Transition metal ether complex

In chemistry, a transition metal ether complex is a coordination complex consisting of a transition metal bonded to one or more ether ligand. The inventory of complexes is extensive.[2] Common ether ligands are diethyl ether and tetrahydrofuran. Common chelating ether ligands include the glymes, dimethoxyethane (dme) and diglyme, and the crown ethers. Being lipophilic, metal-ether complexes often exhibit solubility in organic solvents, a property of interest in synthetic chemistry. In contrast, the di-ether 1,4-dioxane is generally a bridging ligand.
Bonding, structure, reactions[edit]

Ethers are L-type ligands. They are σ-donors that exert weak field ligands. They resemble water ligands as seen in aquo complexes. They do not, however, readily participate in hydrogen bonding. The ether oxygen is nearly trigonal planar in its complexes.[4]
Being weakly basic, ether ligands tend to be easily displaceable. Otherwise, ether ligands are relatively unreactive. Cyclic ethers such as thf can ring-open or even deoxygenated when bound to highly electrophilic metal halides. Thus treatment of tungsten hexachloride with one equivalent of thf gives 1,4-dichlorobutane:[5]
- WCl6 + OC4H8 → WOCl4 + ClCH2CH2CH2CH2Cl
At higher concentrations of thf, polytetrahydrofuran is produced.
Examples[edit]
Homoleptic complexes[edit]
Ethers are relatively bulky ligands, thus homoleptic (i.e., all ligands being the same) ether complexes are uncommon. Examples often feature weakly coordinating anions such as BArF4− and Al(ORF)4−.
Metal halide complexes[edit]

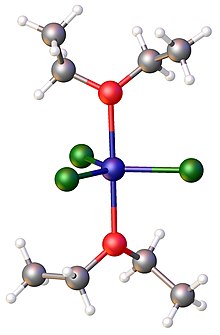
Metal chloride-tetrahydrofuran complexes are especially studied.[12] These compounds are often reagents because they are soluble in organic solvents as well as being anhydrous.
| Formula of metal-chloride-ether complexes |
Coordination sphere |
color |
|---|---|---|
| TiCl4(thf)2 | TiO2Cl4 | yellow[13] |
| TiCl3(thf)3 | TiO3Cl3 | blue[12] |
| [TiCl3(thf)2]2 | TiO2Cl4 | green[14] |
| ZrCl4(thf)2 | ZrO2Cl4 | white[12] |
| HfCl4(thf)2 | HfO2Cl4 | white[12] |
| VCl3(thf)3 | VO3Cl3 | pink[12] |
| VCl3(thf)2]2 | VO2Cl4 | red[14] |
| NbCl4(thf)2 | NbO2Cl4 | yellow[15] |
| Ta3Cl9(thf)4 | TaO2Cl4 and TaOCl5[15] | |
| CrCl3(thf)3 | CrO3Cl3 | pink[12][16] |
| MoCl4(thf)2 | MoO2Cl4 | pink[17] |
| MoCl4(Et2O)2 | MoO2Cl4 | beige[18] |
| MoCl3(thf)3 | MoO3Cl3 | red[17] |
| WCl4(Et2O)2 | WO2Cl4 | yellow[19] |
| MnCl3(thf)3 | MnO3Cl3 | brown-purple[20] |
| TcCl4(thf)2 | TcO2Cl4 | yellow[13] |
| ReCl4(thf)2 | ReO2Cl4 | green[21] |
| Fe4Cl8(thf)6 | FeO2Cl3, FeO2Cl4 | brown[22] |
| FeCl3(thf)2 | FeO2Cl3[23] | the related diethyl ether complex is brown |
| FeCl3(OEt2)2 | FeO2Cl3 | brown[11] |
| Co4Cl8(thf)6 | CoO2Cl3, CoO2Cl4 | blue[24] |
| [CoCl2(dme)]2 | CoO2Cl3[25] | |
| NiCl2(dimethoxyethane)2 | NiCl2O4 | yellow[26] |
| [Cu2Cl4(thf)3]n | CuO2Cl4, CuOCl4 | orange[27] |
| ZnCl2(thf)2 | ZnO2Cl2 | white[16] |
Metal carbonyl complexes[edit]
- M(CO)5(thf) (M = Cr, Mo, W)[4]
- Mo(CO)3(diglyme)
References[edit]
- ^ Duraj, S. A.; Towns; Baker; Schupp, J. (1990). "Structure of cis-Tetrachlorobis(tetrahydrofuran)hafnium(IV)". Acta Crystallographica. C46 (5): 890–2. doi:10.1107/S010827018901382X.
- ^ Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. ISBN 978-0-08-037941-8.
