Etanercept
 | |
| Clinical data | |
|---|---|
| Trade names | Enbrel |
| Biosimilars | etanercept-szzs, etanercept-ykro, Benepali, Brenzys,[1] Erelzi,[2][3] Etacept, Etera,[4] Eticovo, Lifmior, Nepexto,[5] Rymti[6] |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a602013 |
| License data |
|
| Pregnancy category | |
| Routes of administration | Subcutaneous |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 58–76% (SC) |
| Elimination half-life | 70–132 hours |
| Identifiers | |
| CAS Number | |
| PubChem SID | |
| DrugBank | |
| ChemSpider |
|
| UNII | |
| KEGG | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.224.383 |
| Chemical and physical data | |
| Formula | C2224H3475N621O698S36 |
| Molar mass | 51235.07 g·mol−1 |
Etanercept, sold under the brand name Enbrel among others, is a biologic medical product that is used to treat autoimmune diseases by interfering with tumor necrosis factor (TNF), a soluble inflammatory cytokine, by acting as a TNF inhibitor. It has US Food and Drug Administration (FDA) approval to treat rheumatoid arthritis, juvenile idiopathic arthritis and psoriatic arthritis, plaque psoriasis and ankylosing spondylitis. Tumor necrosis factor alpha (TNFα) is the "master regulator" of the inflammatory (immune) response in many organ systems. Autoimmune diseases are caused by an overactive immune response. Etanercept has the potential to treat these diseases by inhibiting TNF-alpha.[17]
Etanercept is a fusion protein produced by recombinant DNA. It fuses the TNF receptor to the constant end of the IgG1 antibody. First, the developers isolated the DNA sequence that codes the human gene for soluble TNF receptor 2, which is a receptor that binds to tumor necrosis factor-alpha. Second, they isolated the DNA sequence that codes the human gene for the Fc end of immunoglobulin G1 (IgG1). Third, they linked the DNA for TNF receptor 2 to the DNA for IgG1 Fc. Finally, they expressed the linked DNA to produce a protein that links the protein for TNF receptor 2 to the protein for IgG1 Fc.[18]
The prototypic fusion protein was first synthesized and shown to be highly active and unusually stable as a modality for blockade of TNF in vivo in the early 1990s by Bruce A. Beutler, an academic researcher then at the University of Texas Southwestern Medical Center at Dallas, and his colleagues.[19][20]
These investigators also patented the protein,[21] selling all rights to its use to Immunex, a Seattle biotechnology company that was acquired by Amgen in 2002.[22]
It is a large molecule, with a molecular weight of 150 kDa, that binds to TNFα and decreases its role in disorders involving excess inflammation in humans and other animals, including autoimmune diseases such as ankylosing spondylitis,[23] juvenile rheumatoid arthritis, psoriasis, psoriatic arthritis, rheumatoid arthritis, and, potentially, in a variety of other disorders mediated by excess TNFα. It is on the World Health Organization's List of Essential Medicines.[24]
Medical uses[edit]
In the United States, etanercept is indicated for:
- Moderate to Severe Rheumatoid Arthritis (RA) (Nov 1998)[14][25]
- Moderate to Severe Polyarticular Juvenile Rheumatoid Arthritis (May 1999)[14][26]
- Psoriatic Arthritis (Jan 2002)[14][27]
- Ankylosing Spondylitis (AS) (July 2003)[14][28][29]
- Moderate to Severe Plaque Psoriasis (April 2004)[14][30]
In the European Union, etanercept is indicated to treat:
- moderate to severe active rheumatoid arthritis[15]
- severe, active and progressive rheumatoid arthritis[15]
- juvenile idiopathic arthritis[15]
- polyarthritis (rheumatoid-factor-positive or -negative) and extended oligoarthritis in children and adolescents[15]
- active and progressive psoriatic arthritis[15]
- enthesitis-related arthritis
- axial spondyloarthritis[15]
- severe active ankylosing spondylitis[15]
- severe non-radiographic axial spondyloarthritis[15]
- moderate to severe plaque psoriasis[15]
- chronic severe plaque psoriasis pediatric plaque psoriasis[15]
Unrecognized uses[edit]
An American physician, Edward Tobinick, has attempted to use etanercept to treat chronic neurological dysfunction after stroke and brain injury[31] and issued U.S.[32] and foreign patents. Writing for Science-Based Medicine, Steven Novella said that it was "unethical for physicians to practice outside of their area of competence and expertise". Tobinick sued Novella in response, and lost.[33] Of this treatment, the American Academy of Neurology advise "there is insufficient evidence to determine its effectiveness and that the treatment may be associated with adverse outcomes and high cost".[34]
Adverse effects[edit]
On May 2, 2008, the US Food and Drug Administration (FDA) placed a black box warning on etanercept due to a number of serious infections associated with the drug.[35] Serious infections and sepsis, including fatalities, have been reported with the use of etanercept including reactivation of latent tuberculosis and hepatitis B infections.[14][36]
Injection site reactions such as redness and pain are common, occurring in approximately 11.4% of cases.[37]
Mechanism of action[edit]
Etanercept reduces the effect of naturally present TNF, and hence is a TNF inhibitor, functioning as a decoy receptor that binds to TNF.[38]
Tumor necrosis factor-alpha (TNFα) is a cytokine produced by lymphocytes and macrophages, two types of white blood cells. It mediates the immune response by attracting additional white blood cells to sites of inflammation and through additional molecular mechanisms that initiate and amplify inflammation. Inhibition of its action by etanercept reduces the inflammatory response, which is especially useful for treating autoimmune diseases.
