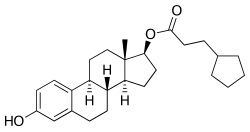
Estradiol cypionate
 | |
 | |
| Clinical data | |
|---|---|
| Pronunciation | /ˌɛstrəˈdaɪoʊl sɪˈpaɪoʊneɪt/ ES-trə-DY-ohl sih-PY-oh-nate[1] |
| Trade names | Depo-Estradiol, Depofemin, Estradep, many others |
| Other names | EC; E2C; Estradiol cipionate; Estradiol cyclopentylpropionate; ECP; Estradiol 17β-cyclopentylpropionate; Estradiol 17β-cyclopentanepropionate |
| Routes of administration | Intramuscular injection, subcutaneous injection[2] |
| Drug class | Estrogen; Estrogen ester |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | IM: High[4] |
| Protein binding | Estradiol: ~98% (to albumin and SHBG)[5][6] |
| Metabolism | Cleavage via esterases in the liver, blood, and tissues[7][8] |
| Metabolites | Estradiol, cypionic acid, and metabolites of estradiol[7][8] |
| Elimination half-life | IM (aqueous suspension): 8–10 days[9][10] |
| Duration of action | IM (oil): 5 mg ≈ 11–14 days[11] IM (aqueous suspension): 5 mg ≈ 14–24 days[9][12][13] |
| Excretion | Urine |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.005.672 |
| Chemical and physical data | |
| Formula | C26H36O3 |
| Molar mass | 396.571 g·mol−1 |
| 3D model (JSmol) | |
| Melting point | 151 to 152 °C (304 to 306 °F) |
| |
| |
Estradiol cypionate (EC), sold under the brand name Depo-Estradiol among others, is an estrogen medication which is used in hormone therapy for menopausal symptoms and low estrogen levels in women, in hormone therapy for trans women, and in hormonal birth control for women.[14][8][15][16] It is given by injection into muscle once every 1 to 4 weeks.[14][17]
Side effects of estradiol cypionate include breast tenderness, breast enlargement, nausea, headache, and fluid retention.[14][8] Estradiol cypionate is an estrogen and hence is an agonist of the estrogen receptor, the biological target of estrogens like estradiol.[8][7] Estradiol cypionate is an estrogen ester and a long-lasting prodrug of estradiol in the body.[14][8][7] Because of this, it is considered to be a natural and bioidentical form of estrogen.[7][18][12]
Estradiol cypionate was first described as well as introduced for medical use in 1952.[19][20] Along with estradiol valerate, it is one of the most commonly used esters of estradiol.[21] Estradiol cypionate has mostly been used in the United States, but is also marketed in a few other countries.[22][23][24] The medication is not available in Europe.[25] It is not currently available as a generic medication in the United States.[26]
Medical uses[edit]
The medical uses of estradiol cypionate are the same as those of estradiol and other estrogens. Examples of indications for the drug include hormone therapy and hormonal contraception. In regard to the latter, estradiol cypionate has been used in combination with medroxyprogesterone acetate as a combined injectable contraceptive.[15][16][27] Along with estradiol valerate, estradiol undecylate, and estradiol benzoate, estradiol cypionate is used as a form of high-dose estrogen therapy in feminizing hormone therapy for transgender women.[28][17][29][30] The medication has been used to induce puberty in girls with delayed puberty due to hypogonadism.[31][25]
Estradiol cypionate is usually used at a dosage of 1 to 5 mg by intramuscular injection every 3 to 4 weeks in the treatment of menopausal symptoms such as hot flashes and vaginal atrophy, at a dosage of 1.5 to 2 mg by intramuscular injection once a month in the treatment of female hypoestrogenism due to hypogonadism, and at a dosage of 2 to 10 mg by intramuscular injection once every 1 or 2 weeks for hormone therapy in transgender women.[14][17][29][28][32] The doses used to induce puberty in girls are 0.2 to 2.5 mg per month, gradually increased over a period of 4 years.[31][25]
| Route/form | Estrogen | Low | Standard | High | |||
|---|---|---|---|---|---|---|---|
| Oral | Estradiol | 0.5–1 mg/day | 1–2 mg/day | 2–4 mg/day | |||
| Estradiol valerate | 0.5–1 mg/day | 1–2 mg/day | 2–4 mg/day | ||||
| Estradiol acetate | 0.45–0.9 mg/day | 0.9–1.8 mg/day | 1.8–3.6 mg/day | ||||
| Conjugated estrogens | 0.3–0.45 mg/day | 0.625 mg/day | 0.9–1.25 mg/day | ||||
| Esterified estrogens | 0.3–0.45 mg/day | 0.625 mg/day | 0.9–1.25 mg/day | ||||
| Estropipate | 0.75 mg/day | 1.5 mg/day | 3 mg/day | ||||
| Estriol | 1–2 mg/day | 2–4 mg/day | 4–8 mg/day | ||||
| Ethinylestradiola | 2.5–10 μg/day | 5–20 μg/day | – | ||||
| Nasal spray | Estradiol | 150 μg/day | 300 μg/day | 600 μg/day | |||
| Transdermal patch | Estradiol | 25 μg/dayb | 50 μg/dayb | 100 μg/dayb | |||
| Transdermal gel | Estradiol | 0.5 mg/day | 1–1.5 mg/day | 2–3 mg/day | |||
| Vaginal | Estradiol | 25 μg/day | – | – | |||
| Estriol | 30 μg/day | 0.5 mg 2x/week | 0.5 mg/day | ||||
| IM or SC injection | Estradiol valerate | – | – | 4 mg 1x/4 weeks | |||
| Estradiol cypionate | 1 mg 1x/3–4 weeks | 3 mg 1x/3–4 weeks | 5 mg 1x/3–4 weeks | ||||
| Estradiol benzoate | 0.5 mg 1x/week | 1 mg 1x/week | 1.5 mg 1x/week | ||||
| SC implant | Estradiol | 25 mg 1x/6 months | 50 mg 1x/6 months | 100 mg 1x/6 months | |||
| Footnotes: a = No longer used or recommended, due to health concerns. b = As a single patch applied once or twice per week (worn for 3–4 days or 7 days), depending on the formulation. Note: Dosages are not necessarily equivalent. Sources: See template. | |||||||
Available forms[edit]
Estradiol cypionate is and has been available as an oil solution for intramuscular injection provided in vials and ampoules at concentrations of 1, 3, and 5 mg/mL (and containing 5, 10, 15, 25, or 50 mg estradiol cypionate total).[26][33][34] The 1 and 3 mg/mL concentrations (containing 5 and 15 mg estradiol cypionate total) have been discontinued in the United States, but the 5 mg/mL concentration (containing 25 mg estradiol cypionate total) remains available.[26][35] Aside from estradiol cypionate, the only other injectable estrogen formulations that remain available in the United States are estradiol valerate (10 mg/mL, 20 mg/mL, and 40 mg/mL in oil) and conjugated estrogens (25 mg/vial in solution).[26]
In addition to single-drug formulations, estradiol cypionate has been marketed in combination with medroxyprogesterone acetate as a microcrystalline aqueous suspension (brand name Lunelle) and in combination with testosterone cypionate as an oil solution (brand name Depo-Testadiol).[26]
| Route | Ingredient | Form | Dose[b] | Brand names[c] |
|---|---|---|---|---|
| Oral | Estradiol | Tablet | 0.1, 0.2, 0.5, 1, 2, 4 mg | Estrace, Ovocyclin |
| Estradiol valerate | Tablet | 0.5, 1, 2, 4 mg | Progynova | |
