaccine (Pfizer-BioNTech or Moderna), albeit only under certain circumstances: only people under 45 who have received a first dose of the AstraZeneca vaccine would be able to receive one of the mRNA vaccines as their second dose.[265][260] A couple of weeks earlier, Quebec had exceptionally allowed for the mixing of Moderna and Pfizer vaccines to go ahead in long-term care homes, as the supply of Moderna doses was low.[266]
On May 20, it was announced that Canada would be performing its own country-wide study on the effectiveness of mixing vaccines.[267]
On June 1, NACI approved the mixing of the three approved vaccines in Canada (Pfizer-BioNTech, Moderna, and AstraZeneca-Oxford). Specifically, they recommended mixing a first dose of the AstraZeneca vaccine with a second dose of one of the mRNA vaccines and mixing a first dose of a Pfizer or Moderna vaccine with a second dose of either mRNA vaccine.[268][269] Two weeks later, they changed their recommendations to suggest that a second dose of one of the mRNA vaccines after a first shot of AstraZeneca is "preferred", due to new evidence that seems to demonstrate an improved immune response on a mixed vaccine schedule.[270][271]
Rollout strategies[edit]
In December 2020, the NACI issued initial recommendations for a multi-stage rollout of vaccines that would prioritize specific populations in sequence, beginning with residents 70 and older, health care workers, and the employees and residents of long-term care facilities (due to the pandemic's disproportionate impact on them); and progressing towards wider age ranges, first responders, essential workers, and residents with comorbidities, before progressing towards the wider general public.[272][273]
| Stage | Recommended prioritization |
|---|---|
| 1 |
|
| 2 |
|
| 3 |
|
Key people[edit]
- Brigadier-General Krista Brodie, Vice President Logistics and Operations, PHAC (began role May 17, 2021)
- Patty Hajdu, Minister of Health
- Harjit S. Sajjan, Minister of National Defence
- Anita Anand, Minister of Public Services and Procurement
- Brigadier-General Simon Bernard, logistics
Former[edit]
- Major-General Dany Fortin, Vice President Logistics and Operations, PHAC (left the role on May 14, 2021, due to an unspecified military investigation[274])
See also[edit]
Notes[edit]
- ^ In an article in the Financial Post on January 14, 2021, responding to the delay in rolling out of Canada's vaccination program, GlaxoSmithKline Canada's Paul Lucas—who served as CEO from 1995 to 2012—said that, fifty years ago, during the Liberal premiership of Pierre Trudeau, there was little interest in chemical patents. Lucas said that this policy discouraged private investment in pharmaceutical innovations in Canada and caused the Canadian pharmaceutical industry to become focused on generic drugs.
- ^ Health policy professor emeritus Joel Lexchin said that the publicly-owned vaccine manufacturer Connaught Laboratories was privatized under the Conservative government of prime minister Brian Mulroney (1984–1993). Connaught was then part of Sanofi Pasteur. GSK acquired the Quebec-based vaccine manufacturer IAF BioChem. Sanofi and GSK—corporations outside Canada controlled decision-making about production. The three biggest vaccine makers Archived February 22, 2021, at the Wayback Machine—GlaxoSmithKline, Merck and Sanofi—lacked the new technologies essential for the production of newer forms of vaccine, that upstarts—such as Moderna and BioNTech—had mastered and by December it was evident that they were incapable of producing an effective COVID-19 vaccine.
- ^ According to Lexchin, newly-elected U.S. President Joe Biden reportedly continued the controversial America First policy of predecessor Donald Trump with respect to vaccine distribution. Canada was unable to obtain vaccine doses from the United States, even though the relevant American vaccine manufacturing facilities are relatively close to the Canada–United States border: Pfizer's facility in Kalamazoo, Michigan and Moderna's facility in Portsmouth, New Hampshire (actually a Lonza Group facility producing under contract).
References[edit]
- ^ a b c "COVID-19 vaccination coverage in Canada". January 15, 2021. Retrieved October 5, 2022.
- ^ "Which COVID-19 vaccine booking system is your public health unit using?". CP24. March 3, 2021. Archived from the original on March 22, 2021. Retrieved March 22, 2021.
- ^ "Most Canadians will get COVID-19 vaccine by September: Trudeau". Al Jazeera. November 27, 2020. Archived from the original on February 17, 2021. Retrieved March 22, 2021.
- ^ a b c Tasker, John Paul (February 4, 2021). "Canada doesn't know how many more Moderna doses will be delivered, or why there are delays". CBC. Archived from the original on May 8, 2021. Retrieved February 4, 2021.
- ^ "Canada to reach 55M vaccine doses by week's end, catching up to U.S. on second doses". CTV News. The Canadian Press. July 12, 2021. Archived from the original on July 12, 2021. Retrieved July 12, 2021.
- ^ Miller, Adam (June 1, 2021). "Canada recommends mixing and matching AstraZeneca, Pfizer and Moderna COVID-19 vaccines". CBC News. Archived from the original on June 1, 2021. Retrieved August 24, 2021.
- ^ "COVID-19 news today: Military to provide medical personnel to help Ontario's health-care system". The Globe and Mail. The Canadian Press. April 26, 2021. Archived from the original on May 6, 2021. Retrieved April 30, 2021.
- ^ "Canada awaiting results of U.S. probe before deciding plans for J&J vaccines". CTV News. The Canadian Press. May 21, 2021. Archived from the original on May 30, 2021. Retrieved May 25, 2021.
- ^ Neustaeter, Brooklyn (June 11, 2021). "Health Canada not releasing more than 300K doses of J&J vaccine over possible quality control issue". CTV News. Archived from the original on July 1, 2021. Retrieved June 28, 2021.
- ^ Thurton, David (November 5, 2021). "Canada obtaining Johnson & Johnson doses as it works to vaccinate the holdouts". CBC News. Retrieved November 5, 2021.
- ^ a b Gillies, Rob (May 5, 2021). "Canada authorizes Pfizer vaccine for age 12 and older". The Washington Post. ISSN 0190-8286. Archived from the original on August 24, 2021. Retrieved May 5, 2021.
- ^ Freeman, Joshua (August 27, 2021). "Health Canada authorizes Moderna COVID-19 vaccine for use in youth 12-17". CP24. Retrieved August 27, 2021.
- ^ "Health Canada authorizes Moderna COVID-19 vaccine as a booster shot". November 12, 2021. Archived from the original on November 12, 2021. Retrieved November 23, 2021.
- ^ Young, Leslie (November 9, 2021). "Pfizer COVID-19 booster dose authorized by Health Canada for all adults 18+ - National". Global News. Retrieved November 19, 2021.
- ^ Young, Leslie (November 29, 2021). "Pfizer COVID-19 vaccine for children aged 5-11 approved by Health Canada". Global News.
- ^ "Novavax's COVID-19 vaccine approved for Canadians 18 and over". CBC News. February 17, 2022.
- ^ a b "Prime Minister announces new support for COVID-19 medical research and vaccine development". Prime Minister of Canada (Press release). April 23, 2020. Archived from the original on May 1, 2020. Retrieved March 22, 2021.
- ^ a b c "Government of Canada announces major steps in treating and preventing COVID-19 through vaccines and therapies". Science and Economic Development Canada (Press release). August 5, 2020. Archived from the original on August 7, 2020. Retrieved March 22, 2021.
