Dorsal raphe nucleus
| Dorsal raphe nucleus | |
|---|---|
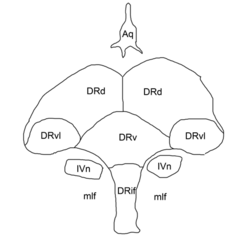 Outline of the dorsal raphe nucleus: DRif interfascicular subnucleus, DRv ventral subnucleus, DRvl ventrolateral subnucleus, DRd dorsal subnucleus, mlf medial longitudinal fasciculus, Aq cerebral aqueduct, IVn trochlear nucleus. | |
| Details | |
| Identifiers | |
| Latin | nucleus raphes posterior, nucleus raphes dorsalis |
| MeSH | D065847 |
| NeuroNames | 512 |
| NeuroLex ID | birnlex_982 |
| TA98 | A14.1.05.604 |
| TA2 | 5957 |
| FMA | 68462 |
| Anatomical terms of neuroanatomy | |
The dorsal raphe nucleus is one of the raphe nuclei. It is situated in the brainstem at the midline. It has rostral and caudal subdivisions:
- The rostral aspect of the dorsal raphe is further divided into interfascicular, ventral, ventrolateral and dorsal subnuclei.
- The projections of the dorsal raphe have been found to vary topographically, and thus the subnuclei differ in their projections.[1]
Anatomy[edit]
Efferents[edit]
The DRN issues serotonergic efferents to the hippocampal formation, limbic lobe, and amygdala (these efferents are involved in regulation of memory processing).[2]
Neurophysiology[edit]
Serotonergic neurotransmission[edit]
The dorsal raphe is the largest serotonergic nucleus and provides a substantial proportion of the serotonin innervation to the forebrain.
Serotonergic neurons are found throughout the dorsal raphe nucleus and tend to be larger than other cells. A substantial population of cells synthesizing substance P are found in the rostral aspects, many of these co-express serotonin and substance P. There is also a population of catecholamine synthesizing neurons in the rostral dorsal raphe, and these cells appear to be relatively large.[3]
The dorsal raphe nucleus is rich in pre-synaptic serotonin 5-HT1A autoreceptors, and it's believed that the action of the selective serotonin reuptake inhibitors (SSRIs) in this region is responsible for the latency of their antidepressant effect.[4]
Research[edit]
Naloxone-induced morphine withdrawal[edit]
The dorsal raphe nucleus has also been implicated in naloxone-induced morphine withdrawal. It is known that endogenous opioid receptors exist in the dorsal raphe nucleus, and that it is a focal point as an ascending and descending regulator. Pourshanazari et al. showed in their 2000 paper that electrical stimulation of the dorsal raphe nucleus can partially alleviate morphine withdrawal symptoms via electrical stimulation of the raphe nucleus in question.[5]
These are fascinating results; however no control was provided for the spread of electrical charge to other parts of the brain stem. It is quite possible that the charge spread to the nucleus raphes magnus and induced analgesia upon the rats. Knowing that the spread of charge across such a short area is very plausible, as is an alternate connection to the raphe magnus, these results could be called into question.
Narcolepsy[edit]
Wu M.F. et al. studied the dorsal raphe nucleus as it pertained to narcolepsy. This is logical, as the raphe nuclei have been known to play a role in the sleep/wake cycle. Cataplexy is the symptom of narcolepsy when full awareness of the environment is maintained, but all muscle tone is lost. This has thought to be a dissociation of what normally happens during REM sleep, when all muscle tone is lost except for the eyes. The dorsal raphe nucleus has been known to project to the lateral hypothalamus, along with the locus coeruleus and the tuberomammillary nucleus. The neurotransmitters of these three aforementioned nuclei, which project to the lateral hypothalamus, are serotonin, norepinephrine and histamine respectively. These neurotransmitters are fully active during waking hours, partially active during non-REM sleep, and have almost ceased during REM sleep. In cats with pontine lesions, their normal atonia is not present, the dorsal raphe nucleus is fully active, as opposed to the cessation of action under normal conditions. A muscle relaxant, known as Mephenesin, reduces activity of the dorsal nucleus, as well as microinjections of carbachol (which induces atonia while awake).[6]
Depression and suicide[edit]
The rostral raphe nuclei, both the median raphe nucleus and particularly the dorsal raphe nucleus have long been implicated in depression. Some studies have suggested that the dorsal raphe may be decreased in size in people with depression and, paradoxically, an increased cell density in those who die by suicide.[7][8][9]
Other animals[edit]
An increased number of cells in the lateral aspects of the dorsal raphe is characteristic of primates (including humans).[citation needed]
Rat[edit]
Research[edit]
Ten percent of the axons from the dorsal raphe nucleus of the rat have been shown to project to the amygdala,[10] while only medium cells seem to project to the caudate and putamen and olfactory bulb.[11][12]
See also[edit]
References[edit]
- ^ O'Hearn E, Molliver ME (December 1984). "Organization of raphe-cortical projections in rat: a quantitative retrograde study". Brain Res. Bull. 13 (6): 709–26. doi:10.1016/0361-9230(84)90232-6. PMID 6099744. S2CID 4761755.