| Transdermal | Estradiol | Patch | 14, 25, 37.5, 50, 60, 75, 100 µg/d | Climara, Vivelle |
| Gel pump | 0.06% (0.52, 0.75 mg/pump) | Elestrin, EstroGel | ||
| Gel packet | 0.1% (0.25, 0.5, 1.0 mg/pk.) | DiviGel, Sandrena | ||
| Emulsion | 0.25% (25 µg/pouch) | Estrasorb | ||
| Spray | 1.53 mg/spray | Evamist, Lenzetto | ||
| Vaginal | Estradiol | Tablet | 10, 25 µg | Vagifem |
| Cream | 0.01% (0.1 mg/gram) | Estrace | ||
| Insert | 4, 10 µg | Imvexxy | ||
| Ring | 2 mg/ring (7.5 µg/d, 3 mon.) | Estring | ||
| Estradiol acetate | Ring | 50, 100 µg/d, 3 months | Femring | |
| Injection[d] | Estradiol | Microspheres | 1 mg/mL | Juvenum E |
| Estradiol benzoate | Oil solution | 0.167, 0.2, 0.333, 1, 1.67, 2, 5, 10, 20, 25 mg/mL | Progynon-B | |
| Estradiol cypionate | Oil solution | 1, 3, 5 mg/mL | Depo-Estradiol | |
| Estradiol valerate | Oil solution | 5, 10, 20, 40 mg/mL | Progynon Depot | |
| Implant | Estradiol | Pellet | 20, 25, 50, 100 mg, 6 mon. | Estradiol Implants |
Notes and sources:
| ||||

Contraindications[edit]
Contraindications of estrogens include coagulation problems, cardiovascular diseases, liver disease, and certain hormone-sensitive cancers such as breast cancer and endometrial cancer, among others.[50][51][52][53]
Side effects[edit]
The side effects of estradiol cypionate are the same as those of estradiol. Examples of such side effects include breast tenderness and enlargement, nausea, vomiting, bloating, edema, headache, migraine, and melasma.[54][55] High-dose estrogen therapy with estradiol cypionate injections may also cause an increased risk of thromboembolism, changes in blood lipid profile, increased insulin resistance, and increased levels of prolactin.[55]
Overdose[edit]
Symptoms of estrogen overdosage may include nausea, vomiting, bloating, increased weight, water retention, breast tenderness, vaginal discharge, heavy legs, and leg cramps.[50] These side effects can be diminished by reducing the estrogen dosage.[50]
Interactions[edit]
Inhibitors and inducers of cytochrome P450 may influence the metabolism of estradiol and by extension circulating estradiol levels.[56]
Pharmacology[edit]

Pharmacodynamics[edit]
Estradiol cypionate is an estradiol ester, or a prodrug of estradiol.[8][7] As such, it is an estrogen, or an agonist of the estrogen receptors.[8][7] The affinity of estradiol valerate for the estrogen receptor has been reported to be 50 times less than that of estradiol,[4] and estradiol valerate and estradiol cypionate have been found to possess similar affinity for the estrogen receptor.[57] Both estradiol cypionate and estradiol valerate are rapidly cleaved into estradiol in the body,[8][58] and estradiol valerate has been found to be unable to reach target tissues in any concentration of significance.[4] As such, estradiol valerate is regarded as essentially inactive in terms of estrogenic effect itself, acting solely as a prodrug to estradiol,[4] and estradiol cypionate is described as a prodrug of estradiol similarly.[7] Estradiol cypionate is of about 46% higher molecular weight than estradiol due to the presence of its C17β cypionate ester, and contains about 69% of the amount of estradiol by weight.[59][22][25] Because estradiol cypionate is a prodrug of estradiol, it is considered to be a natural and bioidentical form of estrogen.[7][18][12]
| Estrogen | Other names | RBA (%)a | REP (%)b | |||
|---|---|---|---|---|---|---|
| ER | ERα | ERβ | ||||
| Estradiol | E2 | 100 | 100 | 100 | ||
| Estradiol 3-sulfate | E2S; E2-3S | ? | 0.02 | 0.04 | ||
| Estradiol 3-glucuronide | E2-3G | ? | 0.02 | 0.09 | ||
| Estradiol 17β-glucuronide | E2-17G | ? | 0.002 | 0.0002 | ||
| Estradiol benzoate | EB; Estradiol 3-benzoate | 10 | 1.1 | 0.52 | ||
| Estradiol 17β-acetate | E2-17A | 31–45 | 24 | ? | ||
| Estradiol diacetate | EDA; Estradiol 3,17β-diacetate | ? | 0.79 | ? | ||
| Estradiol propionate | EP; Estradiol 17β-propionate | 19–26 | 2.6 | ? | ||
| Estradiol valerate | EV; Estradiol 17β-valerate | 2–11 | 0.04–21 | ? | ||
| Estradiol cypionate | EC; Estradiol 17β-cypionate | ?c | 4.0 | ? | ||
| Estradiol palmitate | Estradiol 17β-palmitate | 0 | ? | ? | ||
| Estradiol stearate | Estradiol 17β-stearate | 0 | ? | ? | ||
| Estrone | E1; 17-Ketoestradiol | 11 | 5.3–38 | 14 | ||
| Estrone sulfate | E1S; Estrone 3-sulfate | 2 | 0.004 | 0.002 | ||
| Estrone glucuronide | E1G; Estrone 3-glucuronide | ? | <0.001 | 0.0006 | ||
| Ethinylestradiol | EE; 17α-Ethynylestradiol | 100 | 17–150 | 129 | ||
| Mestranol | EE 3-methyl ether | 1 | 1.3–8.2 | 0.16 | ||
| Quinestrol | EE 3-cyclopentyl ether | ? | 0.37 | ? | ||
| Footnotes: a = Relative binding affinities (RBAs) were determined via in-vitro displacement of labeled estradiol from estrogen receptors (ERs) generally of rodent uterine cytosol. Estrogen esters are variably hydrolyzed into estrogens in these systems (shorter ester chain length -> greater rate of hydrolysis) and the ER RBAs of the esters decrease strongly when hydrolysis is prevented. b = Relative estrogenic potencies (REPs) were calculated from half-maximal effective concentrations (EC50) that were determined via in-vitro β‐galactosidase (β-gal) and green fluorescent protein (GFP) production assays in yeast expressing human ERα and human ERβ. Both mammalian cells and yeast have the capacity to hydrolyze estrogen esters. c = The affinities of estradiol cypionate for the ERs are similar to those of estradiol valerate and estradiol benzoate (figure). Sources: See template page. | ||||||
| Estrogen | Form | Dose (mg) | Duration by dose (mg) | ||
|---|---|---|---|---|---|
| EPD | CICD | ||||
| Estradiol | Aq. soln. | ? | – | <1 d | |
| Oil soln. | 40–60 | – | 1–2 ≈ 1–2 d | ||
| Aq. susp. | ? | 3.5 | 0.5–2 ≈ 2–7 d; 3.5 ≈ >5 d | ||
| Microsph. | ? | – | 1 ≈ 30 d | ||
| Estradiol benzoate | Oil soln. | 25–35 | – | 1.66 ≈ 2–3 d; 5 ≈ 3–6 d | |
| Aq. susp. | 20 | – | 10 ≈ 16–21 d | ||
| Emulsion | ? | – | 10 ≈ 14–21 d | ||
| Estradiol dipropionate | Oil soln. | 25–30 | – | 5 ≈ 5–8 d | |
| Estradiol valerate | Oil soln. | 20–30 | 5 | 5 ≈ 7–8 d; 10 ≈ 10–14 d; 40 ≈ 14–21 d; 100 ≈ 21–28 d | |
| Estradiol benz. butyrate | Oil soln. | ? | 10 | 10 ≈ 21 d | |
| Estradiol cypionate | Oil soln. | 20–30 | – | 5 ≈ 11–14 d | |
| Aq. susp. | ? | 5 | 5 ≈ 14–24 d | ||
| Estradiol enanthate | Oil soln. | ? | 5–10 | 10 ≈ 20–30 d | |
| Estradiol dienanthate | Oil soln. | ? | – | 7.5 ≈ >40 d | |
| Estradiol undecylate | Oil soln. | ? | – | 10–20 ≈ 40–60 d; 25–50 ≈ 60–120 d | |
| Polyestradiol phosphate | Aq. soln. | 40–60 | – | 40 ≈ 30 d; 80 ≈ 60 d; 160 ≈ 120 d | |
| Estrone | Oil soln. | ? | – | 1–2 ≈ 2–3 d | |
| Aq. susp. | ? | – | 0.1–2 ≈ 2–7 d | ||