- ^ a b c Walsh, Marieke; Mcarthur, Greg (March 4, 2021). "Canada's missed shots: How Ottawa's COVID-19 vaccine promises were out of step with reality". The Globe & Mail. Retrieved August 27, 2021.
- ^ Lexchin, Joel (February 8, 2021). "The roots of Canada's COVID-19 vaccine shortage go back decades". The Conversation. Archived from the original on February 22, 2021. Retrieved February 22, 2021.
- ^ Lucas, Paul (January 22, 2021). "Why we're not doing better on COVID vaccines". Financial Post via the National Post. Opinion. Archived from the original on February 9, 2021.
- ^ Kuchler, Hannah; Abboud, Leila (February 15, 2021). "Why the three biggest vaccine makers failed on Covid-19". Financial Post. Archived from the original on February 21, 2021. Retrieved February 22, 2021.
- ^ a b c d e Ling, Justin (May 31, 2021). "Where did Canada's vaccine effort actually go wrong?". Maclean's. Archived from the original on May 31, 2021. Retrieved August 27, 2021.
- ^ a b c Pinkerton, Charlie (January 26, 2021). "Days after announcing deal, Ottawa learned China blocked CanSino's vaccine shipment". iPolitics. Archived from the original on January 26, 2021. Retrieved August 27, 2021.
- ^ Hopper, Tristin (February 5, 2021). "How Ottawa utterly botched Canada's COVID vaccine acquisition". National Post. Retrieved August 27, 2021.
- ^ "Planning ahead: U.S., Canada order 177M more COVID-19 vaccine syringes from BD". FierceBiotech. July 22, 2020. Archived from the original on February 2, 2021. Retrieved February 16, 2021.
- ^ a b Canada, National Research Council (September 18, 2020). "COVID-19 response: Building the infrastructure". Archived from the original on February 2, 2021. Retrieved February 3, 2021.
- ^ Harris, Kathleen (September 25, 2020). "With no successful vaccine candidates yet, Canada signs deal to secure 20M more COVID-19 vaccine doses". CBC News. Archived from the original on March 20, 2021. Retrieved December 15, 2020.
- ^ Health Canada (October 2, 2020). "Health Canada begins first authorization review of a COVID-19 vaccine submission". gcnws. Archived from the original on March 30, 2021. Retrieved December 30, 2020.
- ^ Blanchfield, Mike (November 29, 2020). "PM put all vaccine 'eggs' in one basket, didn't consider other options: O'Toole". CTVNews. Retrieved August 27, 2021.
- ^ "Pfizer and BioNTech to submit request for emergency use approval of their COVID-19 vaccine today". November 20, 2020.[permanent dead link]
- ^ Government of Canada, Health Canada (December 12, 2020). "Authorization of Pfizer-BioNTech COVID-19 Vaccine with English-only Carton and Vial Labels". healthycanadians.gc.ca. Retrieved September 16, 2021.
- ^ Tasker, John Paul (December 9, 2020). "Health Canada approves Pfizer COVID-19 vaccine". CBC News. Archived from the original on January 14, 2021. Retrieved December 9, 2020.
- ^ Tasker, John Paul (November 9, 2020). "Trudeau says promising new Pfizer vaccine could be 'light at the end of the tunnel'". CBC News. Archived from the original on November 9, 2020. Retrieved November 9, 2020.
- ^ Jones, Alexandra Mae (December 10, 2020). "Canada to implement its first national vaccine injury compensation program". CTV News. Archived from the original on December 11, 2020. Retrieved January 17, 2024.
- ^ a b Bilefsky, Dan; Austen, Ian (December 14, 2020). "Canada and the Coronavirus Vaccine: Here's What It Means". The New York Times. Archived from the original on December 15, 2020. Retrieved December 15, 2020.
- ^ "Here's the COVID-19 vaccine rollout plan, province by province". CBC News. December 10, 2020. Archived from the original on December 15, 2020. Retrieved December 15, 2020.
- ^ "When will Canadians get COVID-19 vaccines? The federal and provincial rollout plans so far". The Globe and Mail. December 10, 2020. Archived from the original on December 14, 2020. Retrieved December 15, 2020.
- ^ Jones, Ryan Patrick (December 23, 2020). "Health Canada approves Moderna COVID-19 vaccine". CBC News. Archived from the original on February 18, 2021. Retrieved February 18, 2021.
- ^ a b Aiello, Rachel (December 15, 2020). "Canada locks in thousands more early COVID-19 vaccine doses". CTVNews. Archived from the original on December 16, 2020. Retrieved December 16, 2020.
- ^ Hinchey, Garrett (December 28, 2020). "First doses of Moderna COVID-19 vaccine arrive in Yukon, N.W.T." CBC News. Archived from the original on February 17, 2021. Retrieved February 18, 2021.
- ^ Boynton, Sean (December 28, 2020). "First shipments of Moderna coronavirus vaccine arrive in Yukon, N.W.T." Global News. Archived from the original on February 2, 2021. Retrieved February 16, 2021.
- ^ a b Rabson, Mia (February 2, 2021). "Canada signs deal to produce Novavax COVID-19 vaccine at Montreal plant". CP24. Archived from the original on April 28, 2021. Retrieved February 3, 2021.
- ^ "Canada signs first deal for manufacture of foreign COVID-19 vaccine". Reuters. February 2, 2021. Archived from the original on February 2, 2021. Retrieved February 3, 2021.
- ^ a b Office of the Prime Minister (February 2, 2021). "New support to produce COVID-19 vaccines and treatments in Canada" (Press release). Archived from the original on February 3, 2021. Retrieved February 3, 2021.
- ^ "COVID-19 response: Vaccines and therapeutics". National Research Council Canada. February 2021. Archived from the original on March 18, 2021. Retrieved March 22, 2021.
- ^ "Understanding Canada's Vaccine Injury Support Program: Public Health Agency of Canada Vaccine Confidence Webinar Series". Government of Canada. August 30, 2022. Archived from the original on June 6, 2023. Retrieved January 17, 2024.
- ^ Ljunggren, David; Martell, Allison (January 28, 2021). "Canada Expects Further Pfizer Vaccine Delay Prompting Protests". The Toronto Sun. Reuters. Archived from the original on February 13, 2021. Retrieved February 16, 2021.
- ^ Tasker, John Paul (January 29, 2021). "Moderna Cut Deliveries to Canada". CBC News. Archived from the original on February 2, 2021. Retrieved February 3, 2021.
- ^ Cousins, Ben (February 3, 2021). "Europe approves vaccine delivery to Canada". CTV News. Archived from the original on February 3, 2021. Retrieved February 3, 2021.
- ^ Austen, Ian (February 18, 2021). "Delays Turn Canada's Vaccination Optimism Into Anxiety". The New York Times. p. A6. Archived from the original on February 22, 2021. Retrieved February 22, 2021.
- ^ a b Austen, Ian (February 20, 2021). "Why Canada's Vaccine Rollout Slowed Down". The New York Times. ISSN 0362-4331. Archived from the original on March 11, 2021. Retrieved March 22, 2021.
