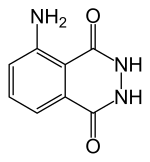
Luminol
| |
| |
| Names | |
|---|---|
| Preferred IUPAC name
5-Amino-2,3-dihydrophthalazine-1,4-dione | |
| Other names
5-Amino-2,3-dihydro-1,4-phthalazinedione
o-Aminophthaloyl hydrazide o-Aminophthalyl hydrazide 3-Aminophthalhydrazide 3-Aminophthalic hydrazide | |
| Identifiers | |
3D model (JSmol)
|
|
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.007.556 |
| EC Number |
|
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C8H7N3O2 | |
| Molar mass | 177.16 g/mol |
| Melting point | 319 °C (606 °F; 592 K) |
| Hazards | |
| NFPA 704 (fire diamond) | |
| Safety data sheet (SDS) | MSDS for luminol |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Luminol (C8H7N3O2) is a chemical that exhibits chemiluminescence, with a blue glow, when mixed with an appropriate oxidizing agent. Luminol is a white-to-pale-yellow crystalline solid that is soluble in most polar organic solvents, but insoluble in water.
Forensic investigators use luminol to detect trace amounts of blood at crime scenes, as it reacts with the iron in hemoglobin. Biologists use it in cellular assays to detect copper, iron, cyanides, as well as specific proteins via western blotting.[2]
When luminol is sprayed evenly across an area, trace amounts of an activating oxidant make the luminol emit a blue glow that can be seen in a darkened room. The glow only lasts about 30 seconds, but can be documented photographically. The glow is stronger in areas receiving more spray; the intensity of the glow does not indicate the amount of blood or other activator present.
Synthesis[edit]
Luminol is synthesized in a two-step process, beginning with 3-nitrophthalic acid.[3][4] First, hydrazine (N2H4) is heated with the 3-nitrophthalic acid in a high-boiling solvent such as triethylene glycol and glycerol. An acyl substitution condensation reaction occurs, with loss of water, forming 3-nitrophthalhydrazide. Reduction of the nitro group to an amino group with sodium dithionite (Na2S2O4), via a transient hydroxylamine intermediate, produces luminol.
The compound was first synthesized in Germany in 1902,[5] but was not named "luminol" until 1934.[3][6]
Chemiluminescence[edit]

To exhibit its luminescence, the luminol must be activated with an oxidant. Usually, a solution containing hydrogen peroxide (H2O2) and hydroxide ions in water is the activator. In the presence of a catalyst such as an iron or periodate compound, the hydrogen peroxide decomposes to form oxygen and water:
- 2 H2O2 → O2 + 2 H2O
- H2O2 + KIO4 → KIO3 + O2 + H2O
Laboratory settings often use potassium ferricyanide or potassium periodate for the catalyst. In the forensic detection of blood, the catalyst is the iron present in hemoglobin.[7] Enzymes in a variety of biological systems may also catalyse the decomposition of hydrogen peroxide.
The exact mechanism of luminol chemiluminescence is a complex multi-step reaction, especially in aqueous conditions. A recent theoretical investigation has been able to elucidate the reaction cascade as shown below.[8] Luminol is first deprotonated in basic conditions, then oxidized to the anionic radical. Which in turn has two paths available to give the key intermediate α-hydroxy- peroxide. After cyclization to the endoperoxide, the mono-anion will undergo decomposition without luminescence, if the pH is too low (< 8.2) for a second deprotonation. The endoperoxide dianion, however can give the retro-Diels-Alder product: 1,2-dioxane-3,6-dione dianion. And after chemiexcitation by two single-electron-transfers (SET) gives 3-aminophthalate dianion in its first singlet excited-state (S1). This highly instable molecule relaxes to the ground state, thereby emitting light of around 425 nm wavelength (purple-blue), the so-called chemiluminescence.
