Umifenovir
You can help expand this article with text translated from the corresponding article in Russian. (June 2020) Click [show] for important translation instructions.
|
This article needs more reliable medical references for verification or relies too heavily on primary sources. (July 2020) |  |
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Arbidol |
| Other names | AR-1I9514 |
| Pregnancy category |
|
| Routes of administration | By mouth (hard capsules, tablets) |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 40% |
| Metabolism | Hepatic, CYP3A4[4] |
| Elimination half-life | 17–21 hours |
| Excretion | 40% excrete as unchanged umifenovir in feces (38.9%) and urine (0.12%)[3] |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| PDB ligand | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.247.800 |
| Chemical and physical data | |
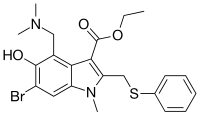
| Formula | C22H25BrN2O3S |
| Molar mass | 477.42 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Umifenovir, sold under the brand name Arbidol, is an antiviral medication for the treatment of influenza and COVID infections used in Russia[5] and China. The drug is manufactured by Pharmstandard (Russian: Фармстандарт). It is not approved by the U.S. Food and Drug Administration (FDA) for the treatment or prevention of influenza.[6]
Chemically, umifenovir features an indole core, functionalized at all but one positions with different substituents. The molecular groups of umifenovir - hydroxy, amino and carboxy - can interact to form various hydrogen-bonded synthons. Antiviral activity and acceptable cytotoxicity profiles [7][8] make it a promising candidate for further research as a potential therapeutic agent for the selective treatment of flavivirus infections [9]. Umifenovir is characterized by only one polymorphic form but can exist in the form of a large number of crystal solvates, the production of which depends on the medium and conditions of synthesis. In this case, the implementation of the crystal solvate or other solid form may be determined by the spatial structure and conformational equilibria in the saturated solution [10][11]. The drug has been shown in studies to inhibit viral entry into target cells [12] and stimulate the immune response.
Status[edit]
Testing of umifenovir's efficacy has mainly occurred in China and Russia,[13][14] and it is well known in these two countries.[15] Some of the Russian tests showed the drug to be effective[13] and a direct comparison with oseltamivir showed similar efficiency in vitro and in a clinical setting.[16] In 2010 Arbidol was the drug brand with the highest sales volume in Russia.[17] In the first quarter of 2020 it had a 16 percent share in the Russian antiviral market.[18]
Mode of action[edit]
Biochemistry[edit]
Umifenovir inhibits membrane fusion of influenza virus.[5] Umifenovir prevents contact between the virus and target host cells. Fusion between the viral envelope (surrounding the viral capsid) and the cell membrane of the target cell is inhibited. This prevents viral entry to the target cell, and therefore protects it from infection.[19]
Some evidence suggests that the drug's actions are more effective at preventing infections from RNA viruses than infections from DNA viruses.[20]
As well as specific antiviral action against both influenza A and influenza B viruses, umifenovir exhibits modulatory effects on the immune system. The drug stimulates a humoral immune response, induces interferon-production, and stimulates the phagocytic function of macrophages.[21]
Clinical application[edit]

Umifenovir is used primarily as an antiviral treatment for influenza. The drug has also been investigated as a candidate drug for treatment of hepatitis C.[22]