- ^ Duan, Zhiming; Zhang, Yan; Zhang, Bin; Zhu, Daoben (2009). "Crystal-to-Crystal Transformation from Antiferromagnetic Chains into a Ferromagnetic Diamondoid Framework". Journal of the American Chemical Society. 131 (20): 6934–6935. doi:10.1021/ja902101x. PMID 19402661.
- ^ a b Schubert, U.; Friedrich, P.; Orama, O. (1978). "Kristall- und molekülstruktur von pentacarbonyltetrahydrofuran-chrom(0)". Journal of Organometallic Chemistry. 144 (2): 175–179. doi:10.1016/S0022-328X(00)84160-1.
- ^ Bianchi, Sabrina; Bortoluzzi, Marco; Castelvetro, Valter; Marchetti, Fabio; Pampaloni, Guido; Pinzino, Calogero; Zacchini, Stefano (2016). "The reactivity of tungsten hexachloride with tetrahydrofuran and 2-methoxyethanol". Polyhedron. 117: 769–776. doi:10.1016/j.poly.2016.07.024. hdl:11568/803763.
- ^ Choukroun, Robert; Moumboko, Pierre; Chevalier, Sandrine; Etienne, Michel; Donnadieu, Bruno (1998). "Cationic Homoleptic Vanadium(II), (IV), and (V) Complexes Arising from Protonolysis of [V(NEt2)4]". Angewandte Chemie International Edition. 37 (22): 3169–3172. doi:10.1002/(SICI)1521-3773(19981204)37:22<3169::AID-ANIE3169>3.0.CO;2-0. PMID 29711321.
- ^ Kong, Gapgoung; Harakas, George N.; Whittlesey, Bruce R. (1995). "An Unusual Transition Metal Cluster Containing a Seven Metal Atom Plane. Syntheses and Crystal Structures of [Mn][Mn7(THF)6(CO)12]2, Mn3(THF)2(CO)10, and [Mn(THF)6][Mn(CO)5]2". Journal of the American Chemical Society. 117 (12): 3502–3509. doi:10.1021/ja00117a019.
- ^ Lichtenberg, Crispin; Adelhardt, Mario; Wörle, Michael; Büttner, Torsten; Meyer, Karsten; Grützmacher, Hansjörg (2015). "Mono- and Dinuclear Neutral and Cationic Iron(II) Compounds Supported by an Amidinato-diolefin Ligand: Characterization and Catalytic Application". Organometallics. 34 (12): 3079–3089. doi:10.1021/acs.organomet.5b00395.
- ^ Schwab, Miriam M.; Himmel, Daniel; Kacprzak, Sylwia; Radtke, Valentin; Kratzert, Daniel; Yassine, Zeinab; Weis, Philippe; Weber, Stefan; Krossing, Ingo (2018). "Reactivity of [Ni(cod)2][Al(ORF)4] towards Small Molecules and Elements". Zeitschrift für Anorganische und Allgemeine Chemie. 644: 50–57. doi:10.1002/zaac.201700367.
- ^ Crochet, Aurélien; Fromm, Katharina M. (2010). "Polyether Adducts of d-Block Metal Compounds as Starting Materials for New Cluster Compounds" (PDF). Zeitschrift für anorganische und allgemeine Chemie. 636 (8): 1484–1496. doi:10.1002/zaac.201000022.
- ^ a b Spandl, Johann; Kusserow, M.; Brüdgam, I. (2003). "Alkoxo-Verbindungen des dreiwertigen Eisen: Synthese und Charakterisierung von [Fe2(Ot Bu)6], [Fe2Cl2(Ot Bu)4], [Fe2Cl4(Ot Bu)2] und [N(n Bu)4]2[Fe6OCl6(OMe)12]". Zeitschrift für anorganische und allgemeine Chemie. 629 (6): 968–974. doi:10.1002/zaac.200300008.
- ^ a b c d e f Manzer, L. E. (1982). Tetrahydrofuran Complexes of Selected Early Transition Metals. Inorganic Syntheses. Vol. 21. pp. 135–140. doi:10.1002/9780470132524.ch31.
- ^ a b Hagenbach, Adelheid; Yegen, Eda; Abram, Ulrich (2006). "Technetium Tetrachloride as a Precursor for Small Technetium(IV) Complexes". Inorganic Chemistry. 45 (18): 7331–7338. doi:10.1021/ic060896u. PMID 16933935.
- ^ a b Sobota, Piotr; Ejfler, Jolanta; Szafert, Sławomir; Szczegot, Krzysztof; Sawka-Dobrowolska, Wanda (1993). "New intermediates for the Synthesis of Olefin Polymerization Catalysts: The Complexes [M2(μ-Cl)2Cl4(THF)4] (M = Ti or V, THF = Tetrahydrofuran); Crystal Structures and Properties". Journal of the Chemical Society, Dalton Transactions (15): 2353–2357. doi:10.1039/dt9930002353.