There are two types of TNF receptors: those found embedded in white blood cells that respond to TNF by releasing other cytokines, and soluble TNF receptors that are used to deactivate TNF and blunt the immune response. In addition, TNF receptors are found on the surface of virtually all nucleated cells (red blood cells, which are not nucleated, do not contain TNF receptors on their surface). Etanercept mimics the inhibitory effects of naturally occurring soluble TNF receptors, the difference being that etanercept, because it is a fusion protein rather than a simple TNF receptor, has a greatly extended half-life in the bloodstream, and therefore a more profound and long-lasting biologic effect than a naturally occurring soluble TNF receptor.[39]
Structure[edit]
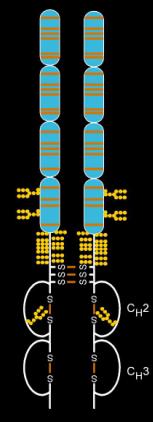
Etanercept is made from the combination of two naturally occurring soluble human 75-kilodalton TNF receptors linked to an Fc portion of an IgG1.[40] The effect is an artificially engineered dimeric fusion protein.[40] Etanercept is a complex molecule containing 6 N-glycans, up to 14 O-glycans and 29 disulfide bridge structures.[41][42][43]
History[edit]
The first etanercept-related patent was filed by Immunex on September 5, 1989.[44] The fusion protein was developed by Bruce A. Beutler, an academic researcher then at the University of Texas Southwestern Medical Center at Dallas, and colleagues, who patented it and licensed the rights in 1995 to Immunex.[45] Another patent on such fusion protein technology from Brian Seed at Massachusetts General Hospital was licensed to Immmunex in 1997.[46]
Etanercept was approved for use in the United States in November 1998.[14][47]
Etanercept was approved for use in the European Union in February 2000.[15]
Society and culture[edit]
Economics[edit]
The US retail price of etanercept has risen over time. In 2008, the cost of etanercept was $1,500 per month or $18,000 per year.[48] By 2011, the cost had exceeded $20,000 per year.[49][50] In 2013, a survey by the International Federation of Health Plans (IFHP) found that the average US cost for etanercept was $2,225 per month, or $26,700 per year.[51] The IFHP report also found wide variation in prices charged to various US health plans, between $1,946 per month at the 25th percentile and $4,006 per month at the 95th percentile.[51]
Etanercept is more expensive in the US than in other countries.[51] As of 2013, average monthly costs in surveyed nations ranged from $1,017 in Switzerland to $1,646 in Canada, compared to an average monthly cost of $2,225 per month in the US.[51]
Amgen sells etanercept within the US and Canada, while Pfizer, Inc. sells the drug outside of the US and Canada.[49] Sales within the US and Canada were $3.5 billion in 2010.[49] Sales of etanercept outside the US and Canada were $3.3 billion in 2010.[52]
Patents[edit]
The patent on etanercept was set to expire on October 23, 2012,[53] but, in the United States, a second patent, granting exclusivity for another 16 years, has been granted.[54]
Before the extension it seemed unlikely that a generic would have been available. As a biologic, etanercept is subject to different laws from those applicable to chemical formulations. Many countries do not permit the manufacture of generic biologics. However, the European Union and the United States (Biologics Price Competition and Innovation Act of 2009) do have in place a system to approve generic biologics (biosimilars) which "requires mandatory clinical testing and periodic review".[55]
In April 2013, the Indian pharma major Cipla made an announcement about launching the first biosimilar of Etanercept in India under the brand name Etacept for the treatment of rheumatic disorders.[56]
Biosimilars[edit]
In January 2016, Benepali was approved for use in the European Union.[57]
In February 2017, Lifmior was approved for use in the European Union.[58] It was withdrawn from the market in February 2020.[59]
In June 2017, Erelzi was approved for use in the European Union.[60]
In March 2019, YLB113 (Etanercept biosimilar by YL Biologics) was approved in Japan.[61]
In May 2020, Nepexto was approved for use in the European Union.[5]
Rymti and Etera were approved for medical use in Australia in October 2020.[7][62][6][4]
References[edit]
- ^ "Health Canada New Drug Authorizations: 2016 Highlights". Health Canada. 14 March 2017. Retrieved 7 April 2024.