- ^ Aiello, Rachel (February 4, 2021). "Canada to keep feeling impacts of COVID-19 vaccine delivery issues: Fortin". CTV News. Archived from the original on February 4, 2021. Retrieved February 4, 2021.
- ^ Jerving, Sara (February 3, 2021). "COVAX releases country-by-country of vaccine distribution figures". Devex. Archived from the original on August 24, 2021. Retrieved February 3, 2021.
- ^ "The Covax Facility: Interim Distribution Forecast – latest as of February 3, 2021" (PDF). COVAX. February 3, 2021. Archived (PDF) from the original on February 18, 2021. Retrieved February 16, 2021.
- ^ a b "Canada negotiates accelerated delivery schedule for Pfizer-BioNTech's COVID-19 vaccine". Public Services and Procurement Canada (Press release). Gatineau, Quebec. February 12, 2021. Archived from the original on February 18, 2021. Retrieved February 18, 2021.
- ^ a b Jones, Ryan Patrick (February 18, 2021). "COVID-19 vaccine deliveries back on track following weeks of delay, says Public Health Agency". CBC News. Archived from the original on March 28, 2021. Retrieved February 18, 2021.
- ^ "COVID-19 Tracker Canada". Vaccination Tracker. December 3, 2020. Archived from the original on February 18, 2021. Retrieved February 18, 2021.
- ^ Austen, Ian (February 18, 2021). "Delays Turn Canada's Vaccination Optimism Into Anxiety". The New York Times. p. A6. Archived from the original on February 21, 2021. Retrieved February 22, 2021.
- ^ a b Forani, Jonathan (February 26, 2021). "Health Canada approves two AstraZeneca COVID-19 vaccines". CTV News. Archived from the original on May 14, 2021. Retrieved February 26, 2021.
- ^ "Canada receives 1st AstraZeneca vaccine shipment as confusion lingers". Global News. The Canadian Press. March 3, 2021. Archived from the original on March 9, 2021. Retrieved March 22, 2021.
- ^ D'Mello, Colin (March 2, 2021). "Ontario won't administer AstraZeneca COVID-19 vaccine to seniors, final plan still in the works". CTV News. Archived from the original on March 3, 2021. Retrieved March 4, 2021.
- ^ Jones, Ryan Patrick (March 5, 2021). "Johnson & Johnson COVID-19 vaccine becomes 4th to receive Health Canada approval". CBC News. Archived from the original on March 19, 2021. Retrieved March 5, 2021.
- ^ Aiello, Rachel (March 5, 2021). "Canada authorizes one-shot COVID-19 vaccine from Johnson & Johnson". CTV News. Archived from the original on March 11, 2021. Retrieved March 22, 2021.
- ^ Sherriff-Scott, Iain (March 5, 2021). "Johnson & Johnson vaccine approved by Health Canada, Pfizer deliveries accelerated". iPolitics. Archived from the original on March 17, 2021. Retrieved March 22, 2021.
- ^ Turnbull, Sarah (March 5, 2021). "Coronavirus: 8M vaccine doses to land in Canada by end of March after Pfizer moves up delivery". CTV News. Archived from the original on March 14, 2021. Retrieved March 22, 2021.
- ^ Aiello, Rachel (March 3, 2021). "Second doses of COVID-19 vaccines can be given up to 4 months after first, NACI now says". CTV News. Archived from the original on May 26, 2021. Retrieved June 2, 2021.
- ^ Jones, Ryan Patrick (March 26, 2021). "1.5 million doses of AstraZeneca vaccine arriving from U.S. on Tuesday, Anand says". CBC News. Archived from the original on March 28, 2021. Retrieved March 28, 2021.
- ^ Anita Anand (March 26, 2021). 10 million shots of the Johnson & Johnson vaccine. Global News.
- ^ Nassar, Hana Mae (April 16, 2021). "Canada secures deal with Pfizer to increase vaccine deliveries". CityNews. Archived from the original on April 28, 2021. Retrieved April 28, 2021.
- ^ "Federal government expecting 1M vaccine doses this week after Moderna delays delivery". CTV News. The Canadian Press. April 19, 2021. Archived from the original on April 28, 2021. Retrieved April 28, 2021.
- ^ "Vaccine hunting? These volunteers want to help; Rogers blames update for outage: CBC's Marketplace Cheat Sheet". CBC News. April 25, 2021. Archived from the original on April 28, 2021. Retrieved August 24, 2021.
- ^ "Having trouble booking your shot? These volunteer 'vaccine hunters' want to help". CBC News. April 20, 2021. Archived from the original on April 26, 2021. Retrieved August 24, 2021.
- ^ Treble, Patricia (April 15, 2021). "Q&A with Vaccine Hunters Canada: 'We're just here to get things done'". Maclean's. Archived from the original on April 28, 2021. Retrieved April 28, 2021.
- ^ Pope, Samantha (April 21, 2021). "'They're heroes': How COVID-19 'vaccine hunters' help Canadians find appointments". National Post. Archived from the original on August 24, 2021. Retrieved April 28, 2021.
- ^ Herhalt, Chris (April 28, 2021). "Toronto partnering with Vaccine Hunters Canada to connect residents with COVID-19 shots". CP24. Archived from the original on April 28, 2021. Retrieved April 28, 2021.
- ^ Robertson, Becky. "Toronto officially partners with crowdsourcing effort Vaccine Hunters Canada". www.blogto.com. Archived from the original on April 28, 2021. Retrieved April 28, 2021.
- ^ Passifiume, Bryan (April 28, 2021). "'ONE MORE CHANNEL': Toronto to provide info to Vaccine Hunters Canada". Toronto Sun. Archived from the original on April 28, 2021. Retrieved April 28, 2021.
- ^ "First J&J COVID-19 vaccines to arrive in Canada Wednesday". CTV News. The Canadian Press. April 28, 2021. Archived from the original on April 28, 2021. Retrieved April 28, 2021.
- ^ Somos, Christy (May 1, 2021). "What you need to know before the J&J vaccines are distributed in Canada". CTV News. Archived from the original on May 3, 2021. Retrieved May 4, 2021.
- ^ Neustaeter, Brooklyn (April 30, 2021). "Health Canada holding J&J COVID-19 vaccines over possible quality control issue". CTV News. Archived from the original on May 4, 2021. Retrieved May 4, 2021.
- ^ Aiello, Rachel (May 4, 2021). "'Get your shot as soon as it's your turn': PM seeks to dispel vaccine concerns". CTV News. Archived from the original on May 4, 2021. Retrieved May 4, 2021.
- ^ Dunham, Jackie (May 4, 2021). "NACI accused of contributing to confusion, hesitancy over 'preferred' vaccines guidance". CTV News. Archived from the original on May 4, 2021. Retrieved May 4, 2021.
- ^ Bensadoun, Emerald (May 3, 2021). "NACI recommends Johnson & Johnson COVID-19 shot for adults 30 and up, says mRNA preferred". Global News. Archived from the original on May 4, 2021. Retrieved May 4, 2021.
- ^ Aiello, Rachel (May 12, 2021). "PM Trudeau says he plans to take second AstraZeneca shot, if it's available". CTV News. Archived from the original on May 13, 2021. Retrieved May 13, 2021.