More recent studies indicate that umifenovir also has in vitro effectiveness at preventing entry of Zaire ebolavirus (Kikwit strain), Tacaribe arenavirus and Kaposi's sarcoma-associated herpesvirus in mammalian cell cultures, while confirming umifenovir's suppressive effect in vitro on Hepatitis B and poliovirus infection of mammalian cells when introduced either in advance of viral infection or during infection.[23][24]
Side effects[edit]
Side effects in children include sensitization to the drug. No known overdose cases have been reported and allergic reactions are limited to people with hypersensitivity. The LD50 is more than 4 g/kg.[25]
Criticism[edit]
In 2007, the Russian Academy of Medical Sciences stated that the effects of Arbidol (umifenovir) are not scientifically proven.[26]
Russian media criticized lobbying attempts by Tatyana Golikova (then-Minister of Healthcare) to promote umifenovir,[27] and the unsubstantiated claim that Arbidol can speed up recovery from flu or cold by 1.3-2.3 days.[28] They also made claims that comparative clinical studies have proven umifenovir to be inefficient.[29][30]
Names[edit]
Arbidol is the INN.[31]
The brand name Arbidol is known as Russian: Арбидол and Chinese: 阿比朵尔.[citation needed]
Research[edit]
In early 2020 umifenovir was touted as a potential treatment for COVID-19 in China, where it was sometimes given to patients in combination with other drugs such as the anti-HIV drug Darunavir.[32][33][34][35] A three-arm RCT study published in May 2020 in the Cell Press journal Clinical Advances found that neither Umifenovir or Lopinavir/Ritonavir helped patients with mild to moderate COVID-19.[36] A similar study comparing Umifenovir directly with Lopinavir/Ritonavir found no difference in fever duration between the two groups but found a lower viral load on day 14 in the Umifenovir group.[37] A systemic review and meta-analysis of 16 studies on Umifenovir published in March 2021 concluded that there is "no significant benefit of using Arbidol compared with non‐antiviral treatment or other therapeutic agents against COVID‐19 disease."[38]
References[edit]
- ^ a b "阿比朵尔抑制新冠,先声药业生产的吗?". 全民健康网 (in Chinese). Archived from the original on 17 June 2020. Retrieved 17 June 2020.
- ^ "ИНСТРУКЦИЯ ПО ПРИМЕНЕНИЮ АРПЕТОЛ (ARPETOL)". Vidal (in Belarusian). Archived from the original on 26 January 2018. Retrieved 17 June 2020.
- ^ "Full Prescribing Information: Arbidol (umifenovir) film-coated tablets 50 and 100 mg: Corrections and Additions". State Register of Medicines (in Russian). Open joint-stock company "Pharmstandard-Tomskchempharm". Retrieved 3 June 2015.[permanent dead link]
- ^ Deng P, Zhong D, Yu K, Zhang Y, Wang T, Chen X (April 2013). "Pharmacokinetics, metabolism, and excretion of the antiviral drug arbidol in humans". Antimicrobial Agents and Chemotherapy. 57 (4): 1743–1755. doi:10.1128/AAC.02282-12. PMC 3623363. PMID 23357765. S2CID 9534025.
- ^ a b Leneva IA, Russell RJ, Boriskin YS, Hay AJ (February 2009). "Characteristics of arbidol-resistant mutants of influenza virus: implications for the mechanism of anti-influenza action of arbidol". Antiviral Research. 81 (2): 132–140. doi:10.1016/j.antiviral.2008.10.009. PMID 19028526.
- ^ "FDA Approved Drugs for Influenza". U.S. Food and Drug Administration. 8 December 2022.
- ^ Leneva I, Kartashova N, Poromov A, Gracheva A, Korchevaya E, Glubokova E, et al. (August 2021). "Antiviral Activity of Umifenovir In Vitro against a Broad Spectrum of Coronaviruses, Including the Novel SARS-CoV-2 Virus". Viruses. 13 (8): 1665. doi:10.3390/v13081665. PMC 8402645. PMID 34452529.
- ^ Haviernik J, Štefánik M, Fojtíková M, Kali S, Tordo N, Rudolf I, et al. (April 2018). "Arbidol (Umifenovir): A Broad-Spectrum Antiviral Drug That Inhibits Medically Important Arthropod-Borne Flaviviruses". Viruses. 10 (4): 184. doi:10.3390/v10040184. PMC 5923478. PMID 29642580.
- ^ Qian X, Qi Z (June 2022). "Mosquito-Borne Flaviviruses and Current Therapeutic Advances". Viruses. 14 (6): 1226. doi:10.3390/v14061226. PMC 9229039. PMID 35746697.
