Nonmetal
| Nonmetals in their periodic table context |

|
| usually/always counted as a nonmetal[1][2][3] |
| sometimes counted as a nonmetal[4][a] |
| status as nonmetal or metal unconfirmed[5] |
| Part of a series on the |
| Periodic table |
|---|
Nonmetals are chemical elements that mostly lack distinctive metallic properties. They range from colorless gases like hydrogen to shiny crystals like iodine. Physically, they are usually lighter (less dense) than metals; brittle or crumbly if solid; and often poor conductors of heat and electricity. Chemically, nonmetals have high electronegativity (meaning they usually attract electrons in a chemical bond); and their oxides tend to be acidic.
Seventeen elements are widely recognized as nonmetals. Additionally, some or all of six borderline elements (metalloids) are sometimes counted as nonmetals.
The two lightest nonmetals, hydrogen and helium, together make up about 98% of the mass of the observable universe. Five nonmetallic elements—hydrogen, carbon, nitrogen, oxygen, and silicon—make up the bulk of Earth's oceans, atmosphere, biosphere, and crust.
The diverse properties of nonmetals enable a range of natural and technological uses. Hydrogen, oxygen, carbon, and nitrogen are essential building blocks for life. Industrial uses of nonmetals include electronics, energy storage, agriculture, and chemical production.
Most nonmetallic elements were not identified until the 18th and 19th centuries. While a distinction between metals and other minerals had existed since antiquity, a basic classification of chemical elements as metallic or nonmetallic emerged only in the late 18th century. Since then over two dozen properties have been suggested as criteria for distinguishing nonmetals from metals.
Definition and applicable elements[edit]
- Unless otherwise noted, this article describes the most stable form of an element in ambient conditions.[b]

Nonmetallic chemical elements are generally described as lacking properties common to metals, namely shininess, pliability, good thermal and electrical conductivity, and a general capacity to form basic oxides.[7][8] There is no widely-accepted precise definition;[9] any list of nonmetals is open to debate and revision.[1] The elements included depend on the properties regarded as most representative of nonmetallic or metallic character.
Fourteen elements are almost always recognized as nonmetals:[1][2]
Three more are commonly classed as nonmetals, but some sources list them as "metalloids",[3] a term which refers to elements regarded as intermediate between metals and nonmetals:[10]
One or more of the six elements most commonly recognized as metalloids are sometimes instead counted as nonmetals:
About 15–20% of the 118 known elements[11] are thus classified as nonmetals.[c]
General properties[edit]
Physical[edit]
of some nonmetallic elements
Nonmetals vary greatly in appearance, being colorless, colored or shiny. For the colorless nonmetals (hydrogen, nitrogen, oxygen, and the noble gases), their electrons are held sufficiently strongly so that no absorption of light happens in the visible part of the spectrum, and all visible light is transmitted.[14] The colored nonmetals (sulfur, fluorine, chlorine, bromine) absorb some colors (wavelengths) and transmit the complementary or opposite colors. For example, chlorine's "familiar yellow-green colour ... is due to a broad region of absorption in the violet and blue regions of the spectrum".[15][d] The shininess of boron, graphitic carbon, silicon, black phosphorus, germanium, arsenic, selenium, antimony, tellurium, and iodine[e] is a result of their structures featuring varying degrees of delocalized (free-moving) electrons that scatter incoming visible light.[18]
About half of nonmetallic elements are gases; most of the rest are solids. Bromine, the only liquid, is usually topped by a layer of its reddish-brown fumes. The gaseous and liquid nonmetals have very low densities, melting and boiling points, and are poor conductors of heat and electricity.[19] The solid nonmetals have low densities and low mechanical and structural strength (being brittle or crumbly),[20] and a wide range of electrical conductivity.[f]
This diversity in form stems from variability in internal structures and bonding arrangements. Nonmetals existing as discrete atoms like xenon, or as small molecules, such as oxygen, sulfur, and bromine, have low melting and boiling points; many are gases at room temperature, as they are held together by weak London dispersion forces acting between their atoms or molecules.[24] In contrast, nonmetals that form giant structures, such as chains of up to 1,000 selenium atoms,[25] sheets of carbon atoms in graphite,[26] or three-dimensional lattices of silicon atoms[27] have higher melting and boiling points, and are all solids, as it takes more energy to overcome their stronger covalent bonds.[28] Nonmetals closer to the left or bottom of the periodic table (and so closer to the metals) often have some weak metallic interactions between their molecules, chains, or layers; this occurs in boron,[29] carbon,[30] phosphorus,[31] arsenic,[32] selenium,[33] antimony,[34] tellurium[35] and iodine.[36]