- ^ Thompson, Nicole (May 12, 2021). "More provinces are limiting the use of the AstraZeneca vaccine". CTV News. Canadian Press. Archived from the original on May 13, 2021. Retrieved May 13, 2021.
- ^ Draaisma, Muriel (May 11, 2021). "Ontario will no longer give AstraZeneca COVID-19 vaccine as 1st dose due to blood clot risk". CBC News. Archived from the original on May 12, 2021. Retrieved May 12, 2021.
- ^ Tasker, John Paul (May 14, 2021). "Partially vaccinated Canadians can socialize outdoors this summer, Tam says". CBC News. Archived from the original on May 16, 2021. Retrieved August 24, 2021.
- ^ "Vaccination error: A dozen youth mistakenly given the wrong COVID-19 shot in Vancouver". CTV News. Vancouver. The Canadian Press. May 31, 2021. Retrieved August 25, 2021.
- ^ Jackson, Hannah (May 22, 2021). "50% of Canadians have now received at least 1 COVID-19 vaccine dose". Global News. Archived from the original on May 25, 2021. Retrieved June 4, 2021.
- ^ Paling, Emma (May 21, 2021). "Canada's percentage of people with 1 COVID-19 vaccine dose now higher than U.S." CBC News. Archived from the original on May 24, 2021. Retrieved August 24, 2021.
- ^ "Ontarians who received 1st AstraZeneca vaccine mid-March can book 2nd shot". Global News. The Canadian Press. May 25, 2021. Archived from the original on May 25, 2021. Retrieved May 25, 2021.
- ^ "B.C. residents who had AstraZeneca vaccine can choose second dose vaccine". CHEK. May 18, 2021. Archived from the original on May 25, 2021. Retrieved May 25, 2021.
- ^ Miller, Adam (May 29, 2021). "Why experts say 2nd vaccine doses should be rolled out now". CBC News. Archived from the original on June 5, 2021. Retrieved June 4, 2021.
- ^ Woo, Andrea; Stone, Laura; Walsh, Marieke (May 25, 2021). "Provinces move to shorten interval between COVID-19 vaccine doses". The Globe and Mail. Archived from the original on June 5, 2021. Retrieved June 4, 2021.
- ^ Canada, Public Health Agency of (May 28, 2021). "NACI updated COVID-19 vaccine statement, May 28, 2021: Summary". Canada.ca. NACI. Archived from the original on June 4, 2021. Retrieved June 4, 2021.
- ^ Ghosh, Twinkle (May 28, 2021). "2nd COVID-19 shots should be offered 'as soon as possible': NACI". Global News. Archived from the original on June 4, 2021. Retrieved June 4, 2021.
- ^ "Canada expects slower week of vaccine shipments with only 600K from Pfizer". Global News. The Canadian Press. May 24, 2021. Archived from the original on May 25, 2021. Retrieved May 25, 2021.
- ^ van Koeverden, Adam (April 19, 2023). Inquiry of Ministry Q-1416 - Vaccine Injury Support Program. Government of Canada.
- ^ Tumilty, Ryan (June 18, 2021). "Canada expects to have 68 million COVID-19 vaccine doses by end of July". National Post. Archived from the original on July 5, 2021. Retrieved June 19, 2021.
- ^ Tasker, John Paul (August 13, 2021). "Federal government to require vaccinations for all federal public servants, air and train passengers". CBC News. Archived from the original on August 13, 2021. Retrieved August 13, 2021.
- ^ Turnbull, Sarah (August 13, 2021). "Feds to make COVID-19 vaccines mandatory across federal public service". CTV News. Archived from the original on August 20, 2021. Retrieved August 13, 2021.
- ^ Treasury Board of Canada Secretariat (August 13, 2021). "Government of Canada to require vaccination of federal workforce and federally regulated transportation sector". Government of Canada (Press release). Retrieved August 24, 2021.
- ^ "Ontario Makes COVID-19 Vaccination Policies Mandatory for High-Risk Settings". Government of Ontario (Press release). Archived from the original on August 23, 2021. Retrieved August 23, 2021.
- ^ "Ontario announces mandatory vaccine plans for health, education workers; 3rd doses for some". CBC News. August 17, 2021. Archived from the original on August 23, 2021. Retrieved August 23, 2021.
- ^ "Mandatory vaccination could be extended beyond health-care workers in Quebec". CTV News. The Canadian Press. August 19, 2021. Archived from the original on August 22, 2021. Retrieved August 23, 2021.
- ^ Allard, Catherine (August 18, 2021). "Le Nouveau-Brunswick rendra la vaccination obligatoire pour tous ses fonctionnaires" [New Brunswick to make vaccination mandatory for all public servants] (in French). Radio-Canada. Archived from the original on August 23, 2021. Retrieved August 23, 2021.
- ^ Sturgeon, Nathalie (August 18, 2021). "N.B. premier mandates vaccines for public sector employees". Global News. Retrieved August 24, 2021.
- ^ "Health Canada authorizes Moderna COVID-19 vaccine for use in youth 12-17". CP24. August 27, 2021.
- ^ Gilmore, Rachel (September 16, 2021). "Pfizer, Moderna COVID-19 vaccines get full Health Canada approval — and new names". Global News. Archived from the original on September 16, 2021. Retrieved September 16, 2021.
- ^ Osman, Laura (October 18, 2021). "Pfizer officially requests Health Canada approval for kids' COVID-19 shot". CTV News.
- ^ Major, Darren (October 21, 2021). "Canada to receive 2.9M Pfizer doses to begin vaccinating 5 to 11 year-olds once approved, Trudeau says". CBC News. Retrieved November 4, 2021.
- ^ Ireland, Nicole (October 29, 2021). "Seniors 70 and older, front-line health workers, people who got 2 AZ doses can be offered boosters, NACI says". CBC News.
- ^ Tunney, Catharine (November 19, 2021). "COVID-19 vaccines for children will start arriving Sunday, government says". CBC News. Retrieved November 22, 2021.
- ^ a b c d e f g h i j k l m n "COVID-19 Tracker Canada - Vaccination Tracker". covid19tracker.ca.
- ^ Osman, Laura (November 19, 2021). "Health Canada approves Pfizer COVID-19 vaccine for kids 5-11". CP24.
- ^ Paas-Lang, Christian (November 21, 2021). "First COVID-19 vaccines for children arrive in Canada". CBC News. Retrieved November 22, 2021.
- ^ "NACI reviewing COVID-19 booster shot program with arrival of Omicron variant". Global News. November 30, 2021.
- ^ Aiello, Rachel (December 1, 2021). "Canada will have vaccine booster supply if NACI recommends wider usage: PM". CTV News.
- ^ "Alberta expands COVID-19 vaccine booster eligibility to all adults in stages". CTV News. Edmonton. December 1, 2021.
- ^ Murphy, Jessica (January 29, 2022). "Freedom Convoy: Why Canadian truckers are protesting in Ottawa". BBC News.
- ^ Pelley, Lauren; Miller, Adam (April 5, 2022). "Canada's vaccine advisory body recommends 'rapid deployment' of 4th doses for 80+". CBC News.
- ^ Boisvert, Nick (July 14, 2022). "Health Canada approves 1st COVID-19 vaccine for youngest kids". CBC News.