Use in criminal investigation[edit]
History[edit]
In 1928, German chemist H. O. Albrecht found that blood, among other substances, enhanced the luminescence of luminol in an alkaline solution of hydrogen peroxide.[9][10] In 1936, Karl Gleu and Karl Pfannstiel confirmed this enhancement in the presence of haematin, a component of blood.[11] In 1937, German forensic scientist Walter Specht made extensive studies of luminol's application to the detection of blood at crime scenes.[12] In 1939, San Francisco pathologists Frederick Proescher and A. M. Moody made three important observations about luminol:[13][14]
- although the test is presumptive, large areas of suspected material can be examined rapidly;
- dried and decomposed blood gave a stronger and more lasting reaction than fresh blood; and
- if the luminescence disappears, it may be reproduced by the application of a fresh luminol-hydrogen peroxide solution; dried bloodstains may thus be made luminescent repeatedly.
Theory[edit]
Crime scene investigators use luminol to find traces of blood, even if someone has cleaned or removed it. The investigator sprays a solution of luminol and the oxidant. The iron in blood catalyses the luminescence. The amount of catalyst necessary to cause the reaction is very small relative to the amount of luminol, allowing detection of even trace amounts of blood. The blue glow lasts for about 30 seconds per application. Detecting the glow requires a fairly dark room. Any glow detected may be documented by a long-exposure photograph.
Drawbacks[edit]
Luminol's use in a crime scene investigation is somewhat hampered by the fact that it reacts to iron- and copper-containing compounds,[15] bleaches, horseradish, fecal matter, or cigarette smoke residue.[14] Application of luminol to a piece of evidence may prevent other tests from being performed on it; however DNA has been successfully extracted from samples exposed to luminol.[16]
Related molecules[edit]
- Luminol: 5-amino-2,3-dihydro-1,4 ; 3-amino-phthalhydrazide ; 1,4-phthalazinedione, 5-amino-2,3-dihydro ; CAS: [521-31-3]
C8H7N3O2 – MW: 177.16
λabs (in 0.1 N NaOH) λmax 1 : 347 nm & λmax 2 : 300 nm; EC (at λmax 1): 7650 L/mol × cm
λabs / λem (MeOH): 355/413 nm - Luminol, sodium salt: sodium 3-amino-phthalhydrazide; CAS: [20666-12-0]
C8H6N3O2Na – MW: 199.12 - Luminol hemihydrate: a hydrate of sodium 3-amino-phthalhydrazide; CAS: [206658-90-4]
C8H6N3O2Na · H2O – MW: 217.16 - Luminol hydrochloride: 3-amino-phthalhydrazide hydrochloride; CAS: [74165-64-3]
C8H7N3O2 · HCl MW: 213.62 - Isoluminol: 4-aminophthalhydrazide; CAS: [3682-14-1]
C8H7N3O2 – MW: 117.16 (Xi) - Isoluminol monohydrate: 4-aminophthalhydrazide monohydrate
C8H7N3O2 · H2O – MW: 195.15 - Isoluminol ABEI: 4-aminophthalhydrazide monohydrate; CAS: [66612-29-1]
See also[edit]
References[edit]
- ^ Merck Index, 11th Edition, 5470.
- ^ Khan, Parvez; Idrees, Danish; MOxley, Michael A.; et al. (May 2014). "Luminol-Based Chemiluminescent Signals: Clinical and Non-clinical Application and Future Uses". Applied Biochemical Biotechnology. 173 (2): 333–355. doi:10.1007/s12010-014-0850-1. PMC 4426882. PMID 24752935.
- ^ a b Ernest Huntress; Lester Stanley; Almon Parker (1934). "The preparation of 3-aminophthalhydrazide for use in the demonstration of chemiluminescence". Journal of the American Chemical Society. 56 (1): 241–242. doi:10.1021/ja01316a077.
- ^ Synthesis of luminol
- ^ See:
- Aloys Josef Schmitz, "Ueber das Hydrazid der Trimesinsäure und der Hemimellithsäure" Archived 2015-01-02 at the Wayback Machine (On the hydrazide of trimesic acid [1,3,5-benzenetricarboxylic acid] and of hemimellitic acid [1,2,3-benzenetricarboxylic acid]), Inaugural Dissertation, Heidelberg University, 1902; pp. 17, 39–43. Schmitz calls luminol "1-amino-2,3-phtalsäurehydrazid".