- ^ "Arthritis". Health Canada. 8 May 2018. Retrieved 13 April 2024.
- ^ "Regulatory Decision Summary for Erelzi". Drug and Health Products Portal. 6 April 2017. Retrieved 13 April 2024.
- ^ a b c "Etera ARTG". Therapeutic Goods Administration (TGA). Archived from the original on 13 June 2021. Retrieved 12 June 2021.
- ^ a b c "Nepexto EPAR". European Medicines Agency. 24 March 2020. Retrieved 4 March 2023.
- ^ a b c "Rymti ARTG". Therapeutic Goods Administration (TGA). Archived from the original on 13 June 2021. Retrieved 12 June 2021.
- ^ a b "Australian Public Assessment Report for Etanercept" (PDF). Feb 2021.
- ^ "Etanercept Use During Pregnancy". Drugs.com. 24 January 2020. Retrieved 13 August 2020.
- ^ "FDA-sourced list of all drugs with black box warnings (Use Download Full Results and View Query links.)". nctr-crs.fda.gov. FDA. Retrieved 22 Oct 2023.
- ^ "Rymti Summary Basis of Decision". Health Canada. 23 October 2014. Retrieved 10 March 2023.
- ^ "Benepali 25 mg solution for injection in pre-filled syringe - Summary of Product Characteristics (SmPC)". (emc). 25 January 2021. Retrieved 12 June 2021.
- ^ "Enbrel 25mg solution for injection in pre-filled pen - Summary of Product Characteristics (SmPC)". (emc). 8 June 2021. Retrieved 12 June 2021.
- ^ "Erelzi 50 mg solution for injection in pre filled pen - Summary of Product Characteristics (SmPC)". (emc). 25 May 2021. Retrieved 12 June 2021.
- ^ a b c d e f g h "Enbrel- etanercept solution Enbrel- etanercept kit". DailyMed. Retrieved 17 April 2021.
- ^ a b c d e f g h i j k l "Enbrel EPAR". European Medicines Agency (EMA). 17 September 2018. Retrieved 2 April 2020. Text was copied from this source which is © European Medicines Agency. Reproduction is authorized provided the source is acknowledged.
- ^ "Nepexto Product information". Union Register of medicinal products. Retrieved 3 March 2023.
- ^ Feldmann M, Maini RN (October 2003). "Lasker Clinical Medical Research Award. TNF defined as a therapeutic target for rheumatoid arthritis and other autoimmune diseases". Nature Medicine. 9 (10): 1245–1250. doi:10.1038/nm939. PMID 14520364. S2CID 52860838.
- ^ "Drugs@FDA: FDA-Approved Drugs".
- ^ Peppel K, Crawford D, Beutler B (December 1991). "A tumor necrosis factor (TNF) receptor-IgG heavy chain chimeric protein as a bivalent antagonist of TNF activity". The Journal of Experimental Medicine. 174 (6): 1483–1489. doi:10.1084/jem.174.6.1483. PMC 2119031. PMID 1660525.
- ^ Peppel K, Poltorak A, Melhado I, Jirik F, Beutler B (November 1993). "Expression of a TNF inhibitor in transgenic mice". Journal of Immunology. 151 (10): 5699–5703. doi:10.4049/jimmunol.151.10.5699. PMID 7693816. S2CID 10859938.
