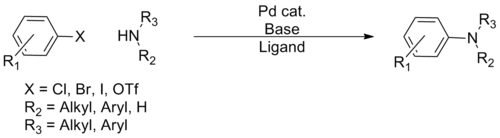Cross-coupling reaction
In organic chemistry, a cross-coupling reaction is a reaction where two different fragments are joined. Cross-couplings are a subset of the more general coupling reactions. Often cross-coupling reactions require metal catalysts. One important reaction type is this:
- R−M + R'−X → R−R' + MX (R, R' = organic fragments, usually aryl; M = main group center such as Li or MgX; X = halide)
These reactions are used to form carbon–carbon bonds but also carbon-heteroatom bonds.[1][2][3][4] Cross-coupling reaction are a subset of coupling reactions.
Richard F. Heck, Ei-ichi Negishi, and Akira Suzuki were awarded the 2010 Nobel Prize in Chemistry for developing palladium-catalyzed coupling reactions.[5][6]
Mechanism[edit]
Many mechanisms exist reflecting the myriad types of cross-couplings, including those that do not require metal catalysts.[7] Often, however, cross-coupling refers to a metal-catalyzed reaction of a nucleophilic partner with an electrophilic partner.

In such cases, the mechanism generally involves reductive elimination of R-R' from LnMR(R') (L = spectator ligand). This intermediate LnMR(R') is formed in a two step process from a low valence precursor LnM. The oxidative addition of an organic halide (RX) to LnM gives LnMR(X). Subsequently, the second partner undergoes transmetallation with a source of R'−. The final step is reductive elimination of the two coupling fragments to regenerate the catalyst and give the organic product. Unsaturated substrates, such as C(sp)−X and C(sp2)−X bonds, couple more easily, in part because they add readily to the catalyst.
Catalysts[edit]

Catalysts are often based on palladium, which is frequently selected due to high functional group tolerance. Organopalladium compounds are generally stable towards water and air. Palladium catalysts can be problematic for the pharmaceutical industry, which faces extensive regulation regarding heavy metals. Many pharmaceutical chemists attempt to use coupling reactions early in production to minimize metal traces in the product.[8] Heterogeneous catalysts based on Pd are also well developed.[9]
Copper-based catalysts are also common, especially for coupling involving heteroatom-C bonds.[10][11]
Iron-,[12] cobalt-,[13] and nickel-based[14] catalysts have been investigated.
Leaving groups[edit]
The leaving group X in the organic partner is usually a halide, although triflate, tosylate and other pseudohalide have been used. Chloride is an ideal group due to the low cost of organochlorine compounds. Frequently, however, C–Cl bonds are too inert, and bromide or iodide leaving groups are required for acceptable rates. The main group metal in the organometallic partner usually is an electropositive element such as tin, zinc, silicon, or boron.
Carbon–carbon cross-coupling[edit]
Many cross-couplings entail forming carbon–carbon bonds.