| Aspect | Metals | Nonmetals |
|---|---|---|
| Appearance and form |
Shiny if freshly prepared or fractured; few colored;[37] all but one solid[38] |
Shiny, colored or transparent;[39] all but one solid or gaseous[38] |
| Density | Often higher | Often lower |
| Elasticity | Mostly malleable and ductile |
Brittle if solid |
| Electrical conductivity[40] |
Good | Poor to good |
| Electronic structure[41] |
Metallic or semimetalic | Semimetallic, semiconductor, or insulator |
The structures of nonmetallic elements differ from those of metals primarily due to variations in valence electron numbers and atomic size. Metals typically have fewer valence electrons than available orbitals, leading them to share electrons with many nearby atoms, resulting in centrosymmetrical crystalline structures.[42] In contrast, nonmetals share only the electrons required to achieve a noble gas electron configuration.[43] For example, nitrogen forms diatomic molecules featuring a triple bonds between each atom, both of which thereby attain the configuration of the noble gas neon. Antimony's larger atomic size prevents triple bonding, resulting in buckled layers in which each antimony atom is singly bonded with three other nearby atoms.[44]
The electrical and thermal conductivities of nonmetals, along with the brittle nature of solid nonmetals, are likewise related to their internal arrangements. Whereas good conductivity and plasticity (malleability, ductility) are ordinarily associated with the presence of free-moving and evenly distributed electrons in metals,[45] the electrons in nonmetals typically lack such mobility.[46] Among nonmetallic elements, good electrical and thermal conductivity is seen only in carbon (as graphite, along its planes), arsenic, and antimony.[g] Good thermal conductivity otherwise occurs only in boron, silicon, phosphorus, and germanium;[21] such conductivity is transmitted though vibrations of the crystalline lattices of these elements.[47] Moderate electrical conductivity is observed in the semiconductors[48] boron, silicon, phosphorus, germanium, selenium, tellurium, and iodine. Plasticity occurs under limited circumstances in carbon, as seen in exfoliated (expanded) graphite[49][50] and carbon nanotube wire,[51] in white phosphorus (soft as wax, pliable and can be cut with a knife, at room temperature),[52] in plastic sulfur,[53] and in selenium which can be drawn into wires from its molten state.[54]
The physical differences between metals and nonmetals arise from internal and external atomic forces. Internally, the positive charge stemming from the protons in an atom's nucleus acts to hold the atom's outer electrons in place. Externally, the same electrons are subject to attractive forces from protons in neighboring atoms. When the external forces are greater than, or equal to, the internal force, the outer electrons are expected to become relatively free to move between atoms, and metallic properties are predicted. Otherwise nonmetallic properties are expected.[55]
Allotropes[edit]
Over half of the nonmetallic elements exhibit a range of less stable allotropic forms, each with distinct physical properties.[56] For example, carbon, the most stable form of which is graphite, can manifest as diamond, buckminsterfullerene,[57] and amorphous[58] and paracrystalline (mixed amorphous and crystalline)[59] variations. Allotropes also occur for nitrogen, oxygen, phosphorus, sulfur, selenium, the six metalloids, and iodine.[60]
Chemical[edit]

| Aspect | Metals | Nonmetals | |
|---|---|---|---|
| Reactivity[61] | Wide range: very reactive to noble | ||
| Oxides | lower | Basic | Acidic; never basic[62] |
| higher | Increasingly acidic | ||
| Compounds with metals[63] |
Alloys | Ionic compounds | |
| Ionization energy[64] | Low to high | Moderate to very high | |
| Electronegativity[65] | Low to high | Moderate to very high | |
Nonmetals have relatively high values of electronegativity, and their oxides are therefore usually acidic. Exceptions may occur if a nonmetal is not very electronegative, or if its oxidation state is low, or both. These non-acidic oxides of nonmetals may be amphoteric (like water, H2O[66]) or neutral (like nitrous oxide, N2O[67][h]), but never basic (as is common with metals).