- ^ Eventova VA, Belov KV, Efimov SV, Khodov IA (January 2023). "Conformational Screening of Arbidol Solvates: Investigation via 2D NOESY". Pharmaceutics. 15 (1): 226. doi:10.3390/pharmaceutics15010226. PMC 9865235. PMID 36678855.
- ^ Belov KV, Dyshin AA, Khodov IA (March 2024). "Conformational analysis of arbidol in supercritical carbon Dioxide: Insights into 'opened' and 'closed' conformer groups". Journal of Molecular Liquids. 397: 124074. doi:10.1016/j.molliq.2024.124074. S2CID 267232952.
- ^ Kadam RU, Wilson IA (January 2017). "Structural basis of influenza virus fusion inhibition by the antiviral drug Arbidol". Proceedings of the National Academy of Sciences of the United States of America. 114 (2): 206–214. Bibcode:2017PNAS..114..206K. doi:10.1073/pnas.1617020114. PMC 5240704. PMID 28003465.
- ^ a b Leneva IA, Fediakina IT, Gus'kova TA, Glushkov RG (2005). "[Sensitivity of various influenza virus strains to arbidol. Influence of arbidol combination with different antiviral drugs on reproduction of influenza virus A]". Terapevticheskii Arkhiv (Russian translation). 77 (8). ИЗДАТЕЛЬСТВО "МЕДИЦИНА": 84–88. PMID 16206613.
- ^ Wang MZ, Cai BQ, Li LY, Lin JT, Su N, Yu HX, et al. (June 2004). "[Efficacy and safety of arbidol in treatment of naturally acquired influenza]". Zhongguo Yi Xue Ke Xue Yuan Xue Bao. Acta Academiae Medicinae Sinicae. 26 (3): 289–293. PMID 15266832.
- ^ Boriskin YS, Leneva IA, Pécheur EI, Polyak SJ (2008). "Arbidol: a broad-spectrum antiviral compound that blocks viral fusion". Current Medicinal Chemistry. 15 (10): 997–1005. doi:10.2174/092986708784049658. PMID 18393857.
- ^ Leneva IA, Burtseva EI, Yatsyshina SB, Fedyakina IT, Kirillova ES, Selkova EP, et al. (February 2016). "Virus susceptibility and clinical effectiveness of anti-influenza drugs during the 2010-2011 influenza season in Russia". International Journal of Infectious Diseases. 43: 77–84. doi:10.1016/j.ijid.2016.01.001. PMID 26775570.
- ^ "Russian Pharmaceutical Market 2010" (PDF). DSM Group. Archived from the original (PDF) on 20 December 2018. Retrieved 17 June 2020.
- ^ "Most popular antiviral medication brands in the Russian pharmaceutical market in 1st quarter 2020 by sales value share". Statista. Retrieved 17 June 2020.
- ^ Boriskin YS, Pécheur EI, Polyak SJ (July 2006). "Arbidol: a broad-spectrum antiviral that inhibits acute and chronic HCV infection". Virology Journal. 3: 56. doi:10.1186/1743-422X-3-56. PMC 1559594. PMID 16854226.
- ^ Shi L, Xiong H, He J, Deng H, Li Q, Zhong Q, et al. (2007). "Antiviral activity of arbidol against influenza A virus, respiratory syncytial virus, rhinovirus, coxsackie virus and adenovirus in vitro and in vivo". Archives of Virology. 152 (8): 1447–1455. doi:10.1007/s00705-007-0974-5. PMID 17497238. S2CID 13595688.
- ^ Glushkov RG, Gus'kova TA, Krylova LI, Nikolaeva IS (1999). "[Mechanisms of arbidole's immunomodulating action]". Vestnik Rossiiskoi Akademii Meditsinskikh Nauk (in Russian) (3): 36–40. PMID 10222830.