| Estriol | Oil soln. | ? | – | 1–2 ≈ 1–4 d | |
| Polyestriol phosphate | Aq. soln. | ? | – | 50 ≈ 30 d; 80 ≈ 60 d | |
Notes and sources
Notes: All aqueous suspensions are of microcrystalline particle size. Estradiol production during the menstrual cycle is 30–640 µg/d (6.4–8.6 mg total per month or cycle). The vaginal epithelium maturation dosage of estradiol benzoate or estradiol valerate has been reported as 5 to 7 mg/week. An effective ovulation-inhibiting dose of estradiol undecylate is 20–30 mg/month. Sources: See template. | |||||
Effects on liver protein synthesis[edit]
A study compared the combination of 5 mg estradiol cypionate and 25 mg medroxyprogesterone acetate as a combined injectable contraceptive (which has been associated with peak estradiol levels of around 300 pg/mL) with an ethinylestradiol-containing combined birth control pill and found that whereas the birth control pill produced significant changes in coagulation parameters, there were no significant prothrombotic effects of the combined injectable contraceptive on levels of fibrinogen, factors VII and X, plasminogen, or the activated prothrombin time.[60] As such, it appears that similarly to depot medroxyprogesterone acetate, combined injectable contraceptives with 5 mg estradiol cypionate and 25 mg medroxyprogesterone acetate have less or no procoagulant effect relative to combined birth control pills.[60]
Pharmacokinetics[edit]
Intramuscular injection[edit]
In contrast to oral administration, which is associated with very low bioavailability (<10%), the bioavailability of both estradiol and estradiol esters like estradiol valerate is complete (i.e., 100%) via intramuscular injection.[4][8] In addition, estradiol esters like estradiol cypionate and estradiol valerate when given as an injection of oil solution or microcrystalline aqueous suspension have a relatively long duration due to the formation of an intramuscular depot from which they are slowly released and absorbed.[4][61][62] Upon intramuscular injection of estradiol cypionate in an oil solution, the solvent (i.e., oil) is absorbed, and a primary microcrystalline depot is formed within the muscle at the site of injection.[8] In addition, a secondary depot may be formed in adipose tissue.[8] The slow release of estradiol cypionate from the tissue depot is caused by the high lipophilicity of the estradiol ester, which in turn is due to its long fatty acid cypionic acid ester moiety.[4] Estradiol cypionate is formulated for use alone and in combination with testosterone cypionate as an oil solution, and for use in combination with medroxyprogesterone acetate as a microcrystalline aqueous suspension.[26][33][34][35] Aqueous suspensions of steroid esters generally have longer durations by intramuscular injection than oil solutions.[62]
A single intramuscular injection of 5 mg estradiol cypionate has been found to result in peak circulating concentrations of 338 pg/mL estradiol and 145 pg/mL estrone, which occurred at about 4 and 5 days post-injection, respectively (see right table).[11] Compared to two other commonly used estradiol esters (which were also assessed in the study), estradiol cypionate had the longest duration, at approximately 11 days, whereas estradiol benzoate and estradiol valerate were found to last for 4 to 5 days and 7 to 8 days, respectively.[11] This is because estradiol cypionate has a more extensive fatty acid chain and in relation to this is comparatively more lipophilic.[8] For a given estradiol ester, the longer or more extensive the fatty acid chain is, the more lipophilic, longer-lasting, and more uniform/plateau-like the resultant levels of estradiol are as well as the lower the peak/maximal levels are (and hence less spike-like).[8]
Estradiol cypionate/medroxyprogesterone acetate (brand names Lunelle, Cyclofem) is a combined injectable contraceptive containing 5 mg estradiol cypionate and 25 mg medroxyprogesterone acetate in microcrystalline aqueous suspension for once-monthly intramuscular administration.[12][13] With this formulations, estradiol levels peak 2 to 3 days post-injection with average maximal circulating levels of about 250 pg/mL.[9][12][13] The elimination half-life of estradiol with these formulations is 8.4 to 10.1 days, and circulating estradiol levels return to a baseline of about 50 pg/mL approximately 14 to 24 days post-injection.[9][12][13][10]
-
Estradiol levels after subcutaneous (s.c.) or intramuscular (i.m.) injection of 5 mg estradiol cypionate in aqueous suspension.[2] Assays were performed using enzyme immunoassay.[2] Source was Sierra-Ramírez et al. (2011).[2]
-
Estradiol levels at steady state (after the 3rd injection) with intramuscular injections of aqueous suspensions of 5 mg estradiol cypionate per month in premenopausal women.[63][9] Assays were performed using enzyme immunoassay and LC-MS/MS.[63][9] Sources were Rahimy et al. (1999) and Thurman et al. (2013).[63][9]
-
Estradiol levels after a single intramuscular injection of 1.0 to 2.0 mg (average 1.67 mg) of estradiol cypionate in oil (Depo-Estradiol) in hypogonadal girls.[64][65] Assays were performed using radioimmunoassay with chromatographic separation.[64][65] Sources were Rosenfield et al. (1973, 1974).[64][65]
-
Estradiol levels after single intramuscular injections of 5 mg of different estradiol esters in oil in premenopausal women.[11] Assays were performed using radioimmunoassay with chromatographic separation.[11] Source was Oriowo et al. (1980).[11]
-
Vaginal cornification with a single intramuscular injection of different estradiol esters in oil solution in women.[68] Source was Schwartz & Soule (1955).[68]
Subcutaneous injection[edit]
Estradiol cypionate in a microcrystalline aqueous suspension has been found to have equivalent effectiveness and virtually identical pharmacokinetics when administered by subcutaneous injection versus intramuscular injection.[2] However, subcutaneous injection is considered to be easier and less painful relative to intramuscular injection, and for these reasons, may result in comparatively greater satisfaction and compliance.[2]
Chemistry[edit]
Estradiol cypionate is a synthetic estrane steroid and the C17β cyclopentylpropionate (cypionate) fatty acid ester of estradiol.[59][22] It is also known as estra-1,3,5(10)-triene-3,17β-diol 17β-cyclopentylpropionate.[59][22] Other common esters of estradiol in use include estradiol valerate, estradiol enantate, and estradiol acetate, the former two of which are C17β esters of estradiol similarly to estradiol cypionate and the latter of which is the C3 acetate ester of estradiol.
