RecBCD
| Exodeoxyribonuclease V | |||||||||
|---|---|---|---|---|---|---|---|---|---|
 | |||||||||
| Identifiers | |||||||||
| EC no. | 3.1.11.5 | ||||||||
| CAS no. | 37350-26-8 | ||||||||
| Databases | |||||||||
| IntEnz | IntEnz view | ||||||||
| BRENDA | BRENDA entry | ||||||||
| ExPASy | NiceZyme view | ||||||||
| KEGG | KEGG entry | ||||||||
| MetaCyc | metabolic pathway | ||||||||
| PRIAM | profile | ||||||||
| PDB structures | RCSB PDB PDBe PDBsum | ||||||||
| |||||||||
Exodeoxyribonuclease V (EC 3.1.11.5, RecBCD, Exonuclease V, Escherichia coli exonuclease V, E. coli exonuclease V, gene recBC endoenzyme, RecBC deoxyribonuclease, gene recBC DNase, gene recBCD enzymes) is an enzyme of E. coli that initiates recombinational repair from potentially lethal double strand breaks in DNA which may result from ionizing radiation, replication errors, endonucleases, oxidative damage, and a host of other factors.[2][3] The RecBCD enzyme is both a helicase that unwinds, or separates the strands of DNA, and a nuclease that makes single-stranded nicks in DNA.[1] It catalyses exonucleolytic cleavage (in the presence of ATP) in either 5′- to 3′- or 3′- to 5′-direction to yield 5′-phosphooligonucleotides.
Structure[edit]
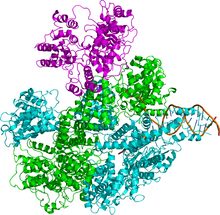
The enzyme complex is composed of three different subunits called RecB, RecC, and RecD and hence the complex is named RecBCD (Figure 1). Before the discovery of the recD gene,[4] the enzyme was known as “RecBC.” Each subunit is encoded by a separate gene:
| gene | chain | protein | function |
|---|---|---|---|
| RecB | β | P08394 | 3'-5' helicase, nuclease |
| RecC | γ | P07648 | Recognizes Chi (crossover hotspot instigator) |
| RecD | α | P04993 | 5'-3' helicase |
Function[edit]
Both the RecD and RecB subunits are helicases, i.e., energy-dependent molecular motors that unwind DNA (or RNA in the case of other proteins). The RecB subunit in addition has a nuclease function.[5] Finally, RecBCD enzyme (perhaps the RecC subunit) recognizes a specific sequence in DNA, 5'-GCTGGTGG-3', known as Chi (sometimes designated with the Greek letter χ).
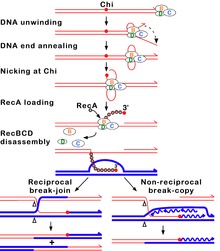
RecBCD is unusual amongst helicases because it has two helicases that travel with different rates[6] and because it can recognize and be altered by the Chi DNA sequence.[7][8] RecBCD avidly binds an end of linear double-stranded (ds) DNA. The RecD helicase travels on the strand with a 5' end at which the enzyme initiates unwinding, and RecB on the strand with a 3' end. RecB is slower than RecD, so that a single-stranded (ss) DNA loop accumulates ahead of RecB (Figure 2). This produces DNA structures with two ss tails (a shorter 3’ ended tail and a longer 5’ ended tail) and one ss loop (on the 3' ended strand) observed by electron microscopy.[9] The ss tails can anneal to produce a second ss loop complementary to the first one; such twin-loop structures were initially referred to as “rabbit ears.”
Mechanism of action[edit]

During unwinding the nuclease in RecB can act in different ways depending on the reaction conditions, notably the ratio of the concentrations of Mg2+ ions and ATP. (1) If ATP is in excess, the enzyme simply nicks the strand with Chi (the strand with the initial 3' end) (Figure 2).[10][11] Unwinding continues and produces a 3' ss tail with Chi near its terminus. This tail can be bound by RecA protein, which promotes strand exchange with an intact homologous DNA duplex.[12] When RecBCD reaches the end of the DNA, all three subunits disassemble and the enzyme remains inactive for an hour or more;[13] a RecBCD molecule that acted at Chi does not attack another DNA molecule. (2) If Mg2+ ions are in excess, RecBCD cleaves both DNA strands endonucleolytically, although the 5' tail is cleaved less often (Figure 3).[14] When RecBCD encounters a Chi site on the 3' ended strand, unwinding pauses and digestion of the 3' tail is reduced.[15] When RecBCD resumes unwinding, it now cleaves the opposite strand (i.e., the 5' tail)[16][17] and loads RecA protein onto the 3’-ended strand.[12] After completing reaction on one DNA molecule, the enzyme quickly attacks a second DNA, on which the same reactions occur as on the first DNA.