- ^ U.S. Patent number: 5,447,851
- ^ "Arthritis Drug Effective for Depression in Psoriasis Sufferers". Archived from the original on 2007-10-20. Retrieved 2008-01-10.
- ^ Braun J, McHugh N, Singh A, Wajdula JS, Sato R (June 2007). "Improvement in patient-reported outcomes for patients with ankylosing spondylitis treated with etanercept 50 mg once-weekly and 25 mg twice-weekly". Rheumatology. 46 (6): 999–1004. doi:10.1093/rheumatology/kem069. PMID 17389658.
- ^ World Health Organization (2019). World Health Organization model list of essential medicines: 21st list 2019. Geneva: World Health Organization. hdl:10665/325771. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
- ^ Siegel YP (November 2, 1998). "Approval of Etanercept for treatment of rheumatoid arthritis" (PDF). Letter to Sally Gould. U.S. Food and Drug Administration (FDA). Retrieved April 14, 2015.
- ^ Weiss KD (May 27, 1999). "Approval of Etanercept for treatment of polyarticular course juvenile rheumatoid arthritis (JRA)" (PDF). Letter to Sally Gould. U.S. Food and Drug Administration (FDA). Retrieved April 14, 2015.
- ^ Weiss KD (January 15, 2002). "Approval of Etanercept for treatment of psoriatic arthritis". Letter to Sally Gould. U.S. Food and Drug Administration (FDA). Retrieved April 14, 2015.
- ^ Keegan P (July 24, 2003). "Approval of Etanercept for treatment of ankylosing spondylitis" (PDF). Letter to Douglas Hunt. U.S. Food and Drug Administration (FDA). Retrieved April 14, 2015.
- ^ Maxwell LJ, Zochling J, Boonen A, Singh JA, Veras MM, Tanjong Ghogomu E, et al. (April 2015). "TNF-alpha inhibitors for ankylosing spondylitis". The Cochrane Database of Systematic Reviews. 4 (4): CD005468. doi:10.1002/14651858.CD005468.pub2. PMID 25887212.
- ^ Walton M (April 30, 2004). "Approval of Etanercept for treatment of moderate to severe plaque psoriasis" (PDF). Letter to Douglas Hunt. U.S. Food and Drug Administration (FDA). Retrieved April 14, 2015.
- ^ "New hope for survivors of stroke and traumatic brain injury". Springer Select. October 31, 2012. Retrieved November 17, 2018.
- ^ US 8900583, Tobinick EL, "Methods for treatment of brain injury utilizing biologics", published 2014-12-02
- ^ "Another Free Speech Win In Libel Lawsuit Disguised As A Trademark Complaint". Above the Law. 2017-02-24. Retrieved 2022-06-27.
- ^ "Practice Advisory: Etanercept for Poststroke Disability" (pdf). American Academy of Neurology. 6 June 2016.
- ^ "Wyeth and Amgen heighten warning of life-threatening infections on skin drug Enbrel". Archived from the original on 2008-05-05. Retrieved 2008-05-02.
- ^ Safety Update on TNF- α Antagonists: Infliximab and Etanercept (PDF). U.S. Food and Drug Administration (FDA). pp. 13–14. Archived from the original (PDF) on 24 September 2015. Retrieved 20 December 2013.
- ^ Kim PJ, Lansang RP, Vender R (July 2023). "A Systematic Review and Meta-Analysis of Injection Site Reactions in Randomized-Controlled Trials of Biologic Injections". Journal of Cutaneous Medicine and Surgery. 27 (4): 358–367. doi:10.1177/12034754231188444. PMC 10486173. PMID 37533141.
- ^ Zalevsky J, Secher T, Ezhevsky SA, Janot L, Steed PM, O'Brien C, et al. (August 2007). "Dominant-negative inhibitors of soluble TNF attenuate experimental arthritis without suppressing innate immunity to infection". Journal of Immunology. 179 (3): 1872–1883. doi:10.4049/jimmunol.179.3.1872. PMID 17641054.
- ^ Madhusudan S, Muthuramalingam SR, Braybrooke JP, Wilner S, Kaur K, Han C, et al. (September 2005). "Study of etanercept, a tumor necrosis factor-alpha inhibitor, in recurrent ovarian cancer". Journal of Clinical Oncology. 23 (25): 5950–5959. doi:10.1200/JCO.2005.04.127. PMID 16135466.