The experimental octanol/water partition coefficient (logP) of estradiol cypionate is 6.9.[69]
| Estrogen | Structure | Ester(s) | Relative mol. weight |
Relative E2 contentb |
log Pc | ||||
|---|---|---|---|---|---|---|---|---|---|
| Position(s) | Moiet(ies) | Type | Lengtha | ||||||
| Estradiol | – | – | – | – | 1.00 | 1.00 | 4.0 | ||
| Estradiol acetate | C3 | Ethanoic acid | Straight-chain fatty acid | 2 | 1.15 | 0.87 | 4.2 | ||
| Estradiol benzoate | C3 | Benzoic acid | Aromatic fatty acid | – (~4–5) | 1.38 | 0.72 | 4.7 | ||
| Estradiol dipropionate | C3, C17β | Propanoic acid (×2) | Straight-chain fatty acid | 3 (×2) | 1.41 | 0.71 | 4.9 | ||
| Estradiol valerate | C17β | Pentanoic acid | Straight-chain fatty acid | 5 | 1.31 | 0.76 | 5.6–6.3 | ||
| Estradiol benzoate butyrate | C3, C17β | Benzoic acid, butyric acid | Mixed fatty acid | – (~6, 2) | 1.64 | 0.61 | 6.3 | ||
| Estradiol cypionate | C17β | Cyclopentylpropanoic acid | Cyclic fatty acid | – (~6) | 1.46 | 0.69 | 6.9 | ||
| Estradiol enanthate | C17β | Heptanoic acid | Straight-chain fatty acid | 7 | 1.41 | 0.71 | 6.7–7.3 | ||
| Estradiol dienanthate | C3, C17β | Heptanoic acid (×2) | Straight-chain fatty acid | 7 (×2) | 1.82 | 0.55 | 8.1–10.4 | ||
| Estradiol undecylate | C17β | Undecanoic acid | Straight-chain fatty acid | 11 | 1.62 | 0.62 | 9.2–9.8 | ||
| Estradiol stearate | C17β | Octadecanoic acid | Straight-chain fatty acid | 18 | 1.98 | 0.51 | 12.2–12.4 | ||
| Estradiol distearate | C3, C17β | Octadecanoic acid (×2) | Straight-chain fatty acid | 18 (×2) | 2.96 | 0.34 | 20.2 | ||
| Estradiol sulfate | C3 | Sulfuric acid | Water-soluble conjugate | – | 1.29 | 0.77 | 0.3–3.8 | ||
| Estradiol glucuronide | C17β | Glucuronic acid | Water-soluble conjugate | – | 1.65 | 0.61 | 2.1–2.7 | ||
| Estramustine phosphated | C3, C17β | Normustine, phosphoric acid | Water-soluble conjugate | – | 1.91 | 0.52 | 2.9–5.0 | ||
| Polyestradiol phosphatee | C3–C17β | Phosphoric acid | Water-soluble conjugate | – | 1.23f | 0.81f | 2.9g | ||
| Footnotes: a = Length of ester in carbon atoms for straight-chain fatty acids or approximate length of ester in carbon atoms for aromatic or cyclic fatty acids. b = Relative estradiol content by weight (i.e., relative estrogenic exposure). c = Experimental or predicted octanol/water partition coefficient (i.e., lipophilicity/hydrophobicity). Retrieved from PubChem, ChemSpider, and DrugBank. d = Also known as estradiol normustine phosphate. e = Polymer of estradiol phosphate (~13 repeat units). f = Relative molecular weight or estradiol content per repeat unit. g = log P of repeat unit (i.e., estradiol phosphate). Sources: See individual articles. | |||||||||
History[edit]
Estradiol cypionate was patented by Upjohn in 1952, with a priority date of 1951.[33] It was first introduced for medical use by Upjohn in 1952 under the brand name Depo-Estradiol in the United States.[19][20][70] Subsequently, it was also marketed in other countries such as European countries and Japan.[33][20][22] The first clinical reports of estradiol cypionate were published in 1952 and thereafter.[71][72][73][74][68] It was initially known as estradiol cyclopentylpropionate (ECP), and did not become known as estradiol cypionate until over a decade later in the mid-to-late 1960s.[72][73][75] Along with estradiol valerate (1954)[20][76] and estradiol benzoate (1933),[77][78][79] estradiol cypionate has become one of the most commonly used esters of estradiol.[21]
When estradiol cypionate was to be combined with medroxyprogesterone acetate as a once-a-month injectable contraceptive, there was a problem in that estradiol cypionate was prepared as an oil solution while medroxyprogesterone acetate was used as a microcrystalline aqueous suspension.[80] This issue was resolved by switching to a microcrystalline aqueous suspension in the case of estradiol cypionate, allowing it to be combined with medroxyprogesterone acetate in a single suspension.[80] As a result, single-drug preparations of estradiol cypionate are oil solutions, while the combination of estradiol cypionate and medroxyprogesterone acetate are microcrystalline aqueous suspensions.[80]
Society and culture[edit]
Generic names[edit]
Estradiol cypionate is the generic name of the drug and its INN and USAN.[59][22][23] It is also known as estradiol cyclopentylpropionate (ECP).[72][73]
Brand names[edit]
Estradiol cypionate has been marketed under the brand names Cicloestradiolo, D-Est, depGynogen, Depo-Estradiol, Depoestra, Depofemin, Depogen, Dura-Estrin, E-Cypionate, E-Ionate, Estradep, Estro-Cyp, Estrofem, Estroject, Estromed-PA, Estronol, Femovirin, Neoginon Depositum, Oestradiol-Retard, Pertradiol, Spendepiol, and T-E Cypionate, among others.[59][22][20][23]
Availability[edit]
Estradiol cypionate is available in the United States.[26][23][25] It was previously marketed in Spain and Italy, but was discontinued in these countries and is no longer available in Europe.[22][25] Estradiol cypionate has mostly been used in the United States, similarly to testosterone cypionate, with both of these medications having been developed by Upjohn, an American pharmaceutical company.[22][24] Besides the United States, estradiol cypionate has been marketed in France, Germany, Italy, Spain, and Japan, among other countries.[33][20][22] Estradiol cypionate for human use is not available in Canada, although it is marketed in several veterinary formulations in this country.[81]
Estradiol cypionate is available in Taiwan in combination with testosterone cypionate.[23] It is also available as a combined injectable contraceptive in combination with medroxyprogesterone acetate in at least 18 countries, mostly in Latin America and Southeast Asia.[82][83][84][16][85][86][87] Estradiol cypionate/testosterone cypionate and estradiol cypionate/medroxyprogesterone acetate were both formerly marketed in the United States, but have been discontinued in this country.[26]
See also[edit]
References[edit]
- ^ "Estradiol: Uses, Dosage & Side Effects". Drugs.com. Retrieved 21 April 2023.