Although neither reaction has been verified by analysis of intracellular DNA, due to the transient nature of reaction intermediates, genetic evidence indicates that the first reaction more nearly mimics that in cells.[2] For example, the activity of Chi is influenced by nucleotides to its 3' side, both in cells and in reactions with ATP in excess but not with Mg2+ in excess [PMIDs 27401752, 27330137]. RecBCD mutants lacking detectable exonuclease activity retain high Chi hotspot activity in cells and nicking at Chi outside cells.[18] A Chi site on one DNA molecule in cells reduces or eliminates Chi activity on another DNA, perhaps reflecting the Chi-dependent disassembly of RecBCD observed in vitro under conditions of excess ATP and nicking of DNA at Chi.[19][20]
Under both reaction conditions, the 3' strand remains intact downstream of Chi. The RecA protein is then actively loaded onto the 3' tail by RecBCD.[12] At some undetermined point RecBCD dissociates from the DNA, although RecBCD can unwind at least 60 kb of DNA without falling off. RecA initiates exchange of the DNA strand to which it is bound with the identical, or nearly identical, strand in an intact DNA duplex; this strand exchange generates a joint DNA molecule, such as a D-loop (Figure 2). The joint DNA molecule is thought to be resolved either by replication primed by the invading 3’ ended strand containing Chi or by cleavage of the D-loop and formation of a Holliday junction. The Holliday junction can be resolved into linear DNA by the RuvABC complex or dissociated by the RecG protein. Each of these events can generate intact DNA with new combinations of genetic markers by which the parental DNAs may differ. This process, homologous recombination, completes the repair of the double-stranded DNA break.
Applications[edit]
RecBCD is a model enzyme for the use of single molecule fluorescence as an experimental technique used to better understand the function of protein-DNA interactions.[21] The enzyme is also useful in removing linear DNA, either single- or double-stranded, from preparations of circular double-stranded DNA, since it requires a DNA end for activity.
References[edit]
- ^ a b Singleton MR, Dillingham MS, Gaudier M, Kowalczykowski SC, Wigley DB (November 2004). "Crystal structure of RecBCD enzyme reveals a machine for processing DNA breaks". Nature. 432 (7014): 187–93. Bibcode:2004Natur.432..187S. doi:10.1038/nature02988. PMID 15538360. S2CID 2916995.
- ^ a b Smith, GR (June 2012). "How RecBCD Enzyme and Chi Promote DNA Break Repair and Recombination: a Molecular Biologist's View". Microbiol Mol Biol Rev. 76 (2): 217–28. doi:10.1128/MMBR.05026-11. PMC 3372252. PMID 22688812.
- ^ Spies M, Kowalczykowski SC (2003). "Homologous recombination by RecBCD and RecF pathways". In Higgins P (ed.). Bacterial Chromosomes. Washington, D.C: ASM Press. pp. 389–403. ISBN 1-55581-232-5.
- ^ Amundsen SK, Taylor AF, Chaudhury AM, Smith GR (August 1986). "recD: the gene for an essential third subunit of exonuclease V." Proc Natl Acad Sci U S A. 83 (15): 5558–62. Bibcode:1986PNAS...83.5558A. doi:10.1073/pnas.83.15.5558. PMC 386327. PMID 3526335.
- ^ Yu M, Souaya J, Julin DA (February 1998). "The 30-kDa C-terminal domain of the RecB protein is critical for the nuclease activity, but not the helicase activity, of the RecBCD enzyme from Escherichia coli". Proc Natl Acad Sci U S A. 95 (3): 981–6. Bibcode:1998PNAS...95..981Y. doi:10.1073/pnas.95.3.981. PMC 18645. PMID 9448271.
- ^ Taylor AF, Smith GR (June 2003). "RecBCD enzyme is a DNA helicase with fast and slow motors of opposite polarity". Nature. 423 (6942): 889–93. Bibcode:2003Natur.423..889T. doi:10.1038/nature01674. PMID 12815437. S2CID 4302346.
- ^ Taylor AF, Smith GR (June 1992). "RecBCD enzyme is altered upon cutting DNA at a Chi recombination hotspot". Proc Natl Acad Sci U S A. 89 (12): 5226–30. Bibcode:1992PNAS...89.5226T. doi:10.1073/pnas.89.12.5226. PMC 49264. PMID 1535156.