- ^ "Regulatory Decision Summary - Spikevax - Health Canada". Health Canada. Archived from the original on July 16, 2022. Retrieved July 16, 2022.
- ^ "Health Canada authorizes first bivalent COVID-19 booster for adults 18 years and older". Health Canada. Government of Canada. September 2022. Archived from the original on September 9, 2022.
- ^ "Quebec rolling out new bivalent COVID-19 vaccine against Omicron". CBC News. September 7, 2022. Retrieved September 9, 2022.
- ^ "Ontario receives first doses of bivalent vaccine: health minister". CTV News. September 8, 2022. Retrieved September 10, 2022.
- ^ Dubois, Stephanie (September 10, 2022). "Bivalent COVID vaccines are here. What happens to the existing vaccines?". CBC News. Retrieved September 10, 2022.
- ^ Zimonjic, Peter; Miller, Adam (October 7, 2022). "Health Canada approves Pfizer's new bivalent COVID-19 vaccine". CBC News.
- ^ "Health Canada approves first bivalent booster for kids aged 5 to 11". CBC News. December 9, 2022.
- ^ Health Canada (September 12, 2023). "Health Canada authorizes Moderna COVID-19 vaccine targeting the Omicron XBB.1.5 subvariant". Government of Canada. Archived from the original on September 15, 2023. Retrieved September 15, 2023.
- ^ Priddy, Frances (September 12, 2023). "Safety and Immunogenicity of Moderna COVID-19 Vaccine (2023-2024 Formula) - Monovalent XBB.1.5 Variant Vaccine" (PDF). Centers for Disease Control and Prevention. Archived (PDF) from the original on September 15, 2023. Retrieved September 15, 2023.
- ^ Winter, Emery; Loe, Megan (September 14, 2023). "Florida Surgeon General's claim that updated COVID vaccines weren't clinically tested, including on humans, is misleading". VERIFY. Archived from the original on September 15, 2023. Retrieved September 15, 2023.
- ^ Judd, Amy (September 12, 2023). "Get ready for significant COVID, flu fall vaccine campaign, B.C. government says". Global News. Archived from the original on September 13, 2023. Retrieved September 15, 2023.
- ^ Health Canada (September 12, 2023). "Moderna Spikevax COVID-19 vaccine". Government of Canada. Archived from the original on September 15, 2023. Retrieved September 15, 2023.
- ^ https://covid-19-newfoundland-and-labrador-gnl.hub.arcgis.com/
- ^ Public Health Agency of Canada (December 3, 2020). "Vaccines and treatments for COVID-19: Vaccine rollout". Government of Canada. Archived from the original on February 16, 2021. Retrieved February 16, 2021.
- ^ a b Alonso, Melissa (October 6, 2021). "Canada issues Covid-19 vaccine mandate for travelers 12 or older on trains and planes". CNN. Retrieved October 7, 2021.
- ^ "COVID-19 Boarding flights and trains in Canada – Travel.gc.ca". December 21, 2021.
- ^ Gilmore, Rachel (January 20, 2022). "As unvaccinated workers sue for wrongful dismissal, Ottawa working on shielding employers". Global News.
- ^ Rahman, Bhromor (October 31, 2021). "Le passeport vaccinal c. la Charte". Le Flagrant Délit (in French).
- ^ Lao, David (August 25, 2021). "Air Canada requiring all employees, new hires to be vaccinated against COVID-19". Global News.
- ^ Kerr, Jaren (August 31, 2021). "Most major accounting firms in Canada to require vaccination for workers in offices". The Globe and Mail.
- ^ Lowe, Julie; Trosow, Samuel E. (August 25, 2021). "Canadian colleges and universities can mandate COVID-19 vaccination without violating Charter rights". The Conversation.
- ^ "Canadian private long-term care providers announce COVID-19 vaccine mandate for staff". CBC News. The Canadian Press. August 26, 2021. Retrieved November 17, 2021.
- ^ Miller, Adam (August 14, 2021). "COVID-19 vaccine mandates are coming — whether Canadians want them or not". CBC News. Retrieved August 28, 2021.
- ^ "B.C. launches proof of vaccination to stop spread of COVID-19" (Press release). Government of British Columbia. August 23, 2021. Retrieved August 29, 2021.
- ^ Roffel, Brittany (October 5, 2021). "B.C. mandates COVID-19 vaccines for public service workers and long-term care visitors". CBC News. Retrieved November 17, 2021.
- ^ Kost, Hannah (September 16, 2021). "What we know about Alberta's new vaccine passport (that's not being called a passport) so far". CBC News. Retrieved November 17, 2021.
- ^ "Alberta Health Services makes COVID-19 vaccine mandatory for all staff". CTV News. Edmonton. August 31, 2021.
- ^ White, Ryan (September 13, 2021). "Alberta universities to mandate vaccinations for students, staff and visitors attending campus". CTV News. Calgary.
- ^ Kury de Castillo, Carolyn (September 21, 2021). "Harassment over COVID-19 restrictions prompts Alberta restaurant to pause indoor dining". Global News.
- ^ "Alberta orders public service employees to be vaccinated". www.hcamag.com.
- ^ "Vaccine mandate ended for Alberta public servants, premier says". CBC News. March 3, 2022. Archived from the original on February 28, 2023.
- ^ "Unvaccinated health-care workers called back to work after AHS COVID vaccine mandate lifted". CBC News. Calgary. March 15, 2022.
- ^ Ghania, Yasmine (September 28, 2021). "Sask.'s proof-of-vaccination policy is now in effect. Here's what you need to know". CBC News. Retrieved November 17, 2021.
- ^ "Vaccinations - COVID-19 | Return to Campus - University of Saskatchewan". covid19.usask.ca. Archived from the original on March 30, 2022. Retrieved September 1, 2021.
- ^ "Mandatory masking, vaccine passports coming to Saskatchewan". 650 CKOM. September 16, 2021.
- ^ "Province Implements Interim Mandatory Masking Effective September 17, Proof of Vaccination Requirement Effective October 1". Government of Saskatchewan (Press release). September 16, 2021. Retrieved November 17, 2021.
- ^ Quon, Alexander (February 8, 2022). "Sask. to end COVID-19 proof of vaccination policy on Feb. 14, mandatory masking to remain until end of month". CBC News.
- ^ "Sask. Ends vaccine mandates for tens of thousands of public servants".
- ^ a b "Manitoba Will Require COVID-19 Vaccinations or Testing for Designated Government Employees". Province of Manitoba (Press release). August 24, 2021.
- ^ Gowriluk, Caitlyn (August 19, 2021). "Manitoba's 2 largest universities, several other schools to require staff and students to be vaccinated". CBC News. Retrieved November 17, 2021.
- ^ "Manitoba Expands Vaccine Passports for Fully Immunized Individuals as of Sept. 3" (Press release). Government of Manitoba. August 27, 2021. Retrieved August 28, 2021.
- ^ "COVID-19 vaccine cards no longer required starting Tuesday at most places in Manitoba". CBC News. February 28, 2022.
- ^ Weeks, Carly (November 5, 2021). "Many Ontario hospitals instituting COVID-19 vaccine requirements in absence of Ford provincial mandate". Globe and Mail.