- Note: Gill states that luminol was prepared as early as 1853. See: Steven K. Gill (1983) "New developments in chemiluminescence research," Aldrichimica Acta 16 (3) : 59–61; has footnote 2 on p. 60. Available at: Aldrichimica Acta Archived 2015-01-08 at the Wayback Machine. However, the sources Gill cites don't mention the preparation of luminol before 1902. Gill probably confused luminol with lophine (2,4,5-triphenyl-1H -imidazole), which the sources he cites do mention. Lophine is also chemiluminescent, and was first prepared in 1844 by Auguste Laurent. (See: Auguste Laurent (1844) "Sur un nouvel alcali organique, la lophine" (On a new organic alkali, lophine), Revue scientifique et industrielle, 18: 272–278.) The chemiluminescence of lophine was first observed by: Radziszewski, Bronisław L. (1877) "Untersuchungen über Hydrobenzamid, Amarin und Lophin" Archived 2015-12-14 at the Wayback Machine (Investigations of hydrobenzamide, amarine, and lophine), Berichte der Deutschen chemischen Gesellschaft zu Berlin, 10 : 70–75. In 1853, Ludwig Teichmann developed a test for blood, but it did not rely on chemiluminescence. See: L Teichmann (1853) "Ueber die Krystallisation der organischen Bestandtheile des Bluts" (On the crystallization of the organic components of blood), Zeitschrift für rationelle Medicin, new series, 3 : 375–388.
- ^ See also: Ernest H. Huntress, Lester N. Stanley, and Almon S. Parker (March 1934) "The oxidation of 3-aminophthalhydrazide ("luminol") as a lecture demonstration of chemiluminescence," Journal of Chemical Education, 11 (3) : 142–145. doi:10.1021/ed011p142
- ^ Ples, Marek. "Chemiluminescence activated by blood". www.weirdscience.eu (in Polish). Archived from the original on 3 January 2015. Retrieved 23 December 2014.
- ^ Yue, Ling; Liu, Yi-Tong (3 September 2020). "Mechanistic Insight into pH-Dependent Luminol Chemiluminescence in Aqueous Solution". The Journal of Physical Chemistry B. 124 (35): 7682–7693. doi:10.1021/acs.jpcb.0c06301. ISSN 1520-6106. PMID 32790377. S2CID 221125324.
- ^ H. O. Albrecht (1928) "Über die Chemiluminescenz des Aminophthalsäurehydrazids" (On the chemiluminescence of aminophthalic acid hydrazide) Zeitschrift für Physikalische Chemie 136: 321–330.
- ^ Stuart H. James and William G. Eckert, Interpretation of Bloodstain Evidence at Crime Scenes, 2nd ed. (Boca Raton, Florida: CRC Press LLC, 1998), page 154 Archived 2017-02-15 at the Wayback Machine.
- ^ Gleu, Karl; Pfannstiel, Karl (1936) "Über 3-aminophthalsaure-hydrazid" Journal für Praktische Chemie 146: 137–150.
- ^ Specht, Walter (1937) "Die Chemiluminescenz des Hämins, ein Hilfsmittel zur Auffindung und Erkennung forensisch wichtiger Blutspuren" (The chemiluminescence of haemin, an aid to the finding and recognition of forensically significant blood traces), Angewandte Chemie 50 (8): 155–157.
- ^ Proescher F. and Moody A.M. (1939) "Detection of blood by means of chemiluminescence," Journal of Laboratory and Clinical Medicine, 24 : 1183–1189.
- ^ a b James, Stuart; Kish, Paul E.; Sutton, Paulette Sutton (2005). Principles of Bloodstsain Pattern Analysis. Boca Raton, London, New York, Singapore: Taylor and Francis Group. p. 376. ISBN 0-8493-2014-3.
- ^ Ples, Marek. "Chemiluminescence of luminol activated by copper compound". www.weirdscience.eu (in Polish). Archived from the original on 3 January 2015. Retrieved 23 December 2014.
- ^ Technical note about Hemaglow Archived January 2, 2015, at the Wayback Machine