- ^ Amundsen SK, Taylor AF, Reddy M, Smith GR (December 2007). "Intersubunit signaling in RecBCD enzyme, a complex protein machine regulated by Chi hot spots". Genes Dev. 21 (24): 3296–307. doi:10.1101/gad.1605807. PMC 2113030. PMID 18079176.
- ^ Taylor A, Smith GR (November 1980). "Unwinding and rewinding of DNA by the RecBC enzyme". Cell. 22 (2 Pt 2): 447–57. doi:10.1016/0092-8674(80)90355-4. PMID 6256081. S2CID 22123298.
- ^ Ponticelli, A. S.; Schultz, D. W.; Taylor, A. F.; Smith, G. R. (May 1985). "Chi-dependent DNA strand cleavage by RecBC enzyme". Cell. 41 (1): 145–151. doi:10.1016/0092-8674(85)90069-8. PMID 3888404. S2CID 38361329.
- ^ Taylor AF, Schultz DW, Ponticelli AS, Smith GR (May 1985). "RecBC enzyme nicking at Chi sites during DNA unwinding: location and orientation-dependence of the cutting". Cell. 41 (1): 153–63. doi:10.1016/0092-8674(85)90070-4. PMID 3888405. S2CID 8551482.
- ^ a b c Anderson DG, Kowalczykowski SC (July 1997). "The translocating RecBCD enzyme stimulates recombination by directing RecA protein onto ssDNA in a Chi-regulated manner". Cell. 90 (1): 77–86. doi:10.1016/S0092-8674(00)80315-3. PMID 9230304.
- ^ Taylor AF, Smith GR (April 1999). "Regulation of homologous recombination: Chi inactivates RecBCD enzyme by disassembly of the three subunits". Genes Dev. 13 (7): 890–900. doi:10.1101/gad.13.7.890. PMC 316601. PMID 10197988.
- ^ Dixon DA, Kowalczykowski SC (April 1993). "The recombination hotspot Chi is a regulatory sequence that acts by attenuating the nuclease activity of the E. coli RecBCD enzyme". Cell. 73 (1): 87–96. doi:10.1016/0092-8674(93)90162-J. PMID 8384931. S2CID 19718817.
- ^ Spies M, Amitani I, Baskin RJ, Kowalczykowski SC (November 2007). "RecBCD enzyme switches lead motor subunits in response to Chi recognition". Cell. 131 (4): 694–705. doi:10.1016/j.cell.2007.09.023. PMC 2151923. PMID 18022364.
- ^ Taylor AF, Smith GR (October 1995). "Strand specificity of nicking of DNA at Chi sites by RecBCD enzyme. Modulation by ATP and magnesium levels". J Biol Chem. 270 (41): 24459–67. doi:10.1074/jbc.270.41.24459. PMID 7592661.
- ^ Anderson DG, Kowalczykowski SC (March 1997). "The recombination hot spot chi is a regulatory element that switches the polarity of DNA degradation by the RecBCD enzyme". Genes Dev. 11 (5): 571–81. doi:10.1101/gad.11.5.571. PMID 9119222.
- ^ Amundsen SK, Smith GR (January 2007). "Chi hotspot activity in Escherichia coli without RecBCD exonuclease activity: implications for the mechanism of recombination". Genetics. 175 (1): 41–54. doi:10.1534/genetics.106.065524. PMC 1774988. PMID 17110484.
- ^ Köppen A, Krobitsch S, Thoms B, Wackernagel W (July 1995). "Interaction with the recombination hot spot Chi in vivo converts the RecBCD enzyme of Escherichia coli into a Chi-independent recombinase by inactivation of the RecD subunit". Proc Natl Acad Sci U S A. 92 (14): 6249–53. Bibcode:1995PNAS...92.6249K. doi:10.1073/pnas.92.14.6249. PMC 41495. PMID 7541534.
- ^ Myers RS, Kuzminov A, Stahl FW (July 1995). "The recombination hot spot Chi activates RecBCD recombination by converting Escherichia coli to a recD mutant phenocopy". Proc Natl Acad Sci U S A. 92 (14): 6244–8. Bibcode:1995PNAS...92.6244M. doi:10.1073/pnas.92.14.6244. PMC 41494. PMID 7603978.
- ^ Bianco PR, Brewer LR, Corzett M, Balhorn R, Yeh Y, Kowalczykowski SC, Baskin RJ (January 2001). "Processive translocation and DNA unwinding by individual RecBCD enzyme molecules". Nature. 409 (6818): 374–8. Bibcode:2001Natur.409..374B. doi:10.1038/35053131. PMID 11201750. S2CID 4399125.
External links[edit]
- Exodeoxyribonuclease+V at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
- Exodeoxyribonuclease+V,+E+coli at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
- EC 3.1.11.5
