Influenza A virus subtype H5N1
| Influenza A virus subtype H5N1 | |
|---|---|

| |
| Colorized transmission electron micrograph of Avian influenza A H5N1 viruses (seen in gold) grown in MDCK cells (seen in green). | |
| Virus classification | |
| (unranked): | Virus |
| Realm: | Riboviria |
| Kingdom: | Orthornavirae |
| Phylum: | Negarnaviricota |
| Class: | Insthoviricetes |
| Order: | Articulavirales |
| Family: | Orthomyxoviridae |
| Genus: | Alphainfluenzavirus |
| Species: | |
| Serotype: | Influenza A virus subtype H5N1
|
| Notable strains | |
 |
Parts of this article (those related to developments over the last 10 to 15 years ) need to be updated. (January 2024) |
Influenza A virus subtype H5N1 (A/H5N1) is a subtype of the influenza A virus, which causes influenza (flu), predominantly in birds. It is enzootic (maintained in the population) in many bird populations, and also panzootic (affecting animals of many species over a wide area).[1] A/H5N1 virus can also infect mammals (including humans) which have been exposed to infected birds; in these cases symptoms are frequently severe or fatal.[2]
A/H5N1 virus is shed in the saliva, mucous, and feces of infected birds; other infected animals may shed bird flu viruses in respiratory secretions and other body fluids (e.g., cow milk).[3] The virus can spread rapidly through poultry flocks and among wild birds.[3] An estimated half a billion farmed birds have been slaughtered in efforts to contain the virus.[2]
Symptoms of A/H5N1 influenza vary according to both the strain of virus underlying the infection, and on the species of bird or mammal affected.[4][5] Classification as either Low Pathogenic Avian Influenza (LPAI) or High Pathogenic Avian Influenza (HPAI) is based on the severity of symptoms in domestic chickens and does not predict severity of symptoms in other species.[6] Chickens infected with LPAI A/H5N1 virus display mild symptoms or are asymptomatic, whereas HPAI A/H5N1 causes serious breathing difficulties, significant drop in egg production, and sudden death.[7]
Humans and other mammals can only become infected after prolonged close contact with infected birds.[8] In mammals including humans, A/H5N1 influenza (whether LPAI or HPAI) is rare. Symptoms of infection vary from mild to severe, including fever, diarrhoea, and cough.[5] Human infections with A/H5N1 virus have been reported in 23 countries since 1997, resulting in severe pneumonia and death in about 50% of cases.[8] As of May 2024, 889 human cases had been identified worldwide with 463 fatalities.[9]
A/H5N1 influenza virus was first identified in farmed birds in Southern China in 1996.[10] Between 1996 and 2018 A/H5N1 coexisted in bird populations with other subtypes of the virus, but since then the highly pathogenic subtype HPAI A(H5N1) has become the dominant strain in bird populations worldwide.[11] Some strains of A/H5N1 which are highly pathogenic to chickens have adapted to cause mild symptoms in ducks and geese,[12][6] and are able to spread rapidly through bird migration.[13] Mammal species which have been recorded with H5N1 infection include cows, seals, goats, and skunks.[14]
Due to the high lethality and virulence of HPAI A(H5N1), its worldwide presence, its increasingly diverse host reservoir, and its significant ongoing mutations, the H5N1 virus is regarded to be the world's largest pandemic threat.[15] Domestic poultry may potentially be protected from specific strains of the virus by vaccination.[16] In the event of a serious outbreak of H5N1 flu among humans, health agencies have prepared "candidate" vaccines which may be used prevent infection and control the outbreak, however it could take several months to ramp up mass production.[3][17][18]
Signs and symptoms[edit]
Birds[edit]
Clinical signs of H5N1 in birds range from mild—decrease in egg production, nasal discharge, coughing and sneezing—to severe, including loss of coordination, energy, and appetite; soft-shelled or misshapen eggs; purple discoloration of the wattles, head, eyelids, combs, and hocks; and diarrhea. Sometimes the first noticeable sign is sudden death.[19]
Humans[edit]
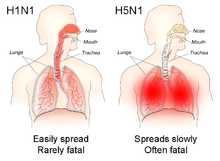
In general, humans who catch a humanized influenza A virus (a human flu virus of type A) usually have symptoms that include fever, cough, sore throat, muscle aches, conjunctivitis, and, in severe cases, breathing problems and pneumonia that may be fatal.[19] The severity of the infection depends in large part on the state of the infected persons' immune systems and whether they had been exposed to the strain before (in which case they would be partially immune). No one knows if these or other symptoms will be the symptoms of a humanized H5N1 flu.
The avian influenza hemagglutinin prefers to bind to alpha-2,3 sialic acid receptors, while the human influenza hemagglutinin prefers to bind to alpha-2,6 sialic acid receptors.[21][22] This means that when the H5N1 strain infects humans, it will replicate in the lower respiratory tract (where alpha-2,3 sialic acid receptors are more plentiful in humans) and consequently cause viral pneumonia.[23][24] As of April 2024, there is no human-adapted form of H5N1 influenza, so all humans who have caught it so far have caught avian H5N1.
The reported mortality rate of highly pathogenic H5N1 avian influenza in a human is high; WHO data indicate 60% of cases classified as H5N1 resulted in death. However, there is some evidence the actual mortality rate of avian flu could be much lower, as there may be many people with milder symptoms who do not seek treatment and are not counted.[25][26]
In one case, a boy with H5N1 experienced diarrhea followed rapidly by a coma without developing respiratory or flu-like symptoms.[27] There have been studies of the levels of cytokines in humans infected by the H5N1 flu virus. Of particular concern is elevated levels of tumor necrosis factor-alpha, a protein associated with tissue destruction at sites of infection and increased production of other cytokines. Flu virus-induced increases in the level of cytokines is also associated with flu symptoms, including fever, chills, vomiting and headache. Tissue damage associated with pathogenic flu virus infection can ultimately result in death.[28] The inflammatory cascade triggered by H5N1 has been called a 'cytokine storm' by some, because of what seems to be a positive feedback process of damage to the body resulting from immune system stimulation. H5N1 induces higher levels of cytokines than the more common flu virus types.[29]
Genetics[edit]
The first known strain of HPAI A(H5N1) (called A/chicken/Scotland/59) killed two flocks of chickens in Scotland in 1959, but that strain was very different from the highly pathogenic strain of H5N1. The dominant strain of HPAI A(H5N1) in 2004 evolved from 1999 to 2002 creating the Z genotype.[30] It has also been called "Asian lineage HPAI A(H5N1)".
Asian lineage HPAI A(H5N1) is divided into two antigenic clades. "Clade 1 includes human and bird isolates from Vietnam, Thailand, and Cambodia and bird isolates from Laos and Malaysia. Clade 2 viruses were first identified in bird isolates from China, Indonesia, Japan, and South Korea before spreading westward to the Middle East, Europe, and Africa. The clade 2 viruses have been primarily responsible for human H5N1 infections that have occurred during late 2005 and 2006, according to WHO. Genetic analysis has identified six subclades of clade 2, three of which have a distinct geographic distribution and have been implicated in human infections: Map
- Subclade 1, Indonesia
- Subclade 2, Europe, Middle East, and Africa (called EMA)
- Subclade 3, China[31][32][33]
A 2007 study focused on the EMA subclade has shed further light on the EMA mutations. "The 36 new isolates reported here greatly expand the amount of whole-genome sequence data available from recent avian influenza (H5N1) isolates. Before our project, GenBank contained only 5 other complete genomes from Europe for the 2004–2006 period, and it contained no whole genomes from the Middle East or northern Africa. Our analysis showed several new findings. First, all European, Middle Eastern, and African samples fall into a clade that is distinct from other contemporary Asian clades, all of which share common ancestry with the original 1997 Hong Kong strain. Phylogenetic trees built on each of the 8 segments show a consistent picture of 3 lineages, as illustrated by the HA tree shown in Figure 1. Two of the clades contain exclusively Vietnamese isolates; the smaller of these, with 5 isolates, we label V1; the larger clade, with 9 isolates, is V2. The remaining 22 isolates all fall into a third, clearly distinct clade, labeled EMA, which comprises samples from Europe, the Middle East, and Africa. Trees for the other 7 segments display a similar topology, with clades V1, V2, and EMA clearly separated in each case. Analyses of all available complete influenza (H5N1) genomes and of 589 HA sequences placed the EMA clade as distinct from the major clades circulating in People's Republic of China, Indonesia, and Southeast Asia."[34]
Terminology[edit]
H5N1 isolates are identified like this actual HPAI A(H5N1) example, A/chicken/Nakorn-Patom/Thailand/CU-K2/04(H5N1):
- A stands for the genus of influenza (A, B or C).
- chicken is the animal species the isolate was found in (note: human isolates lack this component term and are thus identified as human isolates by default)
- Nakorn-Patom/Thailand is the place this specific virus was isolated
- CU-K2 is the laboratory reference number that identifies it from other influenza viruses isolated at the same place and year
- 04 represents the year of isolation 2004
- H5 stands for the fifth of several known types of the protein hemagglutinin.
- N1 stands for the first of several known types of the protein neuraminidase.