Nonmetals tend to gain or share electrons during chemical reactions, in contrast to metals which tend to donate electrons. This behavior is closely related to the stability of electron configurations in the noble gases, which have complete outer shells. Nonmetals generally gain enough electrons to attain the electron configuration of the following noble gas, while metals tend to lose electrons, in some cases achieving the electron configuration of the preceding noble gas. These tendencies in nonmetallic elements are succinctly summarized by the duet and octet rules of thumb.[70]
They typically exhibit higher ionization energies, electron affinities, and standard electrode potentials than metals. Generally, the higher these values are (including electronegativity) the more nonmetallic the element tends to be.[71] For example, the chemically very active nonmetals fluorine, chlorine, bromine, and iodine have an average electronegativity of 3.19—a figure[i] higher than that of any individual metal. On the other hand, the 2.05 average of the chemically weak metalloid nonmetals[j] falls within the 0.70 to 2.54 range of metals.[65]
The chemical distinctions between metals and nonmetals primarily stem from the attractive force between the positive nuclear charge of an individual atom and its negatively charged outer electrons. From left to right across each period of the periodic table, the nuclear charge increases in tandem with the number of protons in the atomic nucleus.[72] Consequently, there is a corresponding reduction in atomic radius[73] as the heightened nuclear charge draws the outer electrons closer to the nucleus core.[74] In metals, the impact of the nuclear charge is generally weaker compared to nonmetallic elements. As a result, in chemical bonding, metals tend to lose electrons, leading to the formation of positively charged ions or polarized atoms, while nonmetals tend to gain these electrons due to their stronger nuclear charge, resulting in negatively charged ions or polarized atoms.[75]
The number of compounds formed by nonmetals is vast.[76] The first 10 places in a "top 20" table of elements most frequently encountered in 895,501,834 compounds, as listed in the Chemical Abstracts Service register for November 2, 2021, were occupied by nonmetals. Hydrogen, carbon, oxygen, and nitrogen collectively appeared in most (80%) of compounds. Silicon, a metalloid, ranked 11th. The highest-rated metal, with an occurrence frequency of 0.14%, was iron, in 12th place.[77] A few examples of nonmetal compounds are: boric acid (H
3BO
3), used in ceramic glazes;[78] selenocysteine (C
3H
7NO
2Se), the 21st amino acid of life;[79] phosphorus sesquisulfide (P4S3), found in strike anywhere matches;[80] and teflon ((C
2F
4)n), used to create non-stick coatings for pans and other cookware.[81]
Complications[edit]
Adding complexity to the chemistry of the nonmetals are anomalies occurring in the first row of each periodic table block; non-uniform periodic trends; higher oxidation states; multiple bond formation; and property overlaps with metals.