- ^ a b c d e f Sierra-Ramírez JA, Lara-Ricalde R, Lujan M, Velázquez-Ramírez N, Godínez-Victoria M, Hernádez-Munguía IA, Padilla A, Garza-Flores J (2011). "Comparative pharmacokinetics and pharmacodynamics after subcutaneous and intramuscular administration of medroxyprogesterone acetate (25 mg) and estradiol cypionate (5 mg)". Contraception. 84 (6): 565–70. doi:10.1016/j.contraception.2011.03.014. PMID 22078184.
- ^ "FDA-sourced list of all drugs with black box warnings (Use Download Full Results and View Query links.)". nctr-crs.fda.gov. FDA. Retrieved 22 Oct 2023.
- ^ a b c d e f g Düsterberg B, Nishino Y (December 1982). "Pharmacokinetic and pharmacological features of oestradiol valerate". Maturitas. 4 (4): 315–24. doi:10.1016/0378-5122(82)90064-0. PMID 7169965.
- ^ Stanczyk FZ, Archer DF, Bhavnani BR (June 2013). "Ethinyl estradiol and 17β-estradiol in combined oral contraceptives: pharmacokinetics, pharmacodynamics and risk assessment". Contraception. 87 (6): 706–727. doi:10.1016/j.contraception.2012.12.011. PMID 23375353.
- ^ Gupta MK, Chia SY (2007). "Ovarian Hormones: Structure, Biosynthesis, Function, Mechanism of Action, and Laboratory Diagnosis". In Falcone T, Hurd WW (eds.). Clinical Reproductive Medicine and Surgery. Elsevier Health Sciences. p. 22. ISBN 978-0-323-03309-1.; Arredondo F, Liu JH (2007). "Menopause". In Falcone T, Hurd WW (eds.). Clinical Reproductive Medicine and Surgery. Elsevier Health Sciences. pp. 362, 388. ISBN 978-0-323-03309-1.
- ^ a b c d e f g h i Oettel M, Schillinger E (6 December 2012). Estrogens and Antiestrogens II: Pharmacology and Clinical Application of Estrogens and Antiestrogen. Springer Science & Business Media. p. 261. ISBN 978-3-642-60107-1.
Natural estrogens considered here include: [...] Esters of 17β-estradiol, such as estradiol valerate, estradiol benzoate and estradiol cypionate. Esterification aims at either better absorption after oral administration or a sustained release from the depot after intramuscular administration. During absorption, the esters are cleaved by endogenous esterases and the pharmacologically active 17β-estradiol is released; therefore, the esters are considered as natural estrogens.
- ^ a b c d e f g h i j k l m n Kuhl H (2005). "Pharmacology of estrogens and progestogens: influence of different routes of administration" (PDF). Climacteric. 8 (Suppl 1): 3–63. doi:10.1080/13697130500148875. PMID 16112947. S2CID 24616324.
- ^ a b c d e f g Thurman A, Kimble T, Hall P, Schwartz JL, Archer DF (June 2013). "Medroxyprogesterone acetate and estradiol cypionate injectable suspension (Cyclofem) monthly contraceptive injection: steady-state pharmacokinetics". Contraception. 87 (6): 738–43. doi:10.1016/j.contraception.2012.11.010. PMID 23265980.
- ^ a b "LUNELLE' Monthly Contraceptive Injection" (PDF). accessdata.fda.gov. 2000. Retrieved 21 April 2023.
- ^ a b c d e f Oriowo MA, Landgren BM, Stenström B, Diczfalusy E (April 1980). "A comparison of the pharmacokinetic properties of three estradiol esters". Contraception. 21 (4): 415–24. doi:10.1016/S0010-7824(80)80018-7. PMID 7389356.
- ^ a b c d e f Seth S, Nagrath A, Deoghare R (15 December 2012). "Injectable Contraceptives till Date". In Arun N, Narendra M, Shikha S (eds.). Progress in Obstetrics and Gynecology--3. Jaypee Brothers Medical Publishers Pvt. Ltd. pp. 416–419. ISBN 978-93-5090-575-3.
- ^ a b c d Rahimy MH, Ryan KK, Hopkins NK (October 1999). "Lunelle monthly contraceptive injection (medroxyprogesterone acetate and estradiol cypionate injectable suspension): steady-state pharmacokinetics of MPA and E2 in surgically sterile women". Contraception. 60 (4): 209–214. doi:10.1016/S0010-7824(99)00086-4. PMID 10640167.
- ^ a b c d e f "Estradiol cypionate injection, USP" (PDF). accessdata.fda.gov. 2005. Retrieved 21 April 2023.
- ^ a b Newton JR, D'arcangues C, Hall PE (1994). "A review of "once-a-month" combined injectable contraceptives". J Obstet Gynaecol (Lahore). 4 (Suppl 1): S1–34. doi:10.3109/01443619409027641. PMID 12290848.
- ^ a b c Bagade O, Pawar V, Patel R, Patel B, Awasarkar V, Diwate S (2014). "Increasing use of long-acting reversible contraception: safe, reliable, and cost-effective birth control" (PDF). World J Pharm Pharm Sci. 3 (10): 364–392. ISSN 2278-4357.
- ^ a b c Smith KP, Madison CM, Milne NM (December 2014). "Gonadal suppressive and cross-sex hormone therapy for gender dysphoria in adolescents and adults". Pharmacotherapy. 34 (12): 1282–1297. doi:10.1002/phar.1487. PMID 25220381. S2CID 26979177.
- ^ a b Cirigliano M (June 2007). "Bioidentical hormone therapy: a review of the evidence". J Womens Health (Larchmt). 16 (5): 600–31. doi:10.1089/jwh.2006.0311. PMID 17627398.
- ^ a b Sittig M (1 January 1988). Pharmaceutical Manufacturing Encyclopedia. William Andrew. pp. 575–576. ISBN 978-0-8155-1144-1. Retrieved 20 May 2012.
- ^ a b c d e f William Andrew Publishing (22 October 2013). Pharmaceutical Manufacturing Encyclopedia (3rd ed.). Elsevier. pp. 1476–1477. ISBN 978-0-8155-1856-3.
- ^ a b Yen SS (1991). Reproductive endocrinology: physiology, pathophysiology, and clinical management. Saunders. ISBN 978-0-7216-3206-3. Retrieved 20 May 2012.
- ^ a b c d e f g h i j Index Nominum 2000: International Drug Directory. Taylor & Francis US. 2000. p. 405. ISBN 978-3-88763-075-1. Retrieved 20 May 2012.