Other examples include: A/duck/Hong Kong/308/78(H5N3), A/avian/NY/01(H5N2), A/chicken/Mexico/31381-3/94(H5N2), and A/shoveler/Egypt/03(H5N2).[35]
As with other avian flu viruses, H5N1 has strains called "highly pathogenic" (HP) and "low-pathogenic" (LP). Avian influenza viruses that cause HPAI are highly virulent, and mortality rates in infected flocks often approach 100%. LPAI viruses have negligible virulence, but these viruses can serve as progenitors to HPAI viruses. The strain of H5N1 responsible for the deaths of birds across the world is an HPAI strain; all other strains of H5N1, including a North American strain that causes no disease at all in any species, are LPAI strains. All HPAI strains identified to date have involved H5 and H7 subtypes. The distinction concerns pathogenicity in poultry, not humans. Normally, a highly pathogenic avian virus is not highly pathogenic to either humans or nonpoultry birds. This[which?] deadly strain of H5N1 is unusual in being deadly to so many species, including some, like domestic cats, never previously susceptible to any influenza virus.[36][failed verification]
[edit]

H5N1 is a subtype of the species Influenza A virus of the genus Alphainfluenzavirus of the family Orthomyxoviridae. Like all other influenza A subtypes, the H5N1 subtype is an RNA virus. It has a segmented genome of eight negative sense, single-strands of RNA, abbreviated as PB2, PB1, PA, HA, NP, NA, MP and NS.[citation needed]
HA codes for hemagglutinin, an antigenic glycoprotein found on the surface of the influenza viruses and is responsible for binding the virus to the cell that is being infected. NA codes for neuraminidase, an antigenic glycosylated enzyme found on the surface of the influenza viruses. It facilitates the release of progeny viruses from infected cells.[37] The hemagglutinin (HA) and neuraminidase (NA) RNA strands specify the structure of proteins that are most medically relevant as targets for antiviral drugs and antibodies. HA and NA are also used as the basis for the naming of the different subtypes of influenza A viruses. This is where the H and N come from in H5N1.
Low pathogenic H5N1[edit]
Low pathogenic avian influenza H5N1 (LPAI H5N1), also called "North American" H5N1, commonly occurs in wild birds. In most cases, it causes minor sickness or no noticeable signs of disease in birds. It is not known to affect humans at all. The only concern about it is that it is possible for it to be transmitted to poultry and in poultry mutate into a highly pathogenic strain.
- 1966 – LPAI H5N1 A/Turkey/Ontario/6613/1966(H5N1) was detected in a flock of infected turkeys in Ontario, Canada.[38][39]
- 1975 – LPAI H5N1 was detected in a wild mallard duck and a wild blue goose in Wisconsin.
- 1981 and 1985 – LPAI H5N1 was detected in ducks by the University of Minnesota conducting a sampling procedure in which sentinel ducks were monitored in cages placed in the wild for a short period of time.
- 1983 – LPAI H5N1 was detected in ring-billed gulls in Pennsylvania.
- 1986 – LPAI H5N1 was detected in a wild mallard duck in Ohio.
- 2005 – LPAI H5N1 was detected in ducks in Manitoba, Canada.
- 2008 – LPAI H5N1 was detected in ducks in New Zealand.
- 2009 – LPAI H5N1 was detected in commercial poultry in British Columbia.[40]
"In the past, there was no requirement for reporting or tracking LPAI H5 or H7 detections in wild birds so states and universities tested wild bird samples independently of USDA. Because of this, the above list of previous detections might not be all inclusive of past LPAI H5N1 detections. However, the World Organization for Animal Health (OIE) recently changed its requirement of reporting detections of avian influenza. Effective in 2006, all confirmed LPAI H5 and H7 AI subtypes must be reported to the OIE because of their potential to mutate into highly pathogenic strains. Therefore, USDA now tracks these detections in wild birds, backyard flocks, commercial flocks and live bird markets."[41]
High mutation rate[edit]
Influenza viruses have a relatively high mutation rate that is characteristic of RNA viruses. The segmentation of its genome facilitates genetic recombination by segment reassortment in hosts infected with two different strains of influenza viruses at the same time.[42][43] A previously uncontagious strain may then be able to pass between humans, one of several possible paths to a pandemic.[citation needed]
The ability of various influenza strains to show species-selectivity is largely due to variation in the hemagglutinin genes. Genetic mutations in the hemagglutinin gene that cause single amino acid substitutions can significantly alter the ability of viral hemagglutinin proteins to bind to receptors on the surface of host cells. Such mutations in avian H5N1 viruses can change virus strains from being inefficient at infecting human cells to being as efficient in causing human infections as more common human influenza virus types.[44] This doesn't mean that one amino acid substitution can cause a pandemic, but it does mean that one amino acid substitution can cause an avian flu virus that is not pathogenic in humans to become pathogenic in humans.[citation needed]
Influenza A virus subtype H3N2 is endemic in pigs in China, and has been detected in pigs in Vietnam, increasing fears of the emergence of new variant strains. The dominant strain of annual flu virus in January 2006 was H3N2, which is now resistant to the standard antiviral drugs amantadine and rimantadine. The possibility of H5N1 and H3N2 exchanging genes through reassortment is a major concern. If a reassortment in H5N1 occurs, it might remain an H5N1 subtype, or it could shift subtypes, as H2N2 did when it evolved into the Hong Kong Flu strain of H3N2.
Both the H2N2 and H3N2 pandemic strains contained avian influenza virus RNA segments. "While the pandemic human influenza viruses of 1957 (H2N2) and 1968 (H3N2) clearly arose through reassortment between human and avian viruses, the influenza virus causing the 'Spanish flu' in 1918 appears to be entirely derived from an avian source".[45]
Prevention[edit]
Vaccine[edit]
There are several H5N1 vaccines for several of the avian H5N1 varieties, but the continual mutation of H5N1 renders them of limited use to date: while vaccines can sometimes provide cross-protection against related flu strains, the best protection would be from a vaccine specifically produced for any future pandemic flu virus strain. Daniel R. Lucey, co-director of the Biohazardous Threats and Emerging Diseases graduate program at Georgetown University has made this point, "There is no H5N1 pandemic so there can be no pandemic vaccine".[46] However, "pre-pandemic vaccines" have been created; are being refined and tested; and do have some promise both in furthering research and preparedness for the next pandemic.[47][48][49] Vaccine manufacturing companies are being encouraged to increase capacity so that if a pandemic vaccine is needed, facilities will be available for rapid production of large amounts of a vaccine specific to a new pandemic strain.
Public health[edit]
"The United States is collaborating closely with eight international organizations, including the World Health Organization (WHO), the Food and Agriculture Organization of the United Nations (FAO), the World Organization for Animal Health (OIE), and 88 foreign governments to address the situation through planning, greater monitoring, and full transparency in reporting and investigating avian influenza occurrences. The United States and these international partners have led global efforts to encourage countries to heighten surveillance for outbreaks in poultry and significant numbers of deaths in migratory birds and to rapidly introduce containment measures. The U.S. Agency for International Development (USAID) and the U.S. Department of State, the U.S. Department of Health and Human Services (HHS), and Agriculture (USDA) are coordinating future international response measures on behalf of the White House with departments and agencies across the federal government".[50]
Together steps are being taken to "minimize the risk of further spread in animal populations", "reduce the risk of human infections", and "further support pandemic planning and preparedness".[50]
Ongoing detailed mutually coordinated onsite surveillance and analysis of human and animal H5N1 avian flu outbreaks are being conducted and reported by the USGS National Wildlife Health Center, the Centers for Disease Control and Prevention, the World Health Organization, the European Commission, and others.[51]
Treatment[edit]
There is no highly effective treatment for H5N1 flu, but oseltamivir (commercially marketed by Roche as Tamiflu) can sometimes inhibit the influenza virus from spreading inside the user's body. This drug has become a focus for some governments and organizations trying to prepare for a possible H5N1 pandemic.[52] On April 20, 2006, Roche AG announced that a stockpile of three million treatment courses of Tamiflu are waiting at the disposal of the World Health Organization to be used in case of a flu pandemic; separately Roche donated two million courses to the WHO for use in developing nations that may be affected by such a pandemic but lack the ability to purchase large quantities of the drug.[53]
However, WHO expert Hassan al-Bushra has said:
- "Even now, we remain unsure about Tamiflu's real effectiveness. As for a vaccine, work cannot start on it until the emergence of a new virus, and we predict it would take six to nine months to develop it. For the moment, we cannot by any means count on a potential vaccine to prevent the spread of a contagious influenza virus, whose various precedents in the past 90 years have been highly pathogenic".[54]
Animal and lab studies suggest that Relenza (zanamivir), which is in the same class of drugs as Tamiflu, may also be effective against H5N1. In a study performed on mice in 2000, "zanamivir was shown to be efficacious in treating avian influenza viruses H9N2, H6N1, and H5N1 transmissible to mammals".[55] In addition, mice studies suggest the combination of zanamivir, celecoxib and mesalazine looks promising producing a 50% survival rate compared to no survival in the placebo arm.[56] While no one knows if zanamivir will be useful or not on a yet to exist pandemic strain of H5N1, it might be useful to stockpile zanamivir as well as oseltamivir in the event of an H5N1 influenza pandemic. Neither oseltamivir nor zanamivir can be manufactured in quantities that would be meaningful once efficient human transmission starts.[57] In September, 2006, a WHO scientist announced that studies had confirmed cases of H5N1 strains resistant to Tamiflu and Amantadine.[58] Tamiflu-resistant strains have also appeared in the EU, which remain sensitive to Relenza.[59][60]
Epidemiology[edit]
The earliest infections of humans by H5N1 coincided with an epizootic (an epidemic in nonhumans) of H5N1 influenza in Hong Kong's poultry population in 1997. This panzootic (a disease affecting animals of many species, especially over a wide area) outbreak was stopped by the killing of the entire domestic poultry population within the territory. However, the disease has continued to spread; outbreaks were reported in Asia again in 2003. On December 21, 2009, the WHO announced a total of 447 cases which resulted in the deaths of 263.[19][61]
Contagiousness[edit]

H5N1 is easily transmissible between birds, facilitating a potential global spread of H5N1. While H5N1 undergoes mutation and reassortment, creating variations which can infect species not previously known to carry the virus, not all of these variant forms can infect humans. H5N1 as an avian virus preferentially binds to a type of galactose receptors that populate the avian respiratory tract from the nose to the lungs and are virtually absent in humans, occurring only in and around the alveoli, structures deep in the lungs where oxygen is passed to the blood. Therefore, the virus is not easily expelled by coughing and sneezing, the usual route of transmission.[22][23][62]
H5N1 is mainly spread by domestic poultry, both through the movements of infected birds and poultry products and through the use of infected poultry manure as fertilizer or feed. Humans with H5N1 have typically caught it from chickens, which were in turn infected by other poultry or waterfowl. Migrating waterfowl (wild ducks, geese and swans) carry H5N1, often without becoming sick.[63][64] Many species of birds and mammals can be infected with HPAI A(H5N1), but the role of animals other than poultry and waterfowl as disease-spreading hosts is unknown.[65]
According to a report by the World Health Organization, H5N1 may be spread indirectly. The report stated the virus may sometimes stick to surfaces or get kicked up in fertilizer dust to infect people.[66]
Virulence[edit]
H5N1 has mutated into a variety of strains with differing pathogenic profiles, some pathogenic to one species but not others, some pathogenic to multiple species. Each specific known genetic variation is traceable to a virus isolate of a specific case of infection. Through antigenic drift, H5N1 has mutated into dozens of highly pathogenic varieties divided into genetic clades which are known from specific isolates, but all belong to genotype Z of avian influenza virus H5N1, now the dominant genotype.[43][42] H5N1 isolates found in Hong Kong in 1997 and 2001 were not consistently transmitted efficiently among birds and did not cause significant disease in these animals. In 2002, new isolates of H5N1 were appearing within the bird population of Hong Kong. These new isolates caused acute disease, including severe neurological dysfunction and death in ducks. This was the first reported case of lethal influenza virus infection in wild aquatic birds since 1961.[67]
Genotype Z emerged in 2002 through reassortment from earlier highly pathogenic genotypes of H5N1[68] that first infected birds in China in 1996, and first infected humans in Hong Kong in 1997.[42][43][69] Genotype Z is endemic in birds in Southeast Asia, has created at least two clades that can infect humans, and is spreading across the globe in bird populations. Mutations occurring within this genotype are increasing their pathogenicity.[70] Birds are also able to shed the virus for longer periods of time before their death, increasing the transmissibility of the virus.