- ^ Pécheur EI, Lavillette D, Alcaras F, Molle J, Boriskin YS, Roberts M, et al. (May 2007). "Biochemical mechanism of hepatitis C virus inhibition by the broad-spectrum antiviral arbidol". Biochemistry. 46 (20): 6050–6059. doi:10.1021/bi700181j. PMC 2532706. PMID 17455911.
- ^ Pécheur EI, Borisevich V, Halfmann P, Morrey JD, Smee DF, Prichard M, et al. (January 2016). "The Synthetic Antiviral Drug Arbidol Inhibits Globally Prevalent Pathogenic Viruses". Journal of Virology. 90 (6): 3086–3092. doi:10.1128/JVI.02077-15. PMC 4810626. PMID 26739045.
- ^ Hulseberg CE, Fénéant L, Szymańska-de Wijs KM, Kessler NP, Nelson EA, Shoemaker CJ, et al. (April 2019). "Arbidol and Other Low-Molecular-Weight Drugs That Inhibit Lassa and Ebola Viruses". Journal of Virology. 93 (8). doi:10.1128/JVI.02185-18. PMC 6450122. PMID 30700611.
- ^ "АРБИДОЛ (ARBIDOL)". Vidal. Archived from the original on 4 February 2009.
- ^ "Resolution". Meetings of the Presidium of the Formulary Committee. Russian Academy of Medical Sciences. 16 March 2007.
- ^ "How do we plant federal ministers". MKRU (in Russian). 21 April 2011.
- ^ Golunov I (23 December 2013). "13 most popular flu cures: do they work?". Professional Journalism Platform (in Russian).
- ^ Svetlana R. "Палка в колеса" [Stick in the wheel]. Esquire (in Russian).
- ^ "Повторение — мать учения" [Repetition - the mother of learning]. Esquire (in Russian).
- ^ Recommended INN: List 65., WHO Drug Information, Vol. 25, No. 1, 2011, page 91
- ^ Ng E (4 February 2020). "Coronavirus: are cocktail therapies for flu and HIV the magic cure?". South China Morning Post.
Bangkok and Hangzhou hospitals put combination remedies to the test.
- ^ Zheng W, Lau M (4 February 2020). "China's health officials say priority is to stop mild coronavirus cases from getting worse". South China Morning Post.
- ^ Lu H (March 2020). "Drug treatment options for the 2019-new coronavirus (2019-nCoV)". BioScience Trends. 14 (1): 69–71. doi:10.5582/bst.2020.01020. PMID 31996494.
- ^ Chen J, Ling Y, Xi X, Liu P, Li F, Li T, et al. (2020-02-21). "Efficacies of lopinavir/ritonavir and arbidol in the treatment of novel coronavirus pneumonia". Chinese Journal of Infectious Diseases (in Chinese). 38: E008. doi:10.3760/cma.j.cn311365-20200210-00050. ISSN 1000-6680.
- ^ Li Y, Xie Z, Lin W, Cai W, Wen C, Guan Y, et al. (December 2020). "Efficacy and Safety of Lopinavir/Ritonavir or Arbidol in Adult Patients with Mild/Moderate COVID-19: An Exploratory Randomized Controlled Trial". Med. 1 (1): 105–113.e4. doi:10.1016/j.medj.2020.04.001. PMC 7235585. PMID 32838353.
- ^ Zhu Z, Lu Z, Xu T, Chen C, Yang G, Zha T, et al. (July 2020). "Arbidol monotherapy is superior to lopinavir/ritonavir in treating COVID-19". The Journal of Infection. 81 (1): e21–e23. doi:10.1016/j.jinf.2020.03.060. PMC 7195393. PMID 32283143.
- ^ Amani B, Amani B, Zareei S, Zareei M (December 2021). "Efficacy and safety of arbidol (umifenovir) in patients with COVID-19: A systematic review and meta-analysis". Immunity, Inflammation and Disease. 9 (4): 1197–1208. doi:10.1002/iid3.502. PMC 8426686. PMID 34347937.
External links[edit]
- "Umifenovir". Drug Information Portal. U.S. National Library of Medicine.