- ^ "Ontario Makes COVID-19 Vaccination Policies Mandatory for High-Risk Settings" (Press release). Government of Ontario. August 17, 2021. Retrieved August 28, 2021.
- ^ Freeman, Joshua (August 19, 2021). "Ontario says all 64K members of the public service must be vaccinated or take regular COVID-19 tests". CTV News. Retrieved August 28, 2021.
- ^ Broad, Paul E.; Eves, Sarah A. (August 17, 2021). "Ontario to Implement Mandatory Vaccinations Policies for Hospitals, Schools, Post-Secondary Institutions, and More". Hicks Morley. Retrieved August 28, 2021.
- ^ Katawazi, Miriam (September 1, 2021). "Ontario reveals vaccine passport system. Here's what you need to know". CTV News. Toronto. Retrieved September 1, 2021.
- ^ Blewett, Taylor (March 1, 2022). "COVID-19: Ontario ends vaccine passport system, indoor capacity limits; 50 new cases in Ottawa". Ottawa Citizen.
- ^ a b "Quebec backs down again on mandatory vaccination in health network". CBC News. November 3, 2021.
- ^ "COVID-19 vaccination passport". Government of Quebec. Retrieved August 28, 2021.
- ^ Lofaro, Joe (October 13, 2021). "Quebec pushes back vaccine deadline to Nov. 15 for health-care workers". CTV News. Montreal.
- ^ MacKinnon, Bobbi-Jean (August 31, 2021). "COVID-19 vaccine mandatory for New Brunswick health-care workers Sept. 7, memo says". CBC News. Retrieved November 17, 2021.
- ^ Bissett, Kevin (August 27, 2021). "N.B. civil servants must get vaccine by Sept. 13 or submit to testing 3 times a week". CTV News. The Canadian Press. Retrieved August 28, 2021.
- ^ "COVID-19 community update". University of New Brunswick (Press release). August 20, 2021.
- ^ MacKinnon, Bobbi-Jean (September 15, 2021). "N.B. COVID-19 roundup: Vaccination proof to be required after record 63 cases, 1 death recorded". CBC News. Retrieved September 16, 2021.
- ^ "Provincial COVID-19 restrictions to be lifted March 14" (Press release). Government of New Brunswick. February 24, 2022. Archived from the original on March 4, 2022. Retrieved March 4, 2022.
- ^ Toolkit, Web Experience (September 28, 2021). "Health PEI Health Care Workers: COVID-19 FAQs". www.princeedwardisland.ca. Archived from the original on November 8, 2021. Retrieved November 8, 2021.
- ^ Chang, Arturo (August 25, 2021). "Holland College will require all students, staff to be vaccinated". CBC News. Retrieved August 28, 2021.
- ^ Bissett, Kevin (September 21, 2021). "Prince Edward Island to impose COVID-19 vaccine passport starting Oct. 5". Global News.
- ^ "PEI Vax Pass Program - Discontinued" (Press release). Government of Prince Edward Island. February 28, 2022. Archived from the original on March 4, 2022. Retrieved March 4, 2022.
- ^ Long, Samantha (September 29, 2021). "Nova Scotia to take 'cautious approach' in final phase of reopening; mandates vaccines for health-care workers, teachers". CTV News. Atlantic.
- ^ "Our plan for fall 2021". Dalhousie University. Archived from the original on September 1, 2021. Retrieved September 1, 2021.
- ^ April, Allan (September 8, 2021). "Nova Scotia to move into Phase 5 on Sept. 15, announces proof of vaccination policy". CTV News.
- ^ "7 COVID-19 deaths reported in N.S. as most vaccine passport restrictions lifted". CBC News. February 28, 2022.
- ^ Roberts, Darrell (September 29, 2021). "Vaccine mandate coming for N.L. public sector workers, says premier". CBC News. Retrieved November 17, 2021.
- ^ Strzelczyk, Florentine (August 20, 2021). "Mandatory mask and vaccination requirements". Memorial University Gazette.
- ^ "Vaccine passports coming, Furey says, as N.L. reports 5 new cases". CBC News. September 7, 2021. Retrieved November 17, 2021.
- ^ "N.L. government reveals draft vaccine passport plan to St. John's business community". CBC News. September 30, 2021. Retrieved November 17, 2021.
- ^ "N.L. vaccine passports mandatory by Oct. 22, but churches can choose masking option". CTV News Atlantic. The Canadian Press. October 7, 2021.
- ^ "Mandatory Vaccination Policy Details" (Press release). Government of Newfoundland and Labrador. October 15, 2021.
- ^ "Public Advisory: Update on COVID-19 in Newfoundland and Labrador" (Press release). Health and Community Services. March 11, 2022. Retrieved March 16, 2022.
- ^ "Yukon announces mandatory COVID-19 vaccine for wide range of workers". CTV News. The Canadian Press. October 15, 2021. Archived from the original on October 15, 2021.
- ^ Proulx, Michel (October 15, 2021). "Yukon premier announces vaccine mandate, passport program starting Nov. 30". CBC News. Archived from the original on October 15, 2021.
- ^ "COVID-19 public health measures continue to ease" (Press release). Yukon Government. March 2, 2022.
- ^ a b "N.W.T. voluntary vaccine passport program set to launch Oct. 22, as COVID-19 cases trend downward". CBC News. October 14, 2021. Retrieved November 17, 2021.
- ^ "Northwest Territories govt orders staff to be fully vaccinated by Nov 30". www.hcamag.com.
- ^ "GNWT eases pandemic restrictions as it moves towards ending the Public Health Emergency" (Press release). Government of Northwest Territories. February 25, 2022. Retrieved March 7, 2022.
- ^ "Nunavut implementing COVID-19 vaccination requirement for many health workers". CBC News. November 30, 2021.
- ^ "Canadian report on COVID-19 vaccine doses distributed". Public Health Agency of Canada. July 14, 2023.
- ^ a b c Public Services and Procurement Canada (December 11, 2020). "Procuring vaccines for COVID-19". Government of Canada. Archived from the original on February 17, 2021. Retrieved February 16, 2021.
- ^ Gordon, Julie (December 4, 2020). "Canada doubles Moderna vaccine order, daily COVID-19 cases could top 10,000 by January". Reuters. Archived from the original on February 2, 2021. Retrieved February 16, 2021.
- ^ George-Cosh, David (November 10, 2020). "Canada has option for 56M more Pfizer vaccine doses: Anand - BNN Bloomberg". BNN. Archived from the original on February 28, 2021. Retrieved February 16, 2021.
- ^ "Coronavirus vaccine tracker: How many people in Canada have received shots?". CTV News. December 29, 2020. Archived from the original on March 22, 2021. Retrieved March 22, 2021.
- ^ Turnbull, Sarah (June 4, 2021). "Feds confirm 9M Pfizer doses arriving in August, option for 3M in September". CTV News. Retrieved August 24, 2021.
- ^ Jones, Ryan Patrick (January 12, 2021). "Government secures another 20M COVID-19 vaccine doses from Pfizer". CBC News. Retrieved March 22, 2021.
- ^ "Procuring vaccines for COVID-19". Government of Canada. Retrieved August 24, 2021.
- ^ Rabson, Mia (December 9, 2020). "Health Canada approves Pfizer's COVID-19 vaccine". The Star. The Canadian Press.