Transmission and host range[edit]
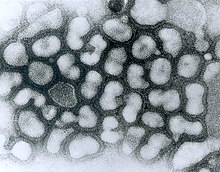
Infected birds transmit H5N1 through their saliva, nasal secretions, feces and blood. Other animals may become infected with the virus through direct contact with these bodily fluids or through contact with surfaces contaminated with them. H5N1 remains infectious after over 30 days at 0 °C (32 °F) (over one month at freezing temperature) or 6 days at 37 °C (99 °F) (one week at human body temperature); at ordinary temperatures it lasts in the environment for weeks. In Arctic temperatures, it does not degrade at all.
Because migratory birds are among the carriers of the highly pathogenic H5N1 virus, it is spreading to all parts of the world. H5N1 is different from all previously known highly pathogenic avian flu viruses in its ability to be spread by animals other than poultry.
In October 2004, researchers discovered H5N1 is far more dangerous than was previously believed. Waterfowl were revealed to be directly spreading this highly pathogenic strain to chickens, crows, pigeons, and other birds, and the virus was increasing its ability to infect mammals, as well. From this point on, avian flu experts increasingly referred to containment as a strategy that can delay, but not ultimately prevent, a future avian flu pandemic.
"Since 1997, studies of influenza A (H5N1) indicate that these viruses continue to evolve, with changes in antigenicity and internal gene constellations; an expanded host range in avian species and the ability to infect felids; enhanced pathogenicity in experimentally infected mice and ferrets, in which they cause systemic infections; and increased environmental stability."[71]
The New York Times, in an article on transmission of H5N1 through smuggled birds, reports Wade Hagemeijer of Wetlands International stating, "We believe it is spread by both bird migration and trade, but that trade, particularly illegal trade, is more important".[72]
On September 29, 2007, researchers reported the H5N1 bird flu virus can also pass through a pregnant woman's placenta to infect the fetus. They also found evidence of what doctors had long suspected—the virus not only affects the lungs, but also passes throughout the body into the gastrointestinal tract, the brain, liver, and blood cells.[73]
In May 2013, North Korea confirmed a H5N1 bird flu outbreak that forced authorities to kill over 160,000 ducks in Pyongyang.[74]
| 2003 | 2004 | 2005 | 2006 | 2007 | 2008 | 2009 | 2010 | 2011 | 2012 | 2013 | 2014 | 2015 | 2016 | 2017 | 2018 | 2019 | 2020 | 2021 | 2022 | 2023 | 2024 | Total | ||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
cases |
deaths |
CFR
|
cases |
deaths |
CFR
|
cases |
deaths |
CFR
|
cases |
deaths |
CFR
|
cases |
deaths |
CFR
|
cases |
deaths |
CFR
|
cases |
deaths |
CFR
|
cases |
deaths |
CFR
|
cases |
deaths |
CFR
|
cases |
deaths |
CFR
|
cases |
deaths |
CFR
|
cases |
deaths |
CFR
|
cases |
deaths |
CFR
|
cases |
deaths |
CFR
|
cases |
deaths |
CFR
|
cases |
deaths |
CFR
|
cases |
deaths |
CFR
|
cases |
deaths |
CFR
|
cases |
deaths |
CFR
|
cases |
deaths |
CFR
|
cases |
deaths |
CFR
|
cases |
deaths |
CFR
|
cases |
deaths |
CFR
|
| 8 | 5 | 62.5% | 8 | 5 | 62.5% | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 1 | 0 | 0% | 2 | 0 | 0% | 3 | 0 | 0% | 1 | 1 | 100% | 1 | 0 | 0% | 8 | 1 | 12.5% | |||||||||||||||||||||||||||||||||||||||||||||||||||
| 4 | 4 | 100% | 2 | 2 | 100% | 1 | 1 | 100% | 1 | 0 | 0% | 1 | 0 | 0% | 1 | 1 | 100% | 8 | 8 | 100% | 3 | 3 | 100% | 26 | 14 | 53.8% | 9 | 4 | 44.4% | 6 | 4 | 66.7% | 5 | 1 | 20.0% | 67 | 42 | 62.7% | ||||||||||||||||||||||||||||||
| 1 | 1 | 100% | 1 | 1 | 100% | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 1[75] | 0 | 0% | 1 | 0 | 0% | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 1 | 1 | 100% | 8 | 5 | 62.5% | 13 | 8 | 61.5% | 5 | 3 | 60.0% | 4 | 4 | 100% | 7 | 4 | 57.1% | 2 | 1 | 50.0% | 1 | 1 | 100% | 2 | 1 | 50.0% | 2 | 2 | 100% | 2 | 0 | 0% | 6 | 1 | 16.7% | 1 | 1 | 100% | 1 | 0 | 0% | 55 | 32 | 58.2% | ||||||||||||||||||||||||
| 1 | 0 | 0% | 1 | 0 | 0% | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 1 | 0 | 0% | 1 | 0 | 0% | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 18 | 10 | 55.6% | 25 | 9 | 36.0% | 8 | 4 | 50.0% | 39 | 4 | 10.3% | 29 | 13 | 44.8% | 39 | 15 | 38.5% | 11 | 5 | 45.5% | 4 | 3 | 75.0% | 37 | 14 | 37.8% | 136 | 39 | 28.7% | 10 | 3 | 30.0% | 3 | 1 | 33.3% | 359 | 120 | 33.4% | ||||||||||||||||||||||||||||||
| 1 | 1 | 100% | 1 | 1 | 100% | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 20 | 13 | 65.0% | 55 | 45 | 81.8% | 42 | 37 | 88.1% | 24 | 20 | 83.3% | 21 | 19 | 90.5% | 9 | 7 | 77.8% | 12 | 10 | 83.3% | 9 | 9 | 100% | 3 | 3 | 100% | 2 | 2 | 100% | 2 | 2 | 100% | 1 | 1 | 100% | 200 | 168 | 84.0% | ||||||||||||||||||||||||||||||
| 3 | 2 | 66.6% | 3 | 2 | 66.6% | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 2 | 2 | 100% | 1 | 0 | 0% | 3 | 2 | 66.7% | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 1 | 0 | 0% | 1 | 0 | 0% | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 1 | 1 | 100% | 1 | 1 | 100% | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 1 | 1 | 100% | 1 | 1 | 100% | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 3 | 1 | 33.3% | 3 | 1 | 33.3% | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 2 | 0 | 0% | 2 | 0 | 0% | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 17 | 12 | 70.6% | 5 | 2 | 40.0% | 3 | 3 | 100% | 25 | 17 | 68.0% | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 12 | 4 | 33.3% | 12 | 4 | 33.3% | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 1 | 0 | 0% | 4 | 0 | 0% | 5 | 0 | 0% | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 1 | 0 | 0% | 1 | 0 | 0% | 2 | 0 | 0% | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 3 | 3 | 100% | 29 | 20 | 69.0% | 61 | 19 | 31.1% | 8 | 5 | 62.5% | 6 | 5 | 83.3% | 5 | 5 | 100% | 7 | 2 | 28.6% | 4 | 2 | 50.0% | 2 | 1 | 50.0% | 2 | 2 | 100% | 1 | 0 | 0% | 1 | 1 | 100% | 129 | 65 | 50.0% | ||||||||||||||||||||||||||||||
| 4 | 4 | 100% | 46 | 32 | 69.6% | 98 | 43 | 43.9% | 115 | 79 | 68.7% | 88 | 59 | 67.0% | 44 | 33 | 75.0% | 73 | 32 | 43.8% | 48 | 24 | 50.0% | 62 | 34 | 54.8% | 32 | 20 | 62.5% | 39 | 25 | 64.1% | 52 | 22 | 42.3% | 145 | 42 | 29.0% | 10 | 3 | 30.0% | 4 | 2 | 50.0% | 0 | 0 | 0% | 1 | 1 | 100% | 1 | 0 | 0% | 2 | 1 | 50.0% | 6 | 1 | 16.7% | 12 | 4 | 33.3% | 7 | 2 | 28.6% | 889 | 463 | 52.1% |
2020–2024 global H5N1 outbreak[edit]
Europe[edit]
A major outbreak of a new strain of H5N1 in wild birds and poultry appeared in Russia in August 2020 and quickly spread to other parts of Europe by October.[76]
Over the winter of 2021 and 2022 avian flu spread among the population of barnacle geese on the Solway Firth, UK, with estimates of up to a third of the Svalbard population being lost;[77][78] pink-footed geese were also affected there and it seems carried the virus to new sites in northern Scotland. The disease was confirmed in sandwich terns in South Africa in April 2022.[79] In late spring 2022 avian flu outbreaks affected many species of wild bird in the United Kingdom, with heavy losses reported among seabirds returning to breed at colonies in the Northern Isles and Outer Hebrides,[80] including great skuas (bonxie) for which outbreaks had initially been reported in 2021[81] (Scotland hosts c. 60% of the world's breeding population) – the 2022 census on St Kilda showed a 64% decline on 2019 with 106 dead birds recorded so far (to 6 June),[82] gannets (1000+ birds reported dead at the Shetlands' Hermaness colony alone,[80] where there are around 26,000 breeding pairs), with many more gannets being reported dead at other colonies (Troup Head, Bass Rock, and St Kilda);[83] the range of species also seems to be expanding, with reports for many species of wildfowl, seabirds (auks, terns and gulls) and scavenging species (corvids and raptors).[84][85]
Elsewhere in Europe the virus killed hundreds (574+) of Dalmatian pelicans in Greece,[86][87] and in Israel around 6000 common cranes were found dead at Hula in December 2021.[88] A report by Scientific Task Force on Avian Influenza and Wild Birds on: "H5N1 Highly Pathogenic Avian Influenza in poultry and wild birds: Winter of 2021/2022 with focus on mass mortality of wild birds in UK and Israel" summarises the situation up to 24 January 2022 and mentions that "H5N8 HPAI is still responsible for poultry and wild bird cases mainly in Asia, H5N1 has now in effect replaced this subtype in Africa and Eurasia in both poultry and wild birds".[89]
The 2022–2023 season was also the worst recorded outbreak in the United Kingdom, with the British government requiring a so-called "poultry lockdown" which required that farmers keep their birds indoors.[90] Meanwhile, an outbreak of H5N1 on a Spanish mink farm led researchers to believe that they had observed the first case of mammal-to-mammal transmission of H5N1.[91] Human cases were reported in Spain in November 2022, and in the UK in May 2023.[76]
Asia[edit]
By November 2020, large outbreaks of the new strain of H5N1 had started to spread into wild birds and farmed poultry across Asia. In February 2023, human cases were reported in Cambodia.[76]
Africa[edit]
Large losses of poultry and wild birds to H5N1 started to occur in Africa in November 2021 and continued through 2022.[76]
Americas[edit]