- ^ a b c d e "Estradiol". Drugs.com.
- ^ a b Llewellyn W (2011). Anabolics. Molecular Nutrition Llc. pp. 426–. ISBN 978-0-9828280-1-4.
- ^ a b c d e f Bruni V, Bucciantini S, Ambroggio S (2017). "From Primary Hypergonadotropic Amenorrhea to "POI": Aetiology and Therapy". Frontiers in Gynecological Endocrinology: Pediatric and Adolescent Gynecological Endocrinology. ISGE Series. Vol. 4. Springer. pp. 67–109. doi:10.1007/978-3-319-41433-1_7. ISBN 978-3-319-41431-7. ISSN 2197-8735.
- ^ a b c d e f g h "Drugs@FDA: FDA Approved Drug Products". United States Food and Drug Administration. Retrieved 31 December 2017.
- ^ Rowlands S (January 2009). "New technologies in contraception" (PDF). BJOG. 116 (2): 230–239. doi:10.1111/j.1471-0528.2008.01985.x. PMID 19076955. S2CID 3415547.
- ^ a b Wesp LM, Deutsch MB (March 2017). "Hormonal and Surgical Treatment Options for Transgender Women and Transfeminine Spectrum Persons". The Psychiatric Clinics of North America. 40 (1): 99–111. doi:10.1016/j.psc.2016.10.006. PMID 28159148.
- ^ a b Rosenthal SM (20 May 2016). "Transgender Youth: Endocrine Management". In Ettner R, Monstrey S, Coleman E (eds.). Principles of Transgender Medicine and Surgery. Routledge. pp. 216–. ISBN 978-1-317-51460-2.
- ^ Israel GE, Tarver DE, Shaffer JD (1 March 2001). "Transgender Hormone Adiministration". Transgender Care: Recommended Guidelines, Practical Information, and Personal Accounts. Temple University Press. pp. 64–. ISBN 978-1-56639-852-7.
- ^ a b Rosenfield RL, Kiess W, de Muinck Keizer-Schrama S (2006). "Physiologic induction of puberty in Turner syndrome with very low-dose estradiol". International Congress Series. 1298: 71–79. doi:10.1016/j.ics.2006.07.003. ISSN 0531-5131.
- ^ American Medical Association. Dept. of Drugs; Council on Drugs (American Medical Association); American Society for Clinical Pharmacology and Therapeutics (1 February 1977). "Estrogens, Progestagens, Oral Contraceptives, and Ovulatory Agents". AMA drug evaluations. Publishing Sciences Group. pp. 540–572. ISBN 978-0-88416-175-2.
Intramuscular: For replacement therapy, (Estradiol, Estradiol Benzoate) 0.5 to 1.5 mg two or three times weekly; (Estradiol Cypionate) 1 to 5 mg weekly for two or three weeks; (Estradiol Dipropionate) 1 to 5 mg every one to two weeks; (Estradiol Valerate) 10 to 40 mg every one to four weeks.
- ^ a b c d e Kleemann A, Engel J, Kutscher B, Reichert D (2014). Pharmaceutical Substances: Syntheses, Patents and Applications of the most relevant APIs (5th ed.). Thieme. pp. 1167–1174. ISBN 978-3-13-179525-0.
- ^ a b Becker KL, ed. (2001). "Endocrine Drugs and Values". Principles and Practice of Endocrinology and Metabolism. Lippincott Williams & Wilkins. pp. 2153–. ISBN 978-0-7817-1750-2.
- ^ a b Food and Drug Administration (2011). Approved Drug Products with Therapeutic Equivalence Evaluations - FDA Orange Book (31st ed.). DrugPatentWatch.com. pp. 586–. ISBN 978-1-934899-81-6.
- ^ "Drugs@FDA: FDA Approved Drug Products". United States Food and Drug Administration. Retrieved 26 July 2018.
- ^ Lobo RA (5 June 2007). Treatment of the Postmenopausal Woman: Basic and Clinical Aspects. Academic Press. pp. 177, 217–226, 770–771. ISBN 978-0-08-055309-2.
- ^ Falcone T, Hurd WW (14 June 2017). Clinical Reproductive Medicine and Surgery: A Practical Guide. Springer. pp. 179–. ISBN 978-3-319-52210-4.
- ^ Becker KL (2001). Principles and Practice of Endocrinology and Metabolism. Lippincott Williams & Wilkins. pp. 889, 1059–1060, 2153. ISBN 978-0-7817-1750-2.
- ^ Kleemann A, Engel J, Kutscher B, Reichert D (14 May 2014). Pharmaceutical Substances, 5th Edition, 2009: Syntheses, Patents and Applications of the most relevant APIs. Thieme. pp. 1167–1174. ISBN 978-3-13-179525-0.
- ^ Muller (19 June 1998). European Drug Index: European Drug Registrations, Fourth Edition. CRC Press. pp. 276, 454–455, 566–567. ISBN 978-3-7692-2114-5.
- ^ Krishna UR, Sheriar NK (1996). Menopause. Orient Blackswan. pp. 70–. ISBN 978-81-250-0910-8.
- ^ "NNR: Products Recently Accepted by the A. M. A. Council on Pharmacy and Chemistry". Journal of the American Pharmaceutical Association (Practical Pharmacy ed.). 10 (11): 692–694. 1949. doi:10.1016/S0095-9561(16)31995-8. ISSN 0095-9561.
- ^ "AERODIOL (Oestradiol hemihydrate 150 micrograms/actuation)" (PDF). Servier Laboratories (Aust) Pty Ltd.
- ^ "Estradiol". Drugs.com.
- ^ Sahin FK, Koken G, Cosar E, Arioz DT, Degirmenci B, Albayrak R, Acar M (2008). "Effect of Aerodiol administration on ocular arteries in postmenopausal women". Gynecol. Endocrinol. 24 (4): 173–7. doi:10.1080/09513590701807431. PMID 18382901.
300 μg 17β-estradiol (Aerodiol®; Servier, Chambrayles-Tours, France) was administered via the nasal route by a gynecologist. This product is unavailable after March 31, 2007 because its manufacturing and marketing are being discontinued.
- ^ Plouffe Jr L, Ravnikar VA, Speroff L, Watts NB (6 December 2012). Comprehensive Management of Menopause. Springer Science & Business Media. pp. 271–. ISBN 978-1-4612-4330-4.
- ^ University of California (1868-1952) (1952). Hospital Formulary and Compendium of Useful Information. University of California Press. pp. 49–. GGKEY:2UAAZRZ5LN0.
{{cite book}}: CS1 maint: numeric names: authors list (link) - ^ Leidenberger FA (17 April 2013). Klinische Endokrinologie für Frauenärzte. Springer-Verlag. pp. 527–. ISBN 978-3-662-08110-5.
- ^ a b c Lauritzen C (September 1990). "Clinical use of oestrogens and progestogens". Maturitas. 12 (3): 199–214. doi:10.1016/0378-5122(90)90004-P. PMID 2215269.
- ^ Lauritzen C (22 June 2005). "Practice of Hormone Substitution". In Lauritzen C, Studd JW (eds.). Current Management of the Menopause. CRC Press. pp. 95–98, 488. ISBN 978-0-203-48612-2.
