Flash evaporation
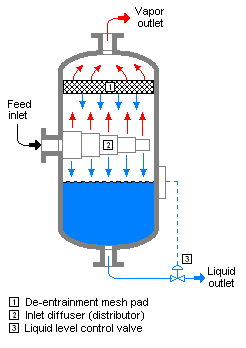
Flash evaporation (or partial evaporation) is the partial vapor that occurs when a saturated liquid stream undergoes a reduction in pressure by passing through a throttling valve or other throttling device. This process is one of the simplest unit operations. If the throttling valve or device is located at the entry into a pressure vessel so that the flash evaporation occurs within the vessel, then the vessel is often referred to as a flash drum.[1][2]
If the saturated liquid is a single-component liquid (for example, propane or liquid ammonia), a part of the liquid immediately "flashes" into vapor. Both the vapor and the residual liquid are cooled to the saturation temperature of the liquid at the reduced pressure. This is often referred to as "auto-refrigeration" and is the basis of most conventional vapor compression refrigeration systems.
If the saturated liquid is a multi-component liquid (for example, a mixture of propane, isobutane and normal butane), the flashed vapor is richer in the more volatile components than is the remaining liquid.
Uncontrolled flash evaporation can result in a boiling liquid expanding vapor explosion (BLEVE).
Flash evaporation of a single-component liquid[edit]
The flash evaporation of a single-component liquid is an isenthalpic process and is often referred to as an adiabatic flash. The following equation, derived from a simple heat balance around the throttling valve or device, is used to predict how much of a single-component liquid is vaporized.
where: = weight ratio of vaporized liquid / total mass = upstream liquid enthalpy at upstream temperature and pressure, J/kg
= flashed vapor enthalpy at downstream pressure and corresponding saturation
temperature, J/kg
= residual liquid enthalpy at downstream pressure and corresponding saturation
temperature, J/kg
If the enthalpy data required for the above equation is unavailable, then the following equation may be used.
where: = weight fraction vaporized = liquid specific heat at upstream temperature and pressure, J/(kg °C) = upstream liquid temperature, °C = liquid saturation temperature corresponding to the downstream pressure, °C
= liquid heat of vaporization at downstream pressure and corresponding saturation
temperature, J/kg
Here, the words "upstream" and "downstream" refer to before and after the liquid passes through the throttling valve or device.
This type of flash evaporation is used in the desalination of brackish water or ocean water by "Multi-Stage Flash Distillation." The water is heated and then routed into a reduced-pressure flash evaporation "stage" where some of the water flashes into steam. This steam is subsequently condensed into salt-free water. The residual salty liquid from that first stage is introduced into a second flash evaporation stage at a pressure lower than the first stage pressure. More water is flashed into steam which is also subsequently condensed into more salt-free water. This sequential use of multiple flash evaporation stages is continued until the design objectives of the system are met. A large part of the world's installed desalination capacity uses multi-stage flash distillation. Typically such plants have 24 or more sequential stages of flash evaporation.
Equilibrium flash of a multi-component liquid[edit]
The equilibrium flash of a multi-component liquid may be visualized as a simple distillation process using a single equilibrium stage. It is very different and more complex than the flash evaporation of single-component liquid. For a multi-component liquid, calculating the amounts of flashed vapor and residual liquid in equilibrium with each other at a given temperature and pressure requires a trial-and-error iterative solution. Such a calculation is commonly referred to as an equilibrium flash calculation. It involves solving the Rachford-Rice equation:[4][5][6][7]
where:
- zi is the mole fraction of component i in the feed liquid (assumed to be known);
- β is the fraction of feed that is vaporised;
- Ki is the equilibrium constant of component i.
The equilibrium constants Ki are in general functions of many parameters, though the most important is arguably temperature; they are defined as:
where:
- xi is the mole fraction of component i in liquid phase;
- yi is the mole fraction of component i in gas phase.
Once the Rachford-Rice equation has been solved for β, the compositions xi and yi can be immediately calculated as:
The Rachford-Rice equation can have multiple solutions for β, at most one of which guarantees that all xi and yi will be positive. In particular, if there is only one β for which:
then that β is the solution; if there are multiple such β's, it means that either Kmax<1 or Kmin>1, indicating respectively that no gas phase can be sustained (and therefore β=0) or conversely that no liquid phase can exist (and therefore β=1).
It is possible to use Newton's method for solving the above water equation, but there is a risk of converging to the wrong value of β; it is important to initialise the solver to a sensible initial value, such as (βmax+βmin)/2 (which is however not sufficient: Newton's method makes no guarantees on stability), or, alternatively, use a bracketing solver such as the bisection method or the Brent method, which are guaranteed to converge but can be slower.
The equilibrium flash of multi-component liquids is very widely utilized in petroleum refineries, petrochemical and chemical plants and natural gas processing plants.
Contrast with spray drying[edit]
Spray drying is sometimes seen as a form of flash evaporation. However, although it is a form of liquid evaporation, it is quite different from flash evaporation.
In spray drying, a slurry of very small solids is rapidly dried by suspension in a hot gas. The slurry is first atomized into very small liquid droplets which are then sprayed into a stream of hot dry air. The liquid rapidly evaporates leaving behind dry powder or dry solid granules. The dry powder or solid granules are recovered from the exhaust air by using cyclones, bag filters or electrostatic precipitators.
Natural flash evaporation[edit]
Natural flash vaporization or flash deposition may occur during earthquakes resulting in deposition of minerals held in supersaturated solutions, sometimes even valuable ore in the case of auriferous, gold-bearing, waters. This results when blocks of rock are rapidly pulled and pushed away from each other by jog faults.[8]
See also[edit]
References[edit]
- ^ Stanley M. Walas (1988). Chemical Process Equipment:Selection and Design. Butterworth-Heinemann. ISBN 0-409-90131-8.
- ^ Gas Processing Suppliers Association (GPSA) (1987). Engineering Data Book (10th Edition, Vol. 1 ed.). Gas Processing Suppliers Association, Tulsa, Oklahoma.
- ^ Vic Marshall; Steve Ruhemann (2001). Fundamentals of Process Safety. IChemE. p. 46. ISBN 9780852954317.
- ^ Harry Kooijman and Ross Taylor (2000). The ChemSep Book (PDF) (2nd ed.). ISBN 3-8311-1068-9. See page 186.
- ^ Analysis of Objective Functions (Pennsylvania State University)
- ^ Flash Calculations using the Soave-Redlich-Kwong equation of state (view full-size image)
- ^ Curtis H. Whitson, Michael L. Michelsen, The Negative Flash, Fluid Phase Equilibria, 53 (1989) 51–71.
- ^ Richard A. Lovett; Nature magazine (March 18, 2013). "Earthquakes Make Gold Veins in an Instant: Pressure changes cause the precious metal to deposit each time the crust moves, a new study finds. The insight suggests that remote sensing could be used to find new deposits in rocks where fault jogs are common". Scientific American. Retrieved March 18, 2013.
External links[edit]
- Vapor and Flash Steam Animation, photos and technical explanation of the difference between Flash Steam and Vaporized fraction.
- Flash Steam Tutorial The benefits of recovering flash steam, how it is done and typical applications.
- Water Desalination Technologies in the Middle East and Western Asia
- Discussion of spray drying
- Flash evaporation program online Flash distillation of the hydrocarbon compounds.