Similar to 2021 reports, outbreaks were noted from gannet colonies in Canada, with thousands of birds dead in June 2022,[92] as well as common eiders and great black-backed gulls.[93] Prior to that there were reports of spread in wild birds in over 30 states in the US, including major mortalities in a double-crested cormorant colony in Barrington, Illinois,[94][95] the virus also spreading to scavengers including three bald eagles in Georgia.[96] Mass die-offs of both birds and mammals were noted in Peru during the 2022–2023 season.[97] In particular, the Peruvian government reported the deaths of approximately 63,000 birds as well as 716 sea lions, with the WHO noting that mammalian spillovers needed to be "monitored closely".[90][98] In the United States, the 2022–2023 avian outbreak was the worst since H5N1 was first detected.[90]
Ecuador entered into a three-month "animal-health emergency" on 29 November 2022, just days after its first case was reported, whereas Argentina and Uruguay both declared "national sanitary emergencies" on 15 February 2023, after their respective first cases were discovered.[99] On 22 May 2023, Brazil, as the world's largest exporter of chicken meat, declared a 180-day emergency following several cases detected in wild birds and created an emergency operations center to plan for and mitigate potential further spread of H5N1.[100] Human cases were reported in Ecuador and Chile.[76]
In March 2024, H5N1 infections were recorded for the first time in deceased and sick livestock located in the United States. Goats and cows in three states became ill after exposure to wild birds and culled poultry.[101] In early April, H5N1 was reported to have spread amongst dairy cow herds in multiple states of the USA, indicating cow-to-cow spread. A dairy worker in Texas also became infected, with conjunctivitis being the main symptom.[102]
Antarctica[edit]
H5N1 was detected in dead birds on the Antarctic mainland for the first time in February 2024.[103]
Mammalian infections[edit]
H5N1 transmission studies in ferrets (2011)[edit]
Novel, contagious strains of H5N1 were created by Ron Fouchier of the Erasmus Medical Center in Rotterdam, the Netherlands, who first presented his work to the public at an influenza conference in Malta in September 2011. Three mutations were introduced into the H5N1 virus genome, and the virus was then passed from the noses of infected ferrets to the noses of uninfected ones, which was repeated 10 times.[104] After these 10 passages the H5N1 virus had acquired the ability of transmission between ferrets via aerosols or respiratory droplets.
After Fouchier offered an article describing this work to the leading academic journal Science, the US National Science Advisory Board for Biosecurity (NSABB) recommended against publication of the full details of the study, and the one submitted to Nature by Yoshihiro Kawaoka of the University of Wisconsin describing related work. However, after additional consultations at the World Health Organization and by the NSABB, the NSABB reversed its position and recommended publication of revised versions of the two papers.[105] However, then the Dutch government declared that this type of manuscripts required Fouchier to apply for an export permit in the light of EU directive 428/2009 on dual use goods.[note 1] After much controversy surrounding the publishing of his research, Fouchier complied (under formal protest) with Dutch government demands to obtain a special permit[106] for submitting his manuscript, and his research appeared in a special issue of the journal Science devoted to H5N1.[107][108][109] The papers by Fouchier and Kawaoka conclude that it is entirely possible that a natural chain of mutations could lead to an H5N1 virus acquiring the capability of airborne transmission between mammals, and that a H5N1 influenza pandemic would not be impossible.[110]
In May 2013, it was reported that scientists at the Harbin Veterinary Research Institute in Harbin, China, had created H5N1 strains which passed between guinea pigs.[111]
In response to Fouchier and Kawaoka's work, a number of scientists expressed concerns with the risks of creating novel potential pandemic pathogens, culminating in the formation of the Cambridge Working Group, a consensus statement calling for an assessment of the risks and benefits of such research.[112][113]
Mammal-to-mammal transmission (2022–2024)[edit]
Although mammals, including humans, had become infected with H5N1 bird flu strains in the past, these cases had ostensibly been caused by direct exposure to infected birds, such as through consumption of birds by wildlife or exposure to infected poultry by farmers. In contrast, the October 2022 mammalian outbreak of H5N1 on a Spanish mink farm showed evidence of being the first recorded case of mammal-to-mammal transmission, with 4 percent of the farm's mink population dying from H5N1-related haemorrhagic pneumonia.[91][114] The mink respiratory tract is particularly well suited to act as a pathway of viral transmission into humans, which has concerned public health professionals due to the production of all but one approved human vaccine requiring the eggs of chickens, which H5N1 kills at a 90–100 percent fatality rate.[115] Infected mink in Spain were also found to have exhibited the "PB2" viral mutation found when H5N1 jumped into pigs over a decade prior, adding to fears that farms could be acting as incubators and/or reservoirs of the virus, similar to the role of minks in SARS-CoV-2.[91]
As of January 2023, fifteen species of wild and captive mammals had become infected with H5N1 throughout the United States.[116] A mass Caspian seal die-off in December 2022, with 700 infected seals found dead along the Caspian Sea coastline of Russia's Dagestan republic, worried researchers regarding the possibility that wild mammal-to-mammal spread had begun.[117] A similar mass die-off of 95% of southern elephant seal pups in 2023 also raised concerns of mammal-to-mammal spread, as nursing pups would have had less exposure to birds.[118]
In April 2024, spread of H5N1 amongst dairy cow herds in nine states of the USA strongly indicated the presence of cow-to-cow transmission.[119] Although mortality in bovines infected with H5N1 is rare, abundant virus shedding in the milk is evident. Around 50% of cats that lived on the affected dairy farms and were fed unpasteurised milk from symptomatic cows died within a few days from severe systemic influenza infection, raising significant concerns of cross-species mammal-to-mammal transmission.[120]
Human infections[edit]
As of April 2024, the WHO reported a total of 889 confirmed human cases which resulted in the deaths of 463 people since 2003.[121]
Human-to-human transmission[edit]
Cambodia (2023)[edit]
Following the February 2023 H5N1 death of an 11-year-old girl from Cambodia's Prey Veng province, her father was confirmed positive for the virus and several close contacts also began showing signs of infection. On 24 February 2023, the WHO expressed concern that the virus had potentially begun to spread among humans and ordered the production of a new human vaccine for H5N1.[122] Following the confirmed infections, the WHO began working with the Cambodian government to determine whether both individuals had gotten the virus directly from infected poultry or if it had indeed been a case of human-to-human transmission.[123] Further sequencing determined that at least one of the two cases was from an older H5N1 clade, 2.3.2.1c, which had circulated as a common H5N1 strain in Cambodia for many years, rather than the more recent clade 2.3.4.4b, which had caused mass poultry deaths since 2020. This older clade had jumped to humans in the past yet hadn't previously resulted in any known human-to-human transmission.[124]
On March 1, 2023, as Taiwan raised its travel alert for Cambodia, the WHO and the U.S. CDC, in concert with Cambodian authorities, determined that both of the individuals had been infected through direct contact with poultry.[125][126]
Society and culture[edit]
H5N1 has had a significant effect on human society, especially the financial, political, social, and personal responses to both actual and predicted deaths in birds, humans, and other animals. Billions of dollars are being raised and spent to research H5N1 and prepare for a potential avian influenza pandemic. Over $10 billion have been spent and over 200 million birds have been killed to try to contain H5N1.[127][128][129][130][131][132][133][134][135]
People have reacted by buying less chicken, causing poultry sales and prices to fall.[136] Many individuals have stockpiled supplies for a possible flu pandemic. International health officials and other experts have pointed out that many unknown questions still hover around the disease.[137]
Dr. David Nabarro, Chief Avian Flu Coordinator for the United Nations, and former Chief of Crisis Response for the World Health Organization has described himself as "quite scared" about H5N1's potential impact on humans. Nabarro has been accused of being alarmist before, and on his first day in his role for the United Nations, he proclaimed the avian flu could kill 150 million people. In an interview with the International Herald Tribune, Nabarro compares avian flu to AIDS in Africa, warning that underestimations led to inappropriate focus for research and intervention.[138]
See also[edit]
- Antigenic shift
- Avian influenza virus
- Favipiravir
- Fujian flu
- H5N1 clinical trials
- H7N9
- Influenza research
- Influenzavirus A
- International Conference on Emerging Infectious Diseases
- National Influenza Centers
- Swine influenza
- Zoonosis
Notes[edit]
- ^ The World Intellectual Property Organization (WIPO) lists strategic goods with prohibited goods or goods that require a special permit for import and export without which the carrier faces pecuniary punishment or up to 5 years' imprisonment.