- ^ Moderna (February 12, 2021). "Canada Purchases Additional 4 Million Doses of Moderna's COVID-19 Vaccine" (Press release) – via Business Wire.
- ^ "Shipment of Moderna COVID-19 vaccine arrives in Canada one day after approval". CBC News. December 24, 2020. Retrieved February 18, 2021.
- ^ Forani, Jonathan (February 26, 2021). "Health Canada approves two AstraZeneca COVID-19 vaccines". CTV News.
- ^ Tasker, John Paul (February 26, 2021). "Health Canada approves AstraZeneca's COVID-19 vaccine". CBC News. Retrieved March 22, 2021.
- ^ Loblaw Companies Limited (March 10, 2021). "Shoppers Drug Mart marks historic COVID-19 milestone with first vaccine in pharmacy in Ontario" (Press release). Retrieved March 22, 2021 – via Newswire.
- ^ Zimonjic, Peter (September 17, 2020). "Drug and vaccine authorizations for COVID-19: List of authorized drugs, vaccines and expanded indications March 3, 2021". Government of Canada. Retrieved March 22, 2021.
- ^ Bourne, Kirby (November 12, 2021). "Alberta receives 5K doses of Janssen COVID-19 vaccine, confirms 841 new cases over 2 days". Global News. Retrieved November 12, 2021.
- ^ "Johnson & Johnson COVID-19 Vaccine Fully Approved by Health Canada to Prevent COVID-19 in Individuals 18 years and Older" (PDF). November 24, 2021. Retrieved November 28, 2021.
- ^ "Novavax's COVID-19 vaccine approved for Canadians 18 and over". CBC News. February 17, 2022.
- ^ https://regina.ctvnews.ca/novavax-vaccine-available-in-sask-monday-1.5854547
- ^ Lofaro, Joe (April 13, 2022). "First batch of Novavax COVID-19 vaccines now available in Quebec at walk-in clinics". CTV News.
- ^ Medicago GSK (March 16, 2021). "Medicago and GSK start Phase 3 trial of adjuvanted COVID-19 vaccine candidate" (Press release). Retrieved March 22, 2021.
- ^ https://covid-vaccine.canada.ca/covifenz/product-details
- ^ "Medicago Covifenz COVID-19 vaccine". Health Canada. February 24, 2022. Retrieved October 19, 2023.
- ^ Lao, David; Bimman, Abigail (December 24, 2020). "Should provinces reserve COVID-19 vaccine 2nd doses or administer them all right away?". Global News. Archived from the original on January 20, 2021. Retrieved March 4, 2021.
- ^ "Pfizer-BioNTech COVID-19 Vaccine Information". United States Centres for Disease Control and Prevention. February 25, 2021. Archived from the original on June 9, 2021. Retrieved March 4, 2021.
- ^ "Information about the Moderna COVID-19 Vaccine". United States Centres for Disease Control and Prevention. January 25, 2021. Archived from the original on June 11, 2021. Retrieved March 4, 2021.
- ^ Lauerman, John; Gale, Jason (January 26, 2021). "What to know about delaying second COVID vaccine shots as countries grapple with limited supplies". Fortune. Archived from the original on March 3, 2021. Retrieved March 4, 2021.
- ^ Plotkin, Stanley A.; Halsey, Neal (January 27, 2021). "Accelerate COVID-19 Vaccine Rollout by Delaying the Second Dose of mRNA Vaccines". Clinical Infectious Diseases. 73 (7): 1320–1321. doi:10.1093/cid/ciab068. PMC 7929065. PMID 33502467.
- ^ Epperly, David (January 5, 2021). "Evidence For COVID-19 Vaccine Deferred Dose 2 Boost Timing". SSRN. doi:10.2139/ssrn.3760833. S2CID 234325346.
- ^ Skowronski, Danuta M.; De Serres, Gaston (April 22, 2021). "Correspondence: Safety and Efficacy of the BNT162b2 mRNA Covid-19 Vaccine". New England Journal of Medicine (384): 1576–1578. doi:10.1056/NEJMc2036242. hdl:10757/655958. S2CID 231954071.
- ^ "More Evidence: Evidence For COVID-19 Vaccine Deferred Dose 2 Boost Timing". ReallyCorrect.com. Archived from the original on May 23, 2021. Retrieved March 9, 2021.
- ^ Serebrin, Jacob (December 31, 2020). "Quebec changes COVID-19 vaccine strategy, won't hold back doses for booster shots". CTV News. The Canadian Press. Archived from the original on January 27, 2021. Retrieved March 4, 2021.
- ^ "Quebec to speed up COVID-19 vaccination campaign by not holding 2nd doses in reserve". CBC News. December 31, 2020. Archived from the original on May 20, 2021. Retrieved March 22, 2021.
- ^ Banerjee, Sidhartha (February 12, 2021). "Health minister says Quebec remains on track to give vaccine booster within 90 days". National Post. The Canadian Press. Retrieved March 4, 2021.
- ^ Zimonjic, Peter (March 3, 2021). "Stretch interval between COVID-19 vaccine doses up to 4 months, national advisory committee recommends". CBC News. Archived from the original on June 10, 2021. Retrieved March 22, 2021.
- ^ a b Kane, Laura Dhillon (March 2, 2021). "B.C. defends plan to delay second dose as Ontario, Alberta consider following suit". CTV News. The Canadian Press. Archived from the original on March 23, 2021. Retrieved March 4, 2021.
- ^ "Drug and vaccine authorizations for COVID-19: List of authorized drugs, vaccines and expanded indications March 3, 2021". Government of Canada. September 17, 2020. Archived from the original on March 24, 2021. Retrieved March 22, 2021.
- ^ Banerjee, Sidhartha (March 25, 2021). "Quebec officials say those already infected with Covid-19 only need one vaccine-shot". National Post. The Canadian Press. Retrieved March 25, 2021.
- ^ "COVID-19 in Sask.: Second dose available to people 80 years and older starting Monday". CBC News. May 23, 2021. Archived from the original on May 24, 2021. Retrieved August 24, 2021.
- ^ Fox, Chris (May 25, 2021). "Solicitor general asks for patience as Ontarians hoping to book second dose of AstraZeneca vaccine run into issues". CP24. Archived from the original on June 26, 2021. Retrieved June 28, 2021.
- ^ "COVID-19 in Sask.: Province distributes last of vaccine stock, reports 260 new cases, 3 deaths". CBC News. January 24, 2021. Archived from the original on February 3, 2021. Retrieved March 22, 2021.
- ^ Slaughter, Graham (January 28, 2021). "Five doses per vial or six? Pfizer's expectations don't match Canada's reality, experts say". CTV News. Archived from the original on February 4, 2021. Retrieved March 4, 2021.
- ^ D'Amore, Rachael (February 10, 2021). "Canada to squeeze extra vaccine dose from Pfizer vials. It's not as easy as it seems". Global News. Archived from the original on May 11, 2021. Retrieved March 4, 2021.
- ^ "Administration of Pfizer-BioNTech COVID-19 Vaccine.pdf" (PDF). January 7, 2021. Archived (PDF) from the original on February 22, 2021. Retrieved March 22, 2021.