| Reaction | Year | Reactant A | Reactant B | Catalyst | Remark | ||
|---|---|---|---|---|---|---|---|
| Cadiot–Chodkiewicz coupling | 1957 | RC≡CH | sp | RC≡CX | sp | Cu | requires base |
| Castro–Stephens coupling | 1963 | RC≡CH | sp | Ar-X | sp2 | Cu | |
| Corey–House synthesis | 1967 | R2CuLi or RMgX | sp3 | R-X | sp2, sp3 | Cu | Cu-catalyzed version by Kochi, 1971 |
| Kumada coupling | 1972 | RMgBr | sp2, sp3 | R-X | sp2 | Pd or Ni or Fe | |
| Heck reaction | 1972 | alkene | sp2 | Ar-X | sp2 | Pd or Ni | requires base |
| Sonogashira coupling | 1975 | ArC≡CH | sp | R-X | sp3 sp2 | Pd and Cu | requires base |
| Negishi coupling | 1977 | R-Zn-X | sp3, sp2, sp | R-X | sp3 sp2 | Pd or Ni | |
| Stille cross coupling | 1978 | R-SnR3 | sp3, sp2, sp | R-X | sp3 sp2 | Pd or Ni | |
| Suzuki reaction | 1979 | R-B(OR)2 | sp2 | R-X | sp3 sp2 | Pd or Ni | requires base |
| Murahashi coupling[15] | 1979 | R-Li | sp2, sp3 | R-X | sp2 | Pd or Ru | |
| Hiyama coupling | 1988 | R-SiR3 | sp2 | R-X | sp3 sp2 | Pd | requires base |
| Fukuyama coupling | 1998 | R-Zn-I | sp3 | RCO(SEt) | sp2 | Pd or Ni | see Liebeskind–Srogl coupling, gives ketones |
| Liebeskind–Srogl coupling | 2000 | R-B(OR)2 | sp3, sp2 | RCO(SEt) Ar-SMe | sp2 | Pd | requires CuTC, gives ketones |
| Cross dehydrogenative coupling | 2004 | R-H | sp, sp2, sp3 | R'-H | sp, sp2, sp3 | Cu, Fe, Pd etc. | requires oxidant or dehydrogenation |
| Decarboxylative cross-coupling | 2000s | R-CO2H | sp2 | R'-X | sp, sp2 | Cu, Pd | Requires little-to-no base |
The restrictions on carbon atom geometry mainly inhibit β-hydride elimination when complexed to the catalyst.[16]
Carbon–heteroatom coupling[edit]
Many cross-couplings entail forming carbon–heteroatom bonds (heteroatom = S, N, O). A popular method is the Buchwald–Hartwig reaction:
-
(Eq.1)
| Reaction | Year | Reactant A | Reactant B | Catalyst | Remark | ||
|---|---|---|---|---|---|---|---|
| Ullmann-type reaction | 1905 | ArO-MM, ArNH2,RS-M,NC-M | sp3 | Ar-X (X = OAr, N(H)Ar, SR, CN) | sp2 | Cu | |
| Buchwald–Hartwig reaction[17] | 1994 | R2N-H | sp3 | R-X | sp2 | Pd | N-C coupling, second generation free amine |
| Chan–Lam coupling[18] | 1998 | Ar-B(OR)2 | sp2 | Ar-NH2 | sp2 | Cu | |
Miscellaneous reactions[edit]
Palladium-catalyzes the cross-coupling of aryl halides with fluorinated arene. The process is unusual in that it involves C–H functionalisation at an electron deficient arene.[19]

Applications[edit]
Cross-coupling reactions are important for the production of pharmaceuticals,[4] examples being montelukast, eletriptan, naproxen, varenicline, and resveratrol.[20] with Suzuki coupling being most widely used.[21] Some polymers and monomers are also prepared in this way.[22]
Reviews[edit]
- Fortman, George C.; Nolan, Steven P. (2011). "N-Heterocyclic carbene (NHC) ligands and palladium in homogeneous cross-coupling catalysis: a perfect union". Chemical Society Reviews. 40 (10): 5151–69. doi:10.1039/c1cs15088j. PMID 21731956.
- Yin; Liebscher, Jürgen (2007). "Carbon−Carbon Coupling Reactions Catalyzed by Heterogeneous Palladium Catalysts". Chemical Reviews. 107 (1): 133–173. doi:10.1021/cr0505674. PMID 17212474. S2CID 36974481.
- Jana, Ranjan; Pathak, Tejas P.; Sigman, Matthew S. (2011). "Advances in Transition Metal (Pd,Ni,Fe)-Catalyzed Cross-Coupling Reactions Using Alkyl-organometallics as Reaction Partners". Chemical Reviews. 111 (3): 1417–1492. doi:10.1021/cr100327p. PMC 3075866. PMID 21319862.
- Molnár, Árpád (2011). "Efficient, Selective, and Recyclable Palladium Catalysts in Carbon−Carbon Coupling Reactions". Chemical Reviews. 111 (3): 2251–2320. doi:10.1021/cr100355b. PMID 21391571.