References[edit]
Citations[edit]
- ^ "Influenza (Avian and other zoonotic)". World Health Organization. 3 October 2023. Retrieved 2024-05-06.
- ^ a b Bourk, India (26 April 2024). "'Unprecedented': How bird flu became an animal pandemic". www.bbc.com. Retrieved 2024-05-08.
- ^ a b c "Prevention and Antiviral Treatment of Bird Flu Viruses in People | Avian Influenza (Flu)". Centers for Disease Control. 2024-04-19. Retrieved 2024-05-08.
- ^ Services, Department of Health & Human. "Bird flu (avian influenza)". www.betterhealth.vic.gov.au. Retrieved 2024-05-09.
- ^ a b "Avian influenza: guidance, data and analysis". GOV.UK. 2021-11-18. Retrieved 2024-05-09.
- ^ a b CDC (2022-06-14). "Avian Influenza in Birds". Centers for Disease Control and Prevention. Retrieved 2024-05-06.
- ^ "Bird flu (avian influenza): how to spot and report it in poultry or other captive birds". Department for Environment, Food & Rural Affairs and Animal and Plant Health Agency. 2022-12-13. Retrieved 2024-05-06.
- ^ a b CDC (2024-02-01). "Influenza Type A Viruses". Centers for Disease Control and Prevention. Retrieved 2024-05-03.
- ^ Devlin, Hannah; correspondent, Hannah Devlin Science (2024-04-18). "Risk of bird flu spreading to humans is 'enormous concern', says WHO". The Guardian. ISSN 0261-3077. Retrieved 2024-05-06.
- ^ "Emergence and Evolution of H5N1 Bird Flu | Avian Influenza (Flu)". Centers for Disease Control. 2023-06-06. Retrieved 2024-05-03.
- ^ Huang, Pan; Sun, Lujia; Li, Jinhao; Wu, Qingyi; Rezaei, Nima; Jiang, Shibo; Pan, Chungen (2023-06-16). "Potential cross-species transmission of highly pathogenic avian influenza H5 subtype (HPAI H5) viruses to humans calls for the development of H5-specific and universal influenza vaccines". Cell Discovery. 9 (1): 1–13. doi:10.1038/s41421-023-00571-x. ISSN 2056-5968. PMC 10275984. PMID 37328456.
- ^ "Highlights in the History of Avian Influenza (Bird Flu) Timeline – 2020-2024 | Avian Influenza (Flu)". Centers for Disease Control. 2024-04-22. Retrieved 2024-05-08.
- ^ Caliendo, V.; Lewis, N. S.; Pohlmann, A.; Baillie, S. R.; Banyard, A. C.; Beer, M.; Brown, I. H.; Fouchier, R. a. M.; Hansen, R. D. E.; Lameris, T. K.; Lang, A. S.; Laurendeau, S.; Lung, O.; Robertson, G.; van der Jeugd, H. (2022-07-11). "Transatlantic spread of highly pathogenic avian influenza H5N1 by wild birds from Europe to North America in 2021". Scientific Reports. 12 (1): 11729. Bibcode:2022NatSR..1211729C. doi:10.1038/s41598-022-13447-z. ISSN 2045-2322. PMID 35821511.
- ^ "Bird flu is bad for poultry and cattle. Why it's not a dire threat for most of us — yet". NBC News. 2024-05-02. Retrieved 2024-05-09.
- ^ McKie, Robin (2024-04-20). "Next pandemic likely to be caused by flu virus, scientists warn". The Observer. ISSN 0029-7712. Retrieved 2024-05-09.
- ^ "Vaccination of poultry against highly pathogenic avian influenza – Available vaccines and vaccination strategies | EFSA". www.efsa.europa.eu. 2023-10-10. Retrieved 2024-05-09.
- ^ "Two possible bird flu vaccines could be available within weeks, if needed". NBC News. 2024-05-01. Retrieved 2024-05-09.
- ^ "Avian influenza (bird flu) | European Medicines Agency". www.ema.europa.eu. Retrieved 2024-05-09.
- ^ a b c Protecting Poultry Workers from Avian Influenza (Bird Flu) (PDF) (Report). U.S. Department of Health and Human Services, Public Health Service, Centers for Disease Control and Prevention, National Institute for Occupational Safety and Health. 2008. doi:10.26616/NIOSHPUB2008128. DHHS (NIOSH) Publication Number 2008–128.
- ^ Korteweg C, Gu J (May 2008). "Pathology, Molecular Biology, and Pathogenesis of Avian Influenza A (H5N1) Infection in Humans". Am. J. Pathol. 172 (5): 1155–1170. doi:10.2353/ajpath.2008.070791. PMC 2329826. PMID 18403604.
- ^ Bertram S, Glowacka I, Steffen I, Kühl A, Pöhlmann S (September 2010). "Novel insights into proteolytic cleavage of influenza virus hemagglutinin". Reviews in Medical Virology. 20 (5): 298–310. doi:10.1002/rmv.657. PMC 7169116. PMID 20629046.
The influenza virus HA binds to alpha 2–3 linked (avian viruses) or alpha 2–6 linked (human viruses) sialic acids presented by proteins or lipids on the host cell surface.
- ^ a b Shinya K, Ebina M, Yamada S, Ono M, Kasai N, Kawaoka Y (March 2006). "Avian flu: influenza virus receptors in the human airway". Nature. 440 (7083): 435–436. Bibcode:2006Natur.440..435S. doi:10.1038/440435a. PMID 16554799. S2CID 9472264.
- ^ a b van Riel D, Munster VJ, de Wit E, Rimmelzwaan GF, Fouchier RA, Osterhaus AD, Kuiken T (2006). "H5N1 Virus Attachment to Lower Respiratory Tract". Science. 312 (5772): 399. doi:10.1126/science.1125548. PMID 16556800. S2CID 33294327.
- ^ Bennett, Nicholas John (13 October 2021). "Avian Influenza (Bird Flu): Background, Pathophysiology, Epidemiology". Medscape Reference. Retrieved 28 April 2024.
The pathophysiology of avian influenza differs from that of normal influenza. Avian influenza is still primarily a respiratory infection but involves more of the lower airways than human influenza typically does. This is likely due to differences in the hemagglutinin protein and the types of sialic acid residues to which the protein binds. Avian viruses tend to prefer sialic acid alpha(2-3) galactose, which, in humans, is found in the terminal bronchi and alveoli. Conversely, human viruses prefer sialic acid alpha(2-6) galactose, which is found on epithelial cells in the upper respiratory tract.
- ^ Leslie Taylor (2006). "Overestimating Avian Flu". Seed Magazine. Archived from the original on 2008-02-20. Retrieved 2008-01-07.
{{cite journal}}: CS1 maint: unfit URL (link) - ^ Anna Thorson, MD; Max Petzold; Nguyen Thi Kim Chuc; Karl Ekdahl, MD (2006). "Is Exposure to Sick or Dead Poultry Associated With Flulike Illness?". Arch Intern Med. 166 (1): 119–123. doi:10.1001/archinte.166.1.119. PMID 16401820.
- ^ de Jong MD, Bach VC, Phan TQ, Vo MH, Tran TT, Nguyen BH, Beld M, Le TP, Truong HK, Nguyen VV, Tran TH, Do QH, Farrar J (2005). "Fatal avian influenza A (H5N1) in a child presenting with diarrhea followed by coma". N. Engl. J. Med. 352 (7): 686–691. doi:10.1056/NEJMoa044307. PMID 15716562. S2CID 17703507.
- ^ Webster, R. G.; Walker, E. J. (2003). "The world is teetering on the edge of a pandemic that could kill a large fraction of the human population". American Scientist. 91 (2): 122. doi:10.1511/2003.2.122. Archived from the original on 2014-03-08.
