Transition metal benzyne complex

Transition metal benzyne complexes are organometallic complexes that contain benzyne ligands (C6H4). Unlike benzyne itself, these complexes are less reactive although they undergo a number of insertion reactions.[2]
Examples[edit]
The studies of metal-benzyne complexes were initiated with the preparation of zirconocene complex by reaction diphenylzirconocene with trimethylphosphine.[3]
- Cp2ZrPh2 + PMe3 → Cp2Zr(C6H4)(PMe3) + PhH
The preparation of Ta(η5-C5Me5)(C6H4)Me2 proceeds similarly, requiring the phenyl complex Ta(η5-C5Me5)(C6H5)Me3. This complex is prepared by treatment of Ta(η5-C5Me5)Me3Cl with phenyllithium.[4] Upon heating, this complex eliminates methane, leaving the benzyne complex:
- Ta(η5-C5Me5)(C6H5)Me3 → Ta(η5-C5Me5)(C6H4)Me2 + CH4
The second example of a benzyne complex is Ni(η2-C6H4)(dcpe) (dcpe = Cy2PCH2CH2PCy2). It is produced by dehalogenation of the bromophenyl complex NiCl(C6H4Br-2)(dcpe) with sodium amalgam. Its coordination geometry is close to trigonal planar.
Reactivity[edit]
Benzyne complexes react with a variety of electrophiles, resulting in insertion into one M-C bond.[5] With trifluoroacetic acid, benzene is lost to give the trifluoroacetate Ni(O2CF3)2(dcpe).[5]
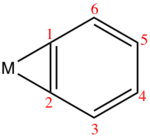
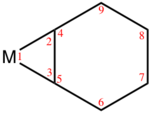
Structural trends[edit]
Several benzyne complexes have been examined by X-ray crystallography.
|
|
References[edit]
- ^ John F. Hartwig; Richard A. Andersen; Robert G. Bergman (1989). "Synthesis of a Highly Reactive (Benzyne)ruthenium Complex. Carbon-Carbon, Carbon-Hydrogen, Nitrogen-Hydrogen and Oxygen-Hydrogen Activation Reactions". J. Am. Chem. Soc. 111 (7): 2717–2719. doi:10.1021/ja00189a058.
- ^ a b c Buchwald, S. L.; Nielsen, R. B. "Group 4 Metal Complexes of Benzynes, Cycloalkynes, Acyclic Alkynes, and Alkenes" Chem. Rev. 1988, volume 88, 1047-1058
- ^ a b Buchwald, S. L.; Watson, B. T. The Trimethylphosphine Adduct of the Zirconocene-Benzyne Complex: Synthesis, Reactions, and X-ray Crystal Structure. J. Am. Chem. Soc. 1986, 108, 7411-7413
- ^ McLain, S. J.; Schrock, R. R.; Sharp, P. R.; Churchill, M. R.; Youngs, W. J. Synthesis of Monomeric Niobium- and Tantalum-Benzyne Complexes and Molecular Structure of Ta(η5-C5Me5)(C6H4)Me2" J. Am. Chem. Soc. 1979, volume 101, 263-265 doi:10.1021/ja00495a067
- ^ a b c d Bennett, M. A.; Hambley, T. W.; Roberts, N. K.; Robertson, G. B. (1985). "Synthesis and Single-Crystal X-ray Study of the Mononuclear η2-Benzyne (Dehydrobenzene) Nickel(0) Complex Ni(η2-C6H4)((C6H11)2PCH2CH2P(C6H11)2). Insertion Reactions with Simple Molecules and X-ray Crystal Structure of the Nickelaidan Complex Ni(CH2CH2C6H4-o)((C6H11)2PCH2CH2P(C6H11)2)"". Organometallics. 4: 1992–2000. doi:10.1021/om00130a012.