- ^ Laurtizen C (2001). "Hormone Substitution Before, During and After Menopause" (PDF). In Fisch FH (ed.). Menopause – Andropause: Hormone Replacement Therapy Through the Ages. Krause & Pachernegg: Gablitz. pp. 67–88. ISBN 978-3-901299-34-6.
- ^ Midwinter A (1976). "Contraindications to estrogen therapy and management of the menopausal syndrome in these cases". In Campbell S (ed.). The Management of the Menopause & Post-Menopausal Years: The Proceedings of the International Symposium held in London 24–26 November 1975 Arranged by the Institute of Obstetrics and Gynaecology, The University of London. MTP Press Limited. pp. 377–382. doi:10.1007/978-94-011-6165-7_33. ISBN 978-94-011-6167-1.
- ^ McLiver B, Tebben PJ, Shah P (23 September 2010). "Endocrinology". In Ghosh AK (ed.). Mayo Clinic Internal Medicine Board Review. OUP USA. pp. 222–. ISBN 978-0-19-975569-1.
- ^ a b Bishop BM (December 2015). "Pharmacotherapy Considerations in the Management of Transgender Patients: A Brief Review". Pharmacotherapy. 35 (12): 1130–9. doi:10.1002/phar.1668. PMID 26684553. S2CID 37001563.
- ^ Cheng ZN, Shu Y, Liu ZQ, Wang LS, Ou-Yang DS, Zhou HH (February 2001). "Role of cytochrome P450 in estradiol metabolism in vitro". Acta Pharmacol. Sin. 22 (2): 148–54. PMID 11741520.
- ^ Dubey RK, Jackson EK, Gillespie DG, Zacharia LC, Imthurn B, Keller PJ (2000). "Clinically used estrogens differentially inhibit human aortic smooth muscle cell growth and mitogen-activated protein kinase activity". Arterioscler. Thromb. Vasc. Biol. 20 (4): 964–72. doi:10.1161/01.atv.20.4.964. PMID 10764660.
- ^ MacLusky NJ, Larner JM, Hochberg RB (January 1989). "Actions of an estradiol-17-fatty acid ester in estrogen target tissues of the rat: comparison with other C-17 metabolites and a pharmacological C-17 ester". Endocrinology. 124 (1): 318–24. doi:10.1210/endo-124-1-318. PMID 2909371.
- ^ a b c d e Elks J (14 November 2014). The Dictionary of Drugs: Chemical Data: Chemical Data, Structures and Bibliographies. Springer. pp. 898–. ISBN 978-1-4757-2085-3.
- ^ a b Kaunitz AM (December 2000). "Injectable contraception. New and existing options". Obstet. Gynecol. Clin. North Am. 27 (4): 741–80. doi:10.1016/S0889-8545(05)70171-6. PMID 11091987.
- ^ Sriram D, Yogiswari P (2007). "Steroids". Medicinal Chemistry. Pearson Education India. p. 427. ISBN 978-81-317-0031-0. Retrieved 20 May 2012.
- ^ a b Crabbé P, Archer S, Benagiano G, Diczfalusy E, Djerassi C, Fried J, Higuchi T (March 1983). "Long-acting contraceptive agents: design of the WHO Chemical Synthesis Programme". Steroids. 41 (3): 243–253. doi:10.1016/0039-128X(83)90095-8. PMID 6658872. S2CID 12896179.
- ^ a b c Rahimy MH, Ryan KK, Hopkins NK (October 1999). "Lunelle monthly contraceptive injection (medroxyprogesterone acetate and estradiol cypionate injectable suspension): steady-state pharmacokinetics of MPA and E2 in surgically sterile women". Contraception. 60 (4): 209–14. doi:10.1016/S0010-7824(99)00086-4. PMID 10640167.
- ^ a b c Rosenfield RL, Fang VS, Dupon C, Kim MH, Refetoff S (October 1973). "The effects of low doses of depot estradiol and testosterone in teenagers with ovarian failure and Turner's syndrome". J. Clin. Endocrinol. Metab. 37 (4): 574–80. doi:10.1210/jcem-37-4-574. PMID 4742538.
- ^ a b c Rosenfield RL, Fang VS (December 1974). "The effects of prolonged physiologic estradiol therapy on the maturation of hypogonadal teen-agers". J Pediatr. 85 (6): 830–7. doi:10.1016/s0022-3476(74)80355-0. PMID 4370903.
- ^ a b Garza-Flores J (April 1994). "Pharmacokinetics of once-a-month injectable contraceptives". Contraception. 49 (4): 347–59. doi:10.1016/0010-7824(94)90032-9. PMID 8013219.
- ^ a b Martins RS, Antunes NJ, Comerlatti G, Caraccio G, Moreno RA, Frecentese F, Caliendo G, De Nucci G (June 2019). "Quantification of estradiol cypionate in plasma by liquid chromatography coupled with tandem mass spectrometry: Application in a pharmacokinetic study in healthy female volunteers". J Pharm Biomed Anal. 170: 273–278. doi:10.1016/j.jpba.2019.03.053. PMID 30947128. S2CID 96433789.
- ^ a b c Schwartz MM, Soule SD (July 1955). "Estradiol 17-beta-cyclopentylpropionate, a long-acting estrogen". Am. J. Obstet. Gynecol. 70 (1): 44–50. doi:10.1016/0002-9378(55)90286-6. PMID 14388061.
- ^ "Estradiol cypionate | C26H36O3 | ChemSpider". www.chemspider.com.
- ^ Ott, A. C. (1952). U.S. Patent No. 2,611,773. Washington, DC: U.S. Patent and Trademark Office. https://patents.google.com/patent/US2611773A/en
- ^ Shearman AM, Vogel M, Mcgavack TH (October 1952). "Responses of the vaginal epithelium of postmenopausal women to single doses of estrogens". Journal of Gerontology. 7 (4): 549–554. doi:10.1093/geronj/7.4.549. PMID 13000121.
- ^ a b c Shearman AM, Mcgavack TH (July 1953). "A comparison of the influence of alpha-estradiol dipropionate and of estradiol cyclopentylpropionate on the vaginal mucosa of nonmenstruating and irregularly menstruating women". American Journal of Obstetrics and Gynecology. 66 (1): 178–181. doi:10.1016/0002-9378(53)90300-7. PMID 13057994.
- ^ a b c Robinson WW (October 1953). "Estradiol cyclopentylpropionate: a new, long-acting, injectable estrogen". The Journal of Clinical Endocrinology and Metabolism. 13 (10): 1279–1280. doi:10.1210/jcem-13-10-1279. PMID 13096552.
- ^ Soule SD, Yanow M (May 1953). "Estradiol 17-cyclopentyl-propionate". Missouri Medicine. 50 (5): 345–346. PMID 13072193.
- ^ Coutinho EM, Carlos de Souza J (March 1968). "Conception control by monthly injections of medroxyprogesterone suspension and long-acting oestrogen". Journal of Reproduction and Fertility. 15 (2): 209–214. doi:10.1530/jrf.0.0150209. PMID 5643482.