- ^ Chan MC, Cheung CY, Chui WH, Tsao SW, Nicholls JM, Chan YO, Chan RW, Long HT, Poon LL, Guan Y, Peiris JS (2005). "Proinflammatory cytokine responses induced by influenza A (H5N1) viruses in primary human alveolar and bronchial epithelial cells". Respir. Res. 6 (1): 135. doi:10.1186/1465-9921-6-135. PMC 1318487. PMID 16283933.
- ^ Harder, T. C.; Werner, O. (2006). "Avian Influenza". In Kamps, B. S.; Hoffman, C.; Preiser, W. (eds.). Influenza Report 2006. Paris: Flying Publisher. ISBN 978-3-924774-51-6. Archived from the original on May 10, 2016.
- ^ Robert G. Webster; Elena A. Govorkova, M.D. (November 23, 2006). "H5N1 Influenza – Continuing Evolution and Spread". NEJM. 355 (21): 2174–2177. doi:10.1056/NEJMp068205. PMID 17124014.
- ^ "WHO changes H5N1 strains for pandemic vaccines, raising concern over virus evolution". Center for Infectious Disease Research and Policy (CIDRAP). August 18, 2006. Archived from the original on February 5, 2012.
- ^ "Antigenic and genetic characteristics of H5N1 viruses and candidate H5N1 vaccine viruses developed for potential use as pre-pandemic vaccines" (PDF). World Health Organization (WHO). August 18, 2006. Archived from the original (PDF) on August 24, 2006.
- ^ Salzberg, Steven L.; Kingsford, Carl; Cattoli, Giovanni; Spiro, David J.; Janies, Daniel A.; Aly, Mona Mehrez; Brown, Ian H.; Couacy-Hymann, Emmanuel; Mia, Gian Mario De; Dung, Do Huu; Guercio, Annalisa; Joannis, Tony; Ali, Ali Safar Maken; Osmani, Azizullah; Padalino, Iolanda (2007). "Genome Analysis Linking Recent European and African Influenza (H5N1) Viruses". Emerging Infectious Diseases. 13 (5): 713–718. doi:10.3201/eid1305.070013. PMC 2432181. PMID 17553249.
- ^ Payungporn S, Chutinimitkul S, Chaisingh A, Damrongwantanapokin S, Nuansrichay B, Pinyochon W, Amonsin A, Donis RO, Theamboonlers A, Poovorawan T (2006). "Discrimination between Highly Pathogenic and Low Pathogenic H5 Avian Influenza A Viruses". Emerging Infectious Diseases. 12 (4): 700–701. doi:10.3201/eid1204.051427. PMC 3294708. PMID 16715581.
- ^ Parker-Pope T (2009-11-05). "The Cat Who Got Swine Flu". The New York Times. Archived from the original on November 8, 2009. Retrieved 2010-04-05.
- ^ Couch, R. (1996). "Chapter 58. Orthomyxoviruses Multiplication". In Baron, S. (ed.). Medical Microbiology. Galveston: The University of Texas Medical Branch at Galveston. ISBN 978-0-9631172-1-2. Archived from the original on May 3, 2009.
- ^ Lang G.; Rouse B. T.; Narayan O.; Ferguson A. E.; Connell M. C. (1968). "A new influenza virus infection in turkeys. I. Isolation and characterization of virus 6213". Can Vet J. 9 (1): 22–29. PMC 1697084. PMID 17421891.
- ^ Ping Jihui; Selman Mohammed; Tyler Shaun; Forbes Nicole; Keleta Liya; Brown Earl G (2012). "Low-pathogenic avian influenza virus A/turkey/Ontario/6213/1966 (H5N1) is the progenitor of highly pathogenic A/turkey/Ontario/7732/1966 (H5N9)". J Gen Virol. 93 (Pt 8): 1649–1657. doi:10.1099/vir.0.042895-0. PMC 3541759. PMID 22592261.
- ^ "Avian Influenza Detected In British Columbia". Canadian Food Inspection Agency (CFIA). January 24, 2009. Archived from the original on January 31, 2009.
- ^ "Avian Influenza Low Pathogenic H5N1 vs. Highly Pathogenic H5N1 – Latest Update". United States Department of Agriculture (USDA). August 17, 2006. Archived from the original on April 9, 2010.
- ^ a b c Kou Z, Lei FM, Yu J, Fan ZJ, Yin ZH, Jia CX, Xiong KJ, Sun YH, Zhang XW, Wu XM, Gao XB, Li TX (2005). "New Genotype of Avian Influenza H5N1 Viruses Isolated from Tree Sparrows in China". J. Virol. 79 (24): 15460–15466. doi:10.1128/JVI.79.24.15460-15466.2005. PMC 1316012. PMID 16306617.
- ^ a b c The World Health Organization Global Influenza Program Surveillance Network. (2005). "Evolution of H5N1 avian influenza viruses in Asia". Emerging Infectious Diseases. 11 (10): 1515–1521. doi:10.3201/eid1110.050644. PMC 3366754. PMID 16318689.
Figure 1 shows a diagramatic representation of the genetic relatedness of Asian H5N1 hemagglutinin genes from various isolates of the virus - ^ Gambaryan A, Tuzikov A, Pazynina G, Bovin N, Balish A, Klimov A (2006). "Fatal Evolution of the receptor binding phenotype of influenza A (H5) viruses". Virology. 344 (2): 432–438. doi:10.1016/j.virol.2005.08.035. PMID 16226289.
- ^ Harder, T. C.; Werner, O. (2006). "Avian Influenza". In Kamps, B. S.; Hoffman, C.; Preiser, W. (eds.). Influenza Report 2006. Paris: Flying Publisher. ISBN 978-3-924774-51-6. Archived from the original on May 10, 2016.
This e-book is under constant revision and is an excellent guide to Avian Influenza - ^ Schultz J (2005-11-28). "Bird flu vaccine won't precede pandemic". United Press International. Archived from the original on February 15, 2006. Retrieved 2006-04-18.
- ^ Enserick, M. (2005-08-12). "Avian Influenza:'Pandemic Vaccine' Appears to Protect Only at High Doses". American Scientist. 91 (2): 122. doi:10.1511/2003.2.122. Archived from the original on 2006-02-28. Retrieved 2006-04-18.
- ^ Walker K (2006-01-27). "Two H5N1 human vaccine trials to begin". Science Daily. Archived from the original on 2006-02-14. Retrieved 2006-04-18.
- ^ Gao W, Soloff AC, Lu X, Montecalvo A, Nguyen DC, Matsuoka Y, Robbins PD, Swayne DE, Donis RO, Katz JM, Barratt-Boyes SM, Gambotto A (2006). "Protection of Mice and Poultry from Lethal H5N1 Avian Influenza Virus through Adenovirus-Based Immunization". J. Virol. 80 (4): 1959–1964. doi:10.1128/JVI.80.4.1959-1964.2006. PMC 1367171. PMID 16439551.
- ^ a b United States Agency for International Development (2006). "Avian Influenza Response: Key Actions to Date". Archived from the original on 2006-04-17. Retrieved 2006-04-18.
- ^ United States Department of Health and Human Services (2002). "Pandemicflu.gov Monitoring outbreaks". Archived from the original on 2006-04-26. Retrieved 2006-04-18.
- ^ Medline Plus (2006-01-12). "Oseltamivir (Systemic)". National Institutes of Health (NIH). Archived from the original on 2006-04-25. Retrieved 2006-04-18.
- ^ Associated Press, "Tamiflu is Set Aside for WHO," The Wall Street Journal, April 20, 2006, p. D6.
- ^ Integrated Regional Information Networks (2006-04-02). "Middle East: Interview with WHO experts Hassan al-Bushra and John Jabbour". Alertnet Reuters foundation. Archived from the original on 2006-04-07. Retrieved 2006-04-18.
- ^ Bernd Sebastian Kamps; Christian Hoffmann. "Zanamivir". Influenza Report. Archived from the original on 2006-10-27. Retrieved 2006-10-15.
- ^ Zheng B.-J. (June 10, 2008). "Delayed antiviral plus immunomodulator treatment still reduces mortality in mice infected by high inoculum of influenza A/H5N1 virus". Proceedings of the National Academy of Sciences. 105 (23): 8091–8096. Bibcode:2008PNAS..105.8091Z. doi:10.1073/pnas.0711942105. PMC 2430364. PMID 18523003.
- ^ "Oseltamivir-resistant H5N1 virus isolated from Vietnamese girl". Center for Infectious Disease Research and Policy (CIDRAP). October 14, 2005. Archived from the original on September 25, 2006. Retrieved 2006-10-15.
- ^ "U.N. Says Bird Flu Awareness Increases". National Public Radio (NPR). October 12, 2006. Retrieved 2006-10-15. [dead link]
- ^ Collins PJ, Haire LF, Lin YP, Liu J, Russell RJ, Walker PA, Skehel JJ, Martin SR, Hay AJ, Gamblin SJ (2008). "Crystal structures of oseltamivir-resistant influenza virus neuraminidase mutants". Nature. 453 (7199): 1258–1261. Bibcode:2008Natur.453.1258C. doi:10.1038/nature06956. PMID 18480754. S2CID 4383625.
- ^ Garcia-Sosa AT, Sild S, Maran U (2008). "Design of Multi-Binding-Site Inhibitors, Ligand Efficiency, and Consensus Screening of Avian Influenza H5N1 Wild-Type Neuraminidase and of the Oseltamivir-Resistant H274Y Variant". J. Chem. Inf. Model. 48 (10): 2074–2080. doi:10.1021/ci800242z. PMID 18847186.
- ^ "Cumulative Number of Confirmed Human Cases for Avian Influenza A/(H5N1) Reported to WHO, 2003–2011" (PDF). World Health Organization (WHO). Archived (PDF) from the original on 2011-10-27.