First row anomaly[edit]
| Condensed periodic table highlighting the first row of each block: s p d and f | |||||||||||||
| Period | s-block | ||||||||||||
| 1 | H 1 |
He 2 |
p-block | ||||||||||
| 2 | Li 3 |
Be 4 |
B 5 |
C 6 |
N 7 |
O 8 |
F 9 |
Ne 10 | |||||
| 3 | Na 11 |
Mg 12 |
d-block |
Al 13 |
Si 14 |
P 15 |
S 16 |
Cl 17 |
Ar 18 | ||||
| 4 | K 19 |
Ca 20 |
Sc-Zn 21-30 |
Ga 31 |
Ge 32 |
As 33 |
Se 34 |
Br 35 |
Kr 36 | ||||
| 5 | Rb 37 |
Sr 38 |
f-block |
Y-Cd 39-48 |
In 49 |
Sn 50 |
Sb 51 |
Te 52 |
I 53 |
Xe 54 | |||
| 6 | Cs 55 |
Ba 56 |
La-Yb 57-70 |
Lu-Hg 71-80 |
Tl 81 |
Pb 82 |
Bi 83 |
Po 84 |
At 85 |
Rn 86 | |||
| 7 | Fr 87 |
Ra 88 |
Ac-No 89-102 |
Lr-Cn 103-112 |
Nh 113 |
Fl 114 |
Mc 115 |
Lv 116 |
Ts 117 |
Og 118 | |||
| Group | (1) | (2) | (3-12) | (13) | (14) | (15) | (16) | (17) | (18) | ||||
| The first-row anomaly strength by block is s >> p > d > f.[82][k] | |||||||||||||
Starting with hydrogen, the first row anomaly primarily arises from the electron configurations of the elements concerned. Hydrogen is particularly notable for its diverse bonding behaviors. It most commonly forms covalent bonds, but it can also lose its single electron in an aqueous solution, leaving behind a bare proton with tremendous polarizing power.[83] Consequently, this proton can attach itself to the lone electron pair of an oxygen atom in a water molecule, laying the foundation for acid-base chemistry.[84] Moreover, a hydrogen atom in a molecule can form a second, albeit weaker, bond with an atom or group of atoms in another molecule. Such bonding, "helps give snowflakes their hexagonal symmetry, binds DNA into a double helix; shapes the three-dimensional forms of proteins; and even raises water's boiling point high enough to make a decent cup of tea."[85]
Hydrogen and helium, as well as boron through neon, have unusually small atomic radii. This phenomenon arises because the 1s and 2p subshells lack inner analogues (meaning there is no zero shell and no 1p subshell), and they therefore experience no electron repulsion effects, unlike the 3p, 4p, and 5p subshells of heavier elements.[86] As a result, ionization energies and electronegativities among these elements are higher than what periodic trends would otherwise suggest. The compact atomic radii of carbon, nitrogen, and oxygen facilitate the formation of double or triple bonds.[87]
While it would normally be expected, on electron configuration consistency grounds, that hydrogen and helium would be placed atop the s-block elements, the significant first row anomaly shown by these two elements justifies alternative placements. Hydrogen is occasionally positioned above fluorine, in group 17, rather than above lithium in group 1. Helium is commonly placed above neon, in group 18, rather than above beryllium in group 2.[88]
Secondary periodicity[edit]
An alternation in certain periodic trends, sometimes referred to as secondary periodicity, becomes evident when descending groups 13 to 15, and to a lesser extent, groups 16 and 17.[89][l] Immediately after the first row of d-block metals, from scandium to zinc, the 3d electrons in the p-block elements—specifically, gallium (a metal), germanium, arsenic, selenium, and bromine—prove less effective at shielding the increasing positive nuclear charge. This same effect is observed with the emergence of fourteen f-block metals located between barium and lutetium, ultimately leading to atomic radii that are smaller than expected for elements from hafnium (Hf) onward.[91]
The Soviet chemist Shchukarev gives two more tangible examples:[92]
- "The toxicity of some arsenic compounds, and the absence of this property in analogous compounds of phosphorus [P] and antimony [Sb]; and the ability of selenic acid [H2SeO4] to bring metallic gold [Au] into solution, and the absence of this property in sulfuric [H2SO4] and [H2TeO4] acids."