- ^ a b Smola MG, Soyer HP, Scharnagl E (October 1991). "Surgical treatment of dermatofibrosarcoma protuberans. A retrospective study of 20 cases with review of literature". European Journal of Surgical Oncology. 17 (5): 447–453. PMID 1936291.
- ^ Houel S, Hilliard M, Yu YQ, McLoughlin N, Martin SM, Rudd PM, et al. (January 2014). "N- and O-glycosylation analysis of etanercept using liquid chromatography and quadrupole time-of-flight mass spectrometry equipped with electron-transfer dissociation functionality". Analytical Chemistry. 86 (1): 576–584. doi:10.1021/ac402726h. PMID 24308717.
- ^ Mukai Y, Nakamura T, Yoshikawa M, Yoshioka Y, Tsunoda S, Nakagawa S, et al. (November 2010). "Solution of the structure of the TNF-TNFR2 complex". Science Signaling. 3 (148): ra83. doi:10.1126/scisignal.2000954. PMID 21081755. S2CID 24226117.
- ^ Lamanna WC, Mayer RE, Rupprechter A, Fuchs M, Higel F, Fritsch C, et al. (June 2017). "The structure-function relationship of disulfide bonds in etanercept". Scientific Reports. 7 (1): 3951. Bibcode:2017NatSR...7.3951L. doi:10.1038/s41598-017-04320-5. PMC 5479810. PMID 28638112.
- ^ Norman P (January 2017). "Enbrel and etanercept biosimilars: a tale of two patent systems". Pharmaceutical Patent Analyst. 6 (1): 5–7. doi:10.4155/ppa-2016-0043. PMID 28201948.
- ^ Gardner J (November 1, 2021). "A three-decade monopoly: How Amgen built a patent thicket around its top-selling drug".
- ^ Kowalczyk L (March 24, 2002). "Lucrative Licensing Deals With Drug, Biotech Firms Are Raising Ethics Issues For Hospitals".
- ^ "Etanercept Product Approval Information - Licensing Action 12/2/98". U.S. Food and Drug Administration (FDA). 1 April 2016. Archived from the original on 18 January 2017. Retrieved 4 June 2020.
- ^ "What's behind the whopping price tags on the newest generation of drugs: The story behind the production of Enbrel, Amgen's popular rheumatoid arthritis drug, provides insights as to why bioengineered drugs are so expensive." Carol M. Ostrom, Seattle Times, August 18, 2008
- ^ a b c Pollock A (23 November 2011). "Amgen's New Enbrel Patent May Undercut Health Care Plan". The New York Times. Retrieved 10 March 2023.
- ^ "Co-pay hike a painful reality; Miracle drug monthly cost jumps from $42 to $600" Archived 2013-01-18 at archive.today, Margery Eagan, Boston Herald, November 3, 2011
- ^ a b c d "2013 Comparative Price Report" (PDF). International Federation of Health Plans. Archived from the original (PDF) on 22 October 2017. Retrieved 24 November 2017.
- ^ "Portions of the 2010 Financial Report". Sec.gov. Retrieved 2019-06-05.
- ^ "Patent Terms Extended Under 35 USC §156". Archived from the original on 2010-02-24. Retrieved 2009-12-09.
- ^ "New Amgen Enbrel patent could block biosimilars until 2028". 2011-11-25. Retrieved 2019-07-14.
- ^ Kaldre I (2008). "The Future of Generic Biologics: Should the United States "Follow-On" the European Pathway?". www.law.duke.edu. Retrieved 2019-06-05.
- ^ "Cipla - Home" (PDF). Cipla.com. Archived from the original (PDF) on 2013-05-01. Retrieved 2019-06-05.
- ^ "Benepali EPAR". European Medicines Agency (EMA). 17 September 2018. Retrieved 2 April 2020.
- ^ "Lifmior EPAR". European Medicines Agency (EMA). 17 September 2018. Retrieved 2 April 2020.
- ^ "Public statement on Lifmior: Withdrawal of the marketing authorisation in the European Union" (PDF). Retrieved 2 April 2020.
- ^ "Erelzi EPAR". European Medicines Agency (EMA). 17 September 2018. Retrieved 2 April 2020.
- ^ "Japanese approval for Lupin's etanercept biosimilar".
- ^ "AusPAR: Etanercept". Therapeutic Goods Administration (TGA). 25 February 2021. Retrieved 12 June 2021.