- ^ "Studies Spot Obstacle to Human Transmission of Bird Flu". Forbes. 2006-03-22. Archived from the original on May 23, 2006. Retrieved 2006-04-18.
- ^ Food and Agricultural Organization of the United Nations (2005). "Wild birds and Avian Influenza". Archived from the original on 2006-11-01. Retrieved 2006-04-18.
- ^ Brstilo M. (2006-01-19). "Highly Pathogenic Avian Influenza in Croatia Follow-up report No. 4". Archived from the original on 2006-05-14. Retrieved 2006-04-18.
- ^ European Food Safety Authority (2006-04-04). "Scientific Statement on Migratory birds and their possible role in the spread of highly pathogenic avian influenza" (PDF). Archived from the original (PDF) on 2006-05-07. Retrieved 2006-04-18.
- ^ "Bird flu may be spread indirectly, WHO says". Reuters. Reuters. 2008-01-17. Archived from the original on 2008-12-01. Retrieved 2009-09-01.
- ^ Sturm-Ramirez KM, Ellis T, Bousfield B, Bissett L, Dyrting K, Rehg JE, Poon L, Guan Y, Peiris M, Webster RG (2004). "Reemerging H5N1 Influenza Viruses in Hong Kong in 2002 Are Highly Pathogenic to Ducks". J. Virol. 78 (9): 4892–45901. doi:10.1128/JVI.78.9.4892-4901.2004. PMC 387679. PMID 15078970.
- ^ Li KS, Guan Y, Wang J, Smith GJ, Xu KM, Duan L, Rahardjo AP, Puthavathana P, Buranathai C, Nguyen TD, Estoepangestie AT, Chaisingh A, Auewarakul P, Long HT, Hanh NT, Webby RJ, Poon LL, Chen H, Shortridge KF, Yuen KY, Webster RG, Peiris JS (2004). "Genesis of a highly pathogenic and potentially pandemic H5N1 influenza virus in eastern Asia". Nature. 430 (6996): 209–213. Bibcode:2004Natur.430..209L. doi:10.1038/nature02746. PMID 15241415. S2CID 4410379.
This was reprinted in 2005: Li KS, Guan Y, Wang J, Smith GJ, Xu KM, Duan L, Rahardjo AP, Puthavathana P, Buranathai C, Nguyen TD, Estoepangestie AT, Chaisingh A, Auewarakul P, Long HT, Hanh NT, Webby RJ, Poon LL, Chen H, Shortridge KF, Yuen KY, Webster RG, Peiris JS (2005). "Today's Pandemic Threat: Genesis of a Highly Pathogenic and Potentially Pandemic H5N1 Influenza Virus in Eastern Asia". In Knobler SL, Mack A, Mahmoud A, Lemon SM (eds.). The Threat of Pandemic Influenza: Are We Ready? Workshop Summary (2005). Washington DC: The National Academies Press. pp. 116–130. Archived from the original on 2006-09-14. - ^ World Health Organization (2005-10-28). "H5N1 avian influenza: timeline" (PDF). Archived from the original (PDF) on 2011-07-27. Retrieved 2006-04-18.
- ^ Chen H, Deng G, Li Z, Tian G, Li Y, Jiao P, Zhang L, Liu Z, Webster RG, Yu K (2004). "The evolution of H5N1 influenza viruses in ducks in southern China". Proc. Natl. Acad. Sci. U.S.A. 101 (28): 10452–10457. Bibcode:2004PNAS..10110452C. doi:10.1073/pnas.0403212101. PMC 478602. PMID 15235128.
- ^ Beigel JH, Farrar J, Han AM, Hayden FG, Hyer R, de Jong MD, Lochindarat S, Nguyen TK, Nguyen TH, Tran TH, Nicoll A, Touch S, Yuen KY; Writing Committee of the World Health Organization (WHO) Consultation on Human Influenza A/H5. (2005). "Avian influenza A (H5N1) infection in humans". N. Engl. J. Med. 353 (13): 1374–1385. CiteSeerX 10.1.1.730.7890. doi:10.1056/NEJMra052211. PMID 16192482.
{{cite journal}}: CS1 maint: multiple names: authors list (link) CS1 maint: numeric names: authors list (link) - ^ Rosenthal E (2006-04-15). "Bird Flu Virus May Be Spread by Smuggling". The New York Times. Archived from the original on 2013-05-20. Retrieved 2006-04-18.
- ^ Gu, Jiang; Xie, Zhigang; Gao, Zhancheng; Liu, Jinhua; Korteweg, Christine; Ye, Juxiang; Lau, Lok Ting; Lu, Jie; Gao, Zifen; Zhang, Bo; McNutt, Michael A. (2007-09-29). "H5N1 infection of the respiratory tract and beyond: a molecular pathology study". The Lancet. 370 (9593): 1137–1145. doi:10.1016/S0140-6736(07)61515-3. ISSN 0140-6736. PMC 7159293. PMID 17905166.
- ^ "North Korea confirms bird flu outbreak at duck farm". Yonhap News. 2013-05-20. Archived from the original on 2014-04-07.
- ^ "Chile detects first case of bird flu in a human". Reuters. 2023-03-29. Retrieved 2023-03-30.
- ^ a b c d e "Avian Influenza Situation Reports". woah.org. World Organisation for Animal Health. Retrieved 26 March 2024.
- ^ Miller, Brittney J. (2022). "Why unprecedented bird flu outbreaks sweeping the world are concerning scientists". Nature. 606 (7912): 18–19. Bibcode:2022Natur.606...18M. doi:10.1038/d41586-022-01338-2. PMID 35618804. S2CID 249096351.
- ^ "Urgent action needed to address impacts of Avian Influenza. Outbreak of bird disease is worst on record".
- ^ "Wilde vogels bezwijken aan vogelgriep: 'Blijf uit de buurt van dode dieren'". 7 June 2022.
- ^ a b "Hundreds of seabirds being lost to avian flu in Shetland". BBC News. 6 June 2022.
- ^ "Bird Flu Update June 2022 – Bird flu updates – Our work – the RSPB Community". June 2022.
- ^ @StKildaNTS (June 8, 2022). "We completed the Great Skua census today on #StKilda and the impact of the current #AvianInfluenza outbreak is clea…" (Tweet) – via Twitter.
- ^ "Avian flu kills birds at St Kilda World Heritage Site". BBC News. 7 June 2022.
- ^ "Huge concern for Scotland's seabirds as number dying from Avian Influenza continues to increase".
- ^ "Dozens of dead birds in suspected flu outbreak at Highlands reserve". BBC News. 13 May 2022.
- ^ "Mass mortality of Dalmatian pelicans observed in Greece due to Avian influenza". 11 March 2022.
- ^ "Bird flu hits world's largest Dalmatian Pelican colony". 16 March 2022.
- ^ "Israel and UK facing record-breaking bird flu outbreaks". 10 January 2022.
- ^ "H5N1 HIGHLY PATHOGENIC AVIAN INFLUENZA IN POULTRY AND WILD BIRDS: WINTER OF 2021/2022 WITH FOCUS ON MASS MORTALITY OF WILD BIRDS IN UK AND ISRAEL" (PDF). cms.int. 2022-01-24. Retrieved 2023-03-02.
- ^ a b c Prater, Erin (8 February 2023). "The spillover of bird flu to mammals must be 'monitored closely,' WHO officials warn: 'We need to be ready to face outbreaks in humans'". Fortune. Archived from the original on 9 February 2023. Retrieved 9 February 2023.
- ^ a b c Nuki, Paul (2 February 2023). "How worried should we be about avian flu?". The Telegraph. Archived from the original on 2 February 2023. Retrieved 5 February 2023.
- ^ "Hundreds of birds dead or dying of avian flu land on Cape Breton shores". Canadian Broadcasting Corporation. 2022-06-06. Archived from the original on 2022-06-06. Retrieved 2023-03-02.
- ^ "Mortalities in colonial seabirds associated with a highly pathogenic H5N1 avian influenza virus in Quebec". 31 May 2022.
- ^ "Hundreds of Birds Found Dead of Bird Flu at Suburban Lake, Experts Fear Bigger Outbreak". 14 April 2022.
- ^ "Bald eagles dying, egg prices rising: Bird flu spreads to more than 30 states". NBC News. 16 April 2022.
- ^ "Avian Influenza Detected in Bald Eagles in Georgia | Department of Natural Resources Division".
- ^ "Sernanp despliega protocolo de monitoreo ante casos de aves y lobos marinos afectados por influenza aviar en áreas naturales protegidas" (in Spanish). National Service of Protected Areas by the State (SERNANP). 6 February 2023. Archived from the original on 7 February 2023. Retrieved 10 February 2023.
- ^ "Bird flu kills sea lions and thousands of pelicans in Peru's protected areas". Reuters. 21 February 2023. Archived from the original on 21 February 2023. Retrieved 21 February 2023.
- ^ Polansek, Tom (15 February 2023). "Bird flu spreads to new countries, threatens non-stop 'war' on poultry". Reuters. Archived from the original on 16 February 2023. Retrieved 17 February 2023.
- ^ Mano, Ana (22 May 2023). "Brazil declares 180-day animal health emergency amid avian flu cases in wild birds". Reuters. Archived from the original on 22 May 2023. Retrieved 23 May 2023.
- ^ Schnirring, Lisa (25 March 2024). "Sick cows in 2 states test positive for avian flu". University of Minnesota. CIDRAP. Retrieved 26 March 2024.
- ^ Schnirring, Lisa (1 April 2024). "Avian flu infects person exposed to sick cows in Texas". University of Minnesota. CIDRAP. Retrieved 2 April 2024.
- ^ Weston, Phoebe (27 February 2024). "Scientists confirm first cases of bird flu on mainland Antarctica". The Guardian. Retrieved 21 March 2024.