- ^ Patestas, Maria A.; Gartner, Leslie P. (2016). A Textbook of Neuroanatomy (2nd ed.). Hoboken, New Jersey: Wiley-Blackwell. p. 432. ISBN 978-1-118-67746-9.
- ^ Baker KG, Halliday GM, Hornung JP, Geffen LB, Cotton RG, Törk I (1991). "Distribution, morphology and number of monoamine-synthesizing and substance P-containing neurons in the human dorsal raphe nucleus". Neuroscience. 42 (3): 757–75. doi:10.1016/0306-4522(91)90043-N. PMID 1720227. S2CID 23034680.
- ^ Briley M, Moret C (October 1993). "Neurobiological mechanisms involved in antidepressant therapies". Clin Neuropharmacol. 16 (5): 387–400. doi:10.1097/00002826-199310000-00002. PMID 8221701.
- ^ Pourshanazari, A.A.; Alaei; Rafati (2000). "Effects of Electrical Stimulation of Nucleus Raphe Dorsalis on initiation of morphine self-administration in rats". Medical Journal of Islamic Academy of Sciences. 13 (2): 63–7.
- ^ Wu MF, John J, Boehmer LN, Yau D, Nguyen GB, Siegel JM (January 2004). "Activity of dorsal raphe cells across the sleep–waking cycle and during cataplexy in narcoleptic dogs". J. Physiol. 554 (Pt 1): 202–15. doi:10.1113/jphysiol.2003.052134. PMC 1664742. PMID 14678502.
- ^ Underwood MD, Khaibulina AA, Ellis SP, Moran A, Rice PM, Mann JJ, Arango V (August 1999). "Morphometry of the dorsal raphe nucleus serotonergic neurons in suicide victims". Biol Psychiatry. 46 (4): 473–83. doi:10.1016/S0006-3223(99)00043-8. PMID 10459396. S2CID 19780741.
- ^ Arango V, Underwood MD, Boldrini M, Tamir H, Kassir SA, Hsiung S, Chen JJ, Mann JJ (December 2001). "Serotonin 1A receptors, serotonin transporter binding and serotonin transporter mRNA expression in the brainstem of depressed suicide victims". Neuropsychopharmacology. 25 (6): 892–903. doi:10.1016/S0893-133X(01)00310-4. hdl:2158/200883. PMID 11750182.
- ^ Matthews PR, Harrison PJ (March 2012). "A morphometric, immunohistochemical, and in situ hybridization study of the dorsal raphe nucleus in major depression, bipolar disorder, schizophrenia, and suicide". J Affect Disord. 137 (1–3): 125–134. doi:10.1016/j.jad.2011.10.043. PMC 3314923. PMID 22129767.
- ^ Ma QP, Yin GF, Ai MK, Han JS (December 1991). "Serotonergic projections from the nucleus raphe dorsalis to the amygdala in the rat". Neurosci. Lett. 134 (1): 21–4. doi:10.1016/0304-3940(91)90499-J. PMID 1815148. S2CID 5713957.
- ^ Steinbusch HW, Nieuwenhuys R, Verhofstad AA, Van der Kooy D (1981). "The nucleus raphe dorsalis of the rat and its projection upon the caudatoputamen. A combined cytoarchitectonic, immunohistochemical and retrograde transport study". J. Physiol. (Paris). 77 (2–3): 157–74. PMID 6169825.
- ^ Petzold GC, Hagiwara A, Murthy VN (June 2009). "Serotonergic modulation of odor input to the mammalian olfactory bulb". Nat. Neurosci. 12 (6): 784–91. doi:10.1038/nn.2335. PMID 19430472. S2CID 33863055.