Higher oxidation states[edit]
Some nonmetallic elements exhibit oxidation states that deviate from those predicted by the octet rule, which typically results in a valency of –3 in group 15, –2 in group 16, –1 in group 17, and 0 in group 18. Examples of such states can include compounds like ammonia (NH3), hydrogen sulfide (H2S), hydrogen fluoride (HF), and elemental xenon (Xe). Meanwhile, the maximum possible oxidation state increases from +5 in group 15, to +8 in group 18. The +5 oxidation state is observable from period 2 onward, in compounds such as nitric acid (HNO3) and phosphorus pentafluoride (PCl5).[m] Higher oxidation states in later groups emerge from period 3 onwards, as seen in sulfur hexafluoride (SF6), iodine heptafluoride (IF7), and xenon tetroxide (XeO4). For heavier nonmetals, their larger atomic radii and lower electronegativity values enable the formation of compounds with higher oxidation numbers, supporting higher bulk coordination numbers.[93]
Multiple bond formation[edit]
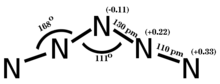
Period 2 nonmetals, particularly carbon, nitrogen, and oxygen, show a propensity to form multiple bonds. The compounds formed by these elements often exhibit unique stoichiometries and structures, as seen in the various nitrogen oxides,[93] which are not commonly found in elements from later periods.
Property overlaps[edit]
While certain elements have traditionally been classified as nonmetals and others as metals, some overlapping of properties occurs. Writing early in the twentieth century, by which time the era of modern chemistry had been well-established,[95] Humphrey[96] observed that:
- ... these two groups, however, are not marked off perfectly sharply from each other; some nonmetals resemble metals in certain of their properties, and some metals approximate in some ways to the non-metals.

Examples of metal-like properties occurring in nonmetallic elements include:
- silicon has an electronegativity (1.9) comparable with metals such as cobalt (1.88), copper (1.9), nickel (1.91) and silver (1.93);[65]
- the electrical conductivity of graphite exceeds that of some metals;[o]
- selenium can be drawn into a wire;[54]
- radon is the most metallic of the noble gases and begins to show some cationic behavior, which is unusual for a nonmetal;[100] and
- in extreme conditions, just over half of nonmetallic elements can form homopolyatomic cations.[p]
Examples of nonmetal-like properties occurring in metals are:
- Tungsten displays some nonmetallic properties, being brittle, having a high electronegativity, and forming only anions in aqueous solution,[102] and predominately acidic oxides.[8][103] These are characteristics more aligned with nonmetals. Even so, tungsten is classified as a metal, illustrating the spectrum of behaviors elements can exhibit within their classifications.
- Gold, the "king of metals" demonstrates several nonmetallic behaviors. It has the highest electrode potential among metals, suggesting a preference for gaining rather than losing electrons. Gold's ionization energy is one of the highest among metals, and its electron affinity and electronegativity are high, with the latter exceeding that of some nonmetals. It forms the Au– auride anion and exhibits a tendency to bond to itself, behaviors which are unexpected for metals. In aurides (MAu, where M = Li–Cs), gold's behavior is similar to that of a halogen, thereby bridging the traditional metal-nonmetal divide.[104]
A relatively recent development involves certain compounds of heavier p-block elements, such as silicon, phosphorus, germanium, arsenic and antimony, exhibiting behaviors typically associated with transition metal complexes. This phenomenon is linked to a small energy gap between their filled and empty molecular orbitals, which are the regions in a molecule where electrons reside and where they can be available for chemical reactions. In such compounds, this closer energy alignment allows for unusual reactivity with small molecules like hydrogen (H2), ammonia (NH3), and ethylene (C2H4), a characteristic previously observed primarily in transition metal compounds. These reactions may open new avenues in catalytic applications.[105]
Types [edit]
Nonmetal classification schemes vary widely, with some accommodating as few as two subtypes and others identifying up to seven. For example, the periodic table in the Encyclopaedia Britannica recognizes noble gases, halogens, and other nonmetals, and splits the elements commonly recognized as metalloids between "other metals" and "other nonmetals".[106] On the other hand, seven of twelve color categories on the Royal Society of Chemistry periodic table include nonmetals.[107][q]
| Group (1, 13−18) | Period | ||||||
| 13 | 14 | 15 | 16 | 1/17 | 18 | (1−6) | |