- ^ a b Babaian-Kibala, Elizabeth; Cotton, F. Albert; Shang, Maoyu (1990). "New Synthetic Routes for the Preparation of Niobium(III) and Tantalum(III) Triangular Cluster Compounds". Inorganic Chemistry. 29 (26): 5148–5156. doi:10.1021/ic00351a005.
- ^ a b Boudjouk, Philip; So, Jeung-Ho (1992). "Solvated and Unsolvated Anhydrous Metal Chlorides from Metal Chloride Hydrates". Inorganic Syntheses. Inorganic Syntheses. Vol. 29. pp. 108–111. doi:10.1002/9780470132609.ch26. ISBN 9780470132609.
- ^ a b Dilworth, Jonathan R.; Richards, Raymond L. (1990). "The Synthesis of Molybdenum and Tungsten Dinitrogen Complexes". Inorganic Syntheses. Inorganic Syntheses. Vol. 28. pp. 33–43. doi:10.1002/9780470132593.ch7. ISBN 9780470132593.
- ^ Maria, Sébastien; Poli, Rinaldo (2014). "Ether Complexes of Molybdenum(III) and Molybdenum(IV) chlorides". Inorganic Syntheses: Volume 36 (PDF). Inorganic Syntheses. pp. 15–18. doi:10.1002/9781118744994.ch03. ISBN 9781118744994.
- ^ Schäfer, André (2021). "The Decades-Old Mystery of Bis(diethyl ether)tungsten(IV) Chloride Solved". Acta Crystallographica Section C Structural Chemistry. 77 (4): 167–168. doi:10.1107/S2053229621002461. PMC 8020884. PMID 33818437.
- ^ Nachtigall, Olaf; Pataki, Astrid; Molski, Matthias; Lentz, Dieter; Spandl, Johann (2015). "Solvates of Manganese Trichloride Revisited - Synthesis, Isolation, and Crystal Structure of MnCl3(THF)3". Zeitschrift für Anorganische und Allgemeine Chemie. 641 (6): 1164–1168. doi:10.1002/zaac.201500106.
- ^ Swidersky, H.-W.; Pebler, J.; Dehnicke, K.; Fenske, D. (1990). "Technetium Tetrachloride as a Precursor for Small Technetium(IV) Complexes". Inorganic Chemistry. 45 (18): 7331–8. doi:10.1021/ic060896u. PMID 16933935.
- ^ Cotton, F. Albert; Luck, Rudy L.; Son, Kyung-Ae (1991). "New polynuclear compounds of iron(II) chloride with oxygen donor ligands Part I. Fe4Cl8(THF)6: Synthesis and a single crystal X-ray structure determination". Inorganica Chimica Acta. 179: 11–15. doi:10.1016/S0020-1693(00)85366-9.
- ^ L. V. Ivakina, N. R. Strel'tsova, V. K. Bel'skii, P. A. Storozhenko, B. M. Bulychev, A. B. Tarasov (1987). Zh.Obshch.Khim. (Russ.J.Gen.Chem.). 57: 1600.
{{cite journal}}: Missing or empty|title=(help)CS1 maint: multiple names: authors list (link) - ^ Sobota, Piotr; Olejnik, Zofia; Utko, Józef; Lis, Tadeusz (1993). "Synthesis, Magnetic Properties and Structure of the [Co4(μ3-Cl)2(μ2-Cl)4Cl2(THF)6] Complex". Polyhedron. 12 (6): 613–616. doi:10.1016/S0277-5387(00)84976-1.
- ^ Petriček, Saša (2011). "Octahedral and Tetrahedral Cobalt(II) Sites in Cobalt Chloride Complexes with Polyethers". Croatica Chemica Acta: 515–520. doi:10.5562/cca1747.
- ^ Ward, Laird G. L. (1972). "Anhydrous Nickel(II) Halides and their Tetrakis(ethanol) and 1,2-Dimethoxyethane Complexes". Inorganic Syntheses. Inorganic Syntheses. Vol. 13. pp. 154–164. doi:10.1002/9780470132449.ch30. ISBN 9780470132449.
- ^ Becker, Sabine; Dürr, Maximilian; Miska, Andreas; Becker, Jonathan; Gawlig, Christopher; Behrens, Ulrich; Ivanović-Burmazović, Ivana; Schindler, Siegfried (2016). "Copper Chloride Catalysis: Do μ4-Oxido Copper Clusters Play a Significant Role?". Inorganic Chemistry. 55 (8): 3759–3766. doi:10.1021/acs.inorgchem.5b02576. PMID 27045752.