- ^ Ghosh, Twinkle (March 3, 2021). "Health Canada allows warmer storage conditions for Pfizer COVID-19 vaccine". Global News. Archived from the original on March 9, 2021. Retrieved March 4, 2021.
- ^ Aiello, Rachel (March 1, 2021). "AstraZeneca COVID-19 vaccine 'not recommended' in people 65 and older: NACI". CTV News. Archived from the original on March 4, 2021. Retrieved March 4, 2021.
- ^ a b "AstraZeneca-Oxford COVID-19 vaccine now recommended for those 65 and older in Canada". CBC News. March 16, 2021. Archived from the original on March 16, 2021. Retrieved March 16, 2021.
- ^ Lowrie, Morgan (March 9, 2021). "Quebec bucks advice from national vaccine panel, will give AstraZeneca to people 65 and up". CBC News. Archived from the original on March 11, 2021. Retrieved March 16, 2021.
- ^ Cousins, Ben (March 29, 2021). "Several provinces halt AstraZeneca vaccine for those under 55 in wake of new NACI guidelines". CTV News. Archived from the original on March 31, 2021. Retrieved April 1, 2021.
- ^ Abraham, Julian; April, Allan (March 29, 2021). "P.E.I. suspends use of AstraZeneca vaccine for those younger than 55". CTV News Atlantic. Archived from the original on April 9, 2021. Retrieved March 29, 2021.
- ^ Miller, Adam (March 29, 2021). "Why Canada is suspending use of AstraZeneca vaccine in people under 55". CBC News. Archived from the original on April 6, 2021. Retrieved April 7, 2021.
- ^ Perreaux, Les (April 20, 2021). "B.C., Alberta, Manitoba and Ontario lower age limits for AstraZeneca COVID-19 vaccine". The Globe & Mail. Archived from the original on April 20, 2021. Retrieved April 21, 2021.
- ^ Ross, Selena (April 20, 2021). "The AstraZeneca vaccine is now available to Quebecers age 45 and up". CTV News Montreal. Archived from the original on April 21, 2021. Retrieved April 21, 2021.
- ^ Blakley, Janelle (April 20, 2021). "Sask. lowers AstraZeneca age to 40; includes teachers, correctional officers in COVID-19 vaccine priority". CTV News Regina. Archived from the original on April 20, 2021. Retrieved April 21, 2021.
- ^ Aiello, Rachel (April 23, 2021). "Canadians aged 30 and older can be offered AstraZeneca vaccine, national vaccine panel says". CTV News. Archived from the original on April 23, 2021. Retrieved April 23, 2021.
- ^ DeClerq, Katherine (May 11, 2021). "Ontario will no longer offer first doses of AstraZeneca COVID-19 vaccine". CTV News. Archived from the original on May 12, 2021. Retrieved May 12, 2021.
- ^ Gilligan, Melissa; Pearson, Heide (May 11, 2021). "Alberta stops giving first doses of AstraZeneca vaccine due to supply issues". Global News. Archived from the original on May 12, 2021. Retrieved May 12, 2021.
- ^ Thompson, Nicole (May 12, 2021). "More provinces are limiting the use of the AstraZeneca vaccine". CTV News. Archived from the original on May 13, 2021. Retrieved May 13, 2021.
- ^ a b "Quebec halts 1st doses of AstraZeneca vaccine, keeps future supply for 2nd only". CBC News. May 13, 2021. Archived from the original on May 13, 2021. Retrieved May 13, 2021.
- ^ D'Amore, Rachel (April 8, 2021). "Vaccine mixing 'possible' in Canada as countries revamp AstraZeneca plans: Njoo". Global News. Archived from the original on May 21, 2021. Retrieved May 21, 2021.
- ^ Allen, Nathan (May 18, 2021). "AstraZeneca 1st dose, Pfizer 2nd: Study says combination is safe and effective". Global News. Archived from the original on May 21, 2021. Retrieved May 21, 2021.
- ^ Shaw, Robert; Stuart, Arabella; Greenland, Melanie; Liu, Xinxue; Van-Tam, Jonathan S. Nguyen; Snape, Matthew D. (May 12, 2021). "Heterologous prime-boost COVID-19 vaccination: initial reactogenicity data". The Lancet. 397 (10289): 2043–2046. doi:10.1016/S0140-6736(21)01115-6. PMC 8115940. PMID 33991480.
- ^ Pelley, Lauren (May 12, 2021). "Mixing COVID-19 vaccine doses leads to more reactions, study finds, which may be 'first sign of success'". CBC News. Archived from the original on May 22, 2021. Retrieved May 21, 2021.
- ^ Comité sur l’immunisation du Québec (May 7, 2021). "Utilisation du vaccin AstraZeneca contre la COVID-19 dans le contexte du signal de thromboses avec thrombocytopénie suite à la vaccination" (PDF). INSPQ (in French). Gouvernement du Québec. Archived from the original (PDF) on May 14, 2021.
- ^ Riopel, Alexis; Cousineau, Marie-Eve (April 22, 2021). "Québec forcé d'interchanger les vaccins en CHSLD" (in French). Le Devoir. Archived from the original on May 4, 2021. Retrieved May 3, 2021.
- ^ Taylor, Brooke (May 20, 2021). "Canadian study to investigate mix-and-match COVID-19 vaccines". CTV News. Archived from the original on May 21, 2021. Retrieved May 21, 2021.
- ^ "INTERCHANGEABILITY OF AUTHORIZED COVID-19 VACCINES" (PDF). National Advisory Committee on Immunization. Government of Canada. Archived from the original (PDF) on June 1, 2021. Retrieved June 1, 2021.
- ^ Miller, Adam (June 1, 2021). "Canada to recommend mixing and matching AstraZeneca, Pfizer and Moderna COVID-19 vaccines". CBC News. Archived from the original on June 1, 2021. Retrieved June 1, 2021.
- ^ NACI (June 17, 2021). "Summary of National Advisory Committee on Immunization statement of June 17, 2021". National Advisory Committee on Immunization (NACI): Statements and publications. Government of Canada. Archived from the original on June 19, 2021. Retrieved June 19, 2021.
- ^ Tasker, John Paul (June 17, 2021). "Vaccine committee says provinces should give AstraZeneca recipients a different vaccine for second dose". CBC News. Archived from the original on June 19, 2021. Retrieved June 19, 2021.
- ^ "COVID-19 vaccine: Guidance on the prioritization of initial doses". Public Health Agency of Canada. December 4, 2020. Archived from the original on May 6, 2021. Retrieved May 4, 2021.
- ^ a b "COVID-19 immunization: Prioritization of key populations guidance". Public Health Agency of Canada. February 15, 2021. Archived from the original on May 6, 2021. Retrieved May 4, 2021.
- ^ Brewster, Murray (May 15, 2021). "Fortin out as Canada's vaccine campaign lead amid military probe into sexual misconduct claim, sources say". CBC News. Archived from the original on May 15, 2021. Retrieved May 15, 2021.
External links[edit]
- COVID-19 Tracker Canada, Vaccination Tracker, December 3, 2020, retrieved February 18, 2021
- Vaccines and treatments for COVID-19: Vaccine rollout, Public Health Agency of Canada, December 3, 2020, retrieved February 18, 2021