- Miyaura, Norio; Suzuki, Akira (1995). "Palladium-Catalyzed Cross-Coupling Reactions of Organoboron Compounds". Chemical Reviews. 95 (7): 2457–2483. CiteSeerX 10.1.1.735.7660. doi:10.1021/cr00039a007.
- Roglans, Anna; Pla-Quintana, Anna; Moreno-Mañas, Marcial (2006). "Diazonium Salts as Substrates in Palladium-Catalyzed Cross-Coupling Reactions". Chemical Reviews. 106 (11): 4622–4643. doi:10.1021/cr0509861. PMID 17091930. S2CID 8128630.
- Korch, Katerina M.; Watson, Donald A. (2019). "Cross-Coupling of Heteroatomic Electrophiles". Chemical Reviews. 119 (13): 8192–8228. doi:10.1021/acs.chemrev.8b00628. PMC 6620169. PMID 31184483.
- Cahiez, Gérard; Moyeux, Alban (2010). "Cobalt-Catalyzed Cross-Coupling Reactions". Chemical Reviews. 110 (3): 1435–1462. doi:10.1021/cr9000786. PMID 20148539.
- Yi, Hong; Zhang, Guoting; Wang, Huamin; Huang, Zhiyuan; Wang, Jue; Singh, Atul K.; Lei, Aiwen (2017). "Recent Advances in Radical C–H Activation/Radical Cross-Coupling". Chemical Reviews. 117 (13): 9016–9085. doi:10.1021/acs.chemrev.6b00620. PMID 28639787.
References[edit]
- ^ Korch, Katerina M.; Watson, Donald A. (2019). "Cross-Coupling of Heteroatomic Electrophiles". Chemical Reviews. 119 (13): 8192–8228. doi:10.1021/acs.chemrev.8b00628. PMC 6620169. PMID 31184483.
- ^ Corbet, Jean-Pierre; Mignani, Gérard (2006). "Selected Patented Cross-Coupling Reaction Technologies". Chemical Reviews. 106 (7): 2651–2710. doi:10.1021/cr0505268. PMID 16836296.
- ^ New Trends in Cross-Coupling: Theory and Applications Thomas Colacot (Editor) 2014 ISBN 978-1-84973-896-5
- ^ a b King, A. O.; Yasuda, N. (2004). "Palladium-Catalyzed Cross-Coupling Reactions in the Synthesis of Pharmaceuticals". Organometallics in Process Chemistry. Topics in Organometallic Chemistry. Vol. 6. Heidelberg: Springer. pp. 205–245. doi:10.1007/b94551. ISBN 978-3-540-01603-8.
- ^ "The Nobel Prize in Chemistry 2010 - Richard F. Heck, Ei-ichi Negishi, Akira Suzuki". NobelPrize.org. 2010-10-06. Retrieved 2010-10-06.
- ^ Johansson Seechurn, Carin C. C.; Kitching, Matthew O.; Colacot, Thomas J.; Snieckus, Victor (2012). "Palladium-Catalyzed Cross-Coupling: A Historical Contextual Perspective to the 2010 Nobel Prize". Angewandte Chemie International Edition. 51 (21): 5062–5085. doi:10.1002/anie.201107017. PMID 22573393. S2CID 20582425.
- ^ Sun, Chang-Liang; Shi, Zhang-Jie (2014). "Transition-Metal-Free Coupling Reactions". Chemical Reviews. 114 (18): 9219–9280. doi:10.1021/cr400274j. PMID 25184859.
- ^ Thayer, Ann (2005-09-05). "Removing Impurities". Chemical & Engineering News. Retrieved 2015-12-11.
- ^ Yin, L.; Liebscher, J. (2007). "Carbon−Carbon Coupling Reactions Catalyzed by Heterogeneous Palladium Catalysts". Chemical Reviews. 107 (1): 133–173. doi:10.1021/cr0505674. PMID 17212474. S2CID 36974481.
- ^ Corbet, Jean-Pierre; Mignani, Gérard (2006). "Selected Patented Cross-Coupling Reaction Technologies". Chemical Reviews. 106 (7): 2651–2710. doi:10.1021/cr0505268. PMID 16836296.