- ^ Harmon, Katherine (2011-09-19). "What Will the Next Influenza Pandemic Look Like?". Scientific American. Archived from the original on 2012-03-02. Retrieved 2012-01-23.
- ^ David Malakoff (March 30, 2012). "Breaking News: NSABB Reverses Position on Flu Papers". Science Insider. Archived from the original on June 30, 2012. Retrieved June 23, 2012.
- ^ Nell Greenfieldboyce (April 24, 2012). "Bird Flu Scientist has Applied for Permit to Export Research". NPR. Archived from the original on June 22, 2012. Retrieved June 23, 2012.
- ^ Nell Greenfieldboyce (June 21, 2012). "Journal Publishes Details on Contagious Bird Flu Created in Lab". National Public Radio (NPR). Archived from the original on June 22, 2012. Retrieved June 23, 2012.
- ^ "H5N1" (Special Issue). Science. June 21, 2012. Archived from the original on June 25, 2012. Retrieved June 23, 2012.
- ^ Herfst, S.; Schrauwen, E. J. A.; Linster, M.; Chutinimitkul, S.; De Wit, E.; Munster, V. J.; Sorrell, E. M.; Bestebroer, T. M.; Burke, D. F.; Smith, D. J.; Rimmelzwaan, G. F.; Osterhaus, A. D. M. E.; Fouchier, R. A. M. (2012). "Airborne Transmission of Influenza A/H5N1 Virus Between Ferrets". Science. 336 (6088): 1534–1541. Bibcode:2012Sci...336.1534H. doi:10.1126/science.1213362. PMC 4810786. PMID 22723413.
- ^ Brown, Eryn (June 21, 2012). "Scientists create bird flu that spreads easily among mammals". Los Angeles Times. Archived from the original on June 23, 2012. Retrieved June 23, 2012.
- ^ Keim, Brandon (2013-05-02). "Chinese Scientists Create New Mutant Bird-Flu Virus". Wired. ISSN 1059-1028. Retrieved 2023-02-07.
- ^ "Scientists Resume Efforts to Create Deadly Flu Virus, with US Government's Blessing". Forbes.
- ^ "From anthrax to bird flu – the dangers of lax security in disease-control labs". TheGuardian.com. 18 July 2014.
- ^ Pelley, Lauren (2 February 2023). "Bird flu keeps spreading beyond birds. Scientists worry it signals a growing threat to humans, too". CBC News. Archived from the original on 2 February 2023. Retrieved 5 February 2023.
- ^ Tufekci, Zeynep (3 February 2023). "An Even Deadlier Pandemic Could Soon Be Here". New York Times. Archived from the original on 5 February 2023. Retrieved 5 February 2023.
- ^ "2022-2023 Detections of Highly Pathogenic Avian Influenza in Mammals". Avian Influenza. USDA APHIS. Archived from the original on 30 January 2023. Retrieved 5 February 2023.
- ^ Merrick, Jane (1 February 2023). "Mass death of seals raises fears bird flu is jumping between mammals, threatening new pandemic". The i newspaper. Archived from the original on 3 February 2023. Retrieved 15 February 2023.
- ^ Kwan, Jacklin (22 January 2024). "Bird flu wipes out over 95% of southern elephant seal pups in 'catastrophic' mass death". livescience.com. Retrieved 23 January 2024.
- ^ Mallapaty, Smriti (2024-04-27). "Bird flu virus has been spreading in US cows for months, RNA reveals". Nature. doi:10.1038/d41586-024-01256-5. PMID 38678111.
- ^ Burrough, Eric; Magstadt, Drew; Main, Rodger (29 April 2024). "Highly Pathogenic Avian Influenza A(H5N1) Clade 2.3.4.4b Virus Infection in Domestic Dairy Cattle and Cats, United States, 2024". Emerging Infectious Diseases. 30 (7). doi:10.3201/eid3007.240508. PMID 38683888. Retrieved 30 April 2024.
- ^ "Avian Influenza Weekly Update Number 943" (PDF). World Health Organization. 19 April 2024. Retrieved 28 April 2024.
- ^ Newey, Sarah (24 February 2023). "Bird flu kills school girl and infects father – 11 others under observation". The Telegraph. Archived from the original on 24 February 2023. Retrieved 24 February 2023.
- ^ Mandavilli, Apoorva; Anthes, Emily (24 February 2023). "Cambodia Investigates After Father and Daughter Infected With Bird Flu". New York Times. Archived from the original on 24 February 2023. Retrieved 24 February 2023.
- ^ "Viruses in Cambodian bird flu cases identified as endemic clade". Reuters. 26 February 2023. Archived from the original on 26 February 2023. Retrieved 27 February 2023.
- ^ Strong, Matthew (1 March 2023). "Taiwan raises Cambodia travel alert after human H5N1 cases | Taiwan News | 2023-03-01 16:02:00". Taiwan News. Archived from the original on 3 March 2023. Retrieved 3 March 2023.
- ^ Cheang, Sopheng (1 March 2023). "Cambodia says recent bird flu cases not spread by humans". ABC News. Archived from the original on 1 March 2023. Retrieved 3 March 2023.
- ^ Rosenthal E, Bradsher K (2006-03-16). "Is Business Ready for a Flu Pandemic?". The New York Times. Archived from the original on 2013-05-02. Retrieved 2012-01-23.
- ^ State.gov Archived 2006-09-14 at the Wayback Machine
- ^ Newswire Archived May 17, 2008, at the Wayback Machine
- ^ US AID Archived 2008-08-15 at the Wayback Machine
- ^ "BMO Financial Group". .bmo.com. Archived from the original on 2009-05-03. Retrieved 2010-04-05.
- ^ "Council on Foreign Relations". Cfr.org. Archived from the original on 2008-10-13. Retrieved 2010-04-05.
- ^ Reuters [dead link] article Vietnam to unveil advanced plan to fight bird flu published on April 28, 2006
- ^ Poultry sector suffers despite absence of bird flu Archived March 30, 2006, at the Wayback Machine
- ^ Barber, Tony (2006-02-13). "Italy imposes controls after bird flu discovery". FT.com. Retrieved 2012-08-19.
- ^ India eNews[usurped] article Pakistani poultry industry demands 10-year tax holiday published May 7, 2006 says "Pakistani poultry farmers have sought a 10-year tax exemption to support their dwindling business after the detection of the H5N1 strain of bird flu triggered a fall in demand and prices, a poultry trader said."
- ^ International Institute for Sustainable Development (IISD) Archived 2006-04-27 at the Wayback Machine Scientific Seminar on Avian Influenza, the Environment and Migratory Birds on 10–11 April 2006 published 14 April 2006.
- ^ McNeil, Donald G. Jr. (March 28, 2006). "The response to bird flu: Too much or not enough? UN expert stands by his dire warnings". International Herald Tribune. Archived from the original on February 20, 2008.
Sources[edit]
- Analysis of the efficacy of an adjuvant-based inactivated pandemic H5N1 influenza virus vaccine. https://link.springer.com/article/10.1007%2Fs00705-019-04147-7 Ainur NurpeisovaEmail authorMarkhabat KassenovNurkuisa RametovKaissar TabynovGourapura J. RenukaradhyaYevgeniy VolginAltynay SagymbayAmanzhol MakbuzAbylay SansyzbayBerik Khairullin
Research Institute for Biological Safety Problems (RIBSP), Zhambyl Region, Republic of Kazakhstan
External links[edit]
- Influenza Research Database – Database of influenza genomic sequences and related information.
- WHO World Health Organization
- WHO's Avian Flu Facts Sheet for 2006
- Epidemic and Pandemic Alert and Response Guide to WHO's H5N1 pages
- Avian Influenza Resources (updated) – tracks human cases and deaths
- National Influenza Pandemic Plans
- WHO Collaborating Centres and Reference Laboratories Centers, names, locations, and phone numbers
- FAO Avian Influenza portal Archived 2012-01-26 at the Wayback Machine Information resources, animations, videos, photos
- FAO Food and Agriculture Organisation – Bi-weekly Avian Influenza Maps – tracks animal cases and deaths
- FAO Bird Flu disease card
- FAO Socio-Economic impact of AI Projects, Information resources
- OIE World Organisation for Animal Health – tracks animal cases and deaths
- Official outbreak reports by country Archived 2012-12-13 at the Wayback Machine
- Official outbreak reports by week
- Chart of outbreaks by country Archived 2012-04-19 at the Wayback Machine
- Health-EU Portal EU response to Avian Influenza.
- Avian influenza – Q & A's factsheet from European Centre for Disease Prevention and Control
- United Kingdom
- Exotic Animal Disease Generic Contingency Plan – DEFRA generic contingency plan for controlling and eradicating an outbreak of an exotic animal disease. PDF hosted by BBC.
- UK Influenza Pandemic Contingency Plan by the National Health Service – a government entity. PDF hosted by BBC
- UK Department of Health Archived 2009-07-09 at the Wayback Machine
- United States
- Center for Infectious Disease Research and Policy Archived 2013-06-17 at the Wayback Machine Avian Influenza (Bird Flu): Implications for Human Disease – An overview of Avian Influenza
- PandemicFlu.Gov U.S. Government's avian flu information site
- USAID U.S. Agency for International Development – Avian Influenza Response
- CDC, Centers for Disease Control and Prevention – responsible agency for avian influenza in humans in US – Facts About Avian Influenza (Bird Flu) and Avian Influenza A (H5N1) Virus
- USGS – NWHC National Wildlife Health Center – responsible agency for avian influenza in animals in US
- Wildlife Disease Information Node A part of the National Biological Information Infrastructure and partner of the NWHC, this agency collects and distributes news and information about wildlife diseases such as avian influenza and coordinates collaborative information sharing efforts.
- HHS U.S. Department of Health & Human Services's Pandemic Influenza Plan