- ^ Duetsch LL (1969). Research and development, market power, and patent policy in ethical drugs. University of Wisconsin--Madison. p. 95.
- ^ Kaufman C (1933). "Die Behandlung der Amenorrhöe mit Hohen Dosen der Ovarialhormone". Klinische Wochenschrift. 12 (40): 1557–1562. doi:10.1007/BF01765673. ISSN 0023-2173. S2CID 25856898.
- ^ Buschbeck H (2009). "Neue Wege der Hormontherapie in der Gynäkologie" [New ways of hormonal therapy in gynecology]. Deutsche Medizinische Wochenschrift. 60 (11): 389–393. doi:10.1055/s-0028-1129842. ISSN 0012-0472. S2CID 72668930.
- ^ Biskind MS (1935). "Commercial Glandular Products". Journal of the American Medical Association. 105 (9): 667. doi:10.1001/jama.1935.92760350007009a. ISSN 0002-9955.
Progynon-B, Schering Corporation: This is crystalline hydroxyestrin benzoate obtained by hydrogenation of theelin and subsequent conversion to the benzoate. [...] Progynon-B is marketed in ampules containing 1 cc. of a sesame oil solution of hydroxyestrin benzoate of either 2,500, 5,000, 10,000 or 50,000 international units.
- ^ a b c Goldzieher JW, Goldzieher JW, Fotherby K (1994). Pharmacology of the contraceptive steroids. Raven Press. p. 164. ISBN 978-0-7817-0097-9.
- ^ "Drug Product Database online query". Health Canada. 25 April 2012. Retrieved 19 August 2022.
- ^ Sweetman SC, ed. (2009). "Sex hormones and their modulators". Martindale: The Complete Drug Reference (36th ed.). London: Pharmaceutical Press. p. 2082. ISBN 978-0-85369-840-1.
- ^ "Micromedex Products: Please Login". www.micromedexsolutions.com. Retrieved 21 April 2023.
- ^ IARC Working Group on the Evaluation of Carcinogenic Risks to Humans; International Agency for Research on Cancer (1 January 1999). Hormonal Contraception and Post-menopausal Hormonal Therapy (PDF). IARC. p. 65. ISBN 978-92-832-1272-0. Archived from the original (PDF) on 28 August 2021. Retrieved 24 February 2019.
- ^ Senanayake P, Potts M (2008). "Hormonal Contraception". Atlas of Contraception (Second ed.). CRC Press. p. 51. ISBN 9780203347324. Archived from the original on 2017-01-02.
- ^ IARC Working Group on the Evaluation of Carcinogenic Risks to Humans; World Health Organization; International Agency for Research on Cancer (2007). Combined Estrogen-progestogen Contraceptives and Combined Estrogen-progestogen Menopausal Therapy. World Health Organization. pp. 431–. ISBN 978-92-832-1291-1.
- ^ Klitsch M (1995). "Still waiting for the contraceptive revolution". Fam Plann Perspect. 27 (6): 246–53. doi:10.2307/2136177. JSTOR 2136177. PMID 8666089. S2CID 38860459.

![Estradiol levels after subcutaneous (s.c.) or intramuscular (i.m.) injection of 5 mg estradiol cypionate in aqueous suspension.[2] Assays were performed using enzyme immunoassay.[2] Source was Sierra-Ramírez et al. (2011).[2]](http://upload.wikimedia.org/wikipedia/commons/thumb/4/44/Estradiol_levels_after_a_single_subcutaneous_or_intramuscular_injection_of_5_mg_estradiol_cypionate.png/300px-Estradiol_levels_after_a_single_subcutaneous_or_intramuscular_injection_of_5_mg_estradiol_cypionate.png)
![Estradiol levels at steady state (after the 3rd injection) with intramuscular injections of aqueous suspensions of 5 mg estradiol cypionate per month in premenopausal women.[63][9] Assays were performed using enzyme immunoassay and LC-MS/MSTooltip liquid chromatography–tandem mass spectrometry.[63][9] Sources were Rahimy et al. (1999) and Thurman et al. (2013).[63][9]](http://upload.wikimedia.org/wikipedia/commons/thumb/c/c7/Estradiol_levels_at_steady_state_with_intramuscular_injections_of_5_mg_estradiol_cypionate_per_month_in_premenopausal_women.png/300px-Estradiol_levels_at_steady_state_with_intramuscular_injections_of_5_mg_estradiol_cypionate_per_month_in_premenopausal_women.png)
![Estradiol levels after a single intramuscular injection of 1.0 to 2.0 mg (average 1.67 mg) of estradiol cypionate in oil (Depo-Estradiol) in hypogonadal girls.[64][65] Assays were performed using radioimmunoassay with chromatographic separation.[64][65] Sources were Rosenfield et al. (1973, 1974).[64][65]](http://upload.wikimedia.org/wikipedia/commons/thumb/0/02/Estradiol_levels_after_a_single_intramuscular_injection_of_1.0_to_2.0-mg_estradiol_cypionate_in_hypogonadal_girls.png/300px-Estradiol_levels_after_a_single_intramuscular_injection_of_1.0_to_2.0-mg_estradiol_cypionate_in_hypogonadal_girls.png)
![Estradiol levels after single intramuscular injections of 5 mg of different estradiol esters in oil in premenopausal women.[11] Assays were performed using radioimmunoassay with chromatographic separation.[11] Source was Oriowo et al. (1980).[11]](http://upload.wikimedia.org/wikipedia/commons/thumb/3/33/Estradiol_levels_after_a_single_5_mg_intramuscular_injection_of_estradiol_esters.png/300px-Estradiol_levels_after_a_single_5_mg_intramuscular_injection_of_estradiol_esters.png)
![Simplified curves of estradiol levels after injection of different estradiol esters in oil solution in women.[66] Source was Garza-Flores (1994).[66]](http://upload.wikimedia.org/wikipedia/commons/thumb/8/8c/Idealized_curves_of_estradiol_levels_after_injection_of_different_estradiol_esters_in_women.png/293px-Idealized_curves_of_estradiol_levels_after_injection_of_different_estradiol_esters_in_women.png)
![Estradiol cypionate levels after a single injection of 5 mg microcrystalline estradiol cypionate in aqueous suspension in women.[67] Assays were performed using LC-MS/MSTooltip liquid chromatography–tandem mass spectrometry. Source was Martins et al. (2019).[67]](http://upload.wikimedia.org/wikipedia/commons/thumb/1/19/Estradiol_cypionate_levels_after_a_single_intramuscular_injection_of_5_mg_microcrystalline_estradiol_cypionate_in_aqueous_suspension_in_women.png/300px-Estradiol_cypionate_levels_after_a_single_intramuscular_injection_of_5_mg_microcrystalline_estradiol_cypionate_in_aqueous_suspension_in_women.png)
![Vaginal cornification with a single intramuscular injection of different estradiol esters in oil solution in women.[68] Source was Schwartz & Soule (1955).[68]](http://upload.wikimedia.org/wikipedia/commons/thumb/2/23/Vaginal_cornification_with_a_single_intramuscular_injection_of_different_estradiol_esters_in_women.png/300px-Vaginal_cornification_with_a_single_intramuscular_injection_of_different_estradiol_esters_in_women.png)