- ^ Evano, Gwilherm; Blanchard, Nicolas; Toumi, Mathieu (2008). "Copper-Mediated Coupling Reactions and Their Applications in Natural Products and Designed Biomolecules Synthesis". Chemical Reviews. 108 (8): 3054–3131. doi:10.1021/cr8002505. PMID 18698737.
- ^ Robin B. Bedford (2015). "How Low Does Iron Go? Chasing the Active Species in Fe-Catalyzed Cross-Coupling Reactions". Acc. Chem. Res. 48 (5): 1485–1493. doi:10.1021/acs.accounts.5b00042. PMID 25916260.
- ^ Cahiez, GéRard; Moyeux, Alban (2010). "Cobalt-Catalyzed Cross-Coupling Reactions". Chemical Reviews. 110 (3): 1435–1462. doi:10.1021/cr9000786. PMID 20148539.
- ^ Rosen, Brad M.; Quasdorf, Kyle W.; Wilson, Daniella A.; Zhang, Na; Resmerita, Ana-Maria; Garg, Neil K.; Percec, Virgil (2011). "Nickel-Catalyzed Cross-Couplings Involving Carbon−Oxygen Bonds". Chemical Reviews. 111 (3): 1346–1416. doi:10.1021/cr100259t. PMC 3055945. PMID 21133429.
- ^ Murahashi, Shunichi; Yamamura, Masaaki; Yanagisawa, Kenichi; Mita, Nobuaki; Kondo, Kaoru (1979). "Stereoselective synthesis of alkenes and alkenyl sulfides from alkenyl halides using palladium and ruthenium catalysts". The Journal of Organic Chemistry. 44 (14): 2408–2417. doi:10.1021/jo01328a016. ISSN 0022-3263.
- ^ Clayden, J.; Greeves, N.; Warren, S. Organic Chemistry, 2nd ed.; Oxford UP: Oxford, U.K., 2012. pp. 1069-1102.
- ^ Ruiz-Castillo, P.; Buchwald, S. L. (2016). "Applications of Palladium-Catalyzed C–N Cross-Coupling Reactions". Chemical Reviews. 116 (19): 12564–12649. doi:10.1021/acs.chemrev.6b00512. PMC 5070552. PMID 27689804.
- ^ Jennifer X. Qiao; Patrick Y.S. Lam (2011). "Recent Advances in Chan–Lam Coupling Reaction: Copper-Promoted C–Heteroatom Bond Cross-Coupling Reactions with Boronic Acids and Derivatives". In Dennis G. Hall (ed.). Boronic Acids: Preparation and Applications in Organic Synthesis, Medicine and Materials. Wiley-VCH. pp. 315–361. doi:10.1002/9783527639328.ch6. ISBN 9783527639328.
- ^ M. Lafrance; C. N. Rowley; T. K. Woo; K. Fagnou (2006). "Catalytic Intermolecular Direct Arylation of Perfluorobenzenes". J. Am. Chem. Soc. 128 (27): 8754–8756. CiteSeerX 10.1.1.631.607. doi:10.1021/ja062509l. PMID 16819868.
- ^ Cornils, Boy; Börner, Armin; Franke, Robert; Zhang, Baoxin; Wiebus, Ernst; Schmid, Klaus (2017). "Hydroformylation". Applied Homogeneous Catalysis with Organometallic Compounds. pp. 23–90. doi:10.1002/9783527651733.ch2. ISBN 9783527328970.
- ^ Roughley, Stephen D.; Jordan, Allan M. (2011). "The Medicinal Chemist's Toolbox: An Analysis of Reactions Used in the Pursuit of Drug Candidates". Journal of Medicinal Chemistry. 54 (10): 3451–3479. doi:10.1021/jm200187y. PMID 21504168.
- ^ Hartwig, J. F. Organotransition Metal Chemistry, from Bonding to Catalysis; University Science Books: New York, 2010. ISBN 1-891389-53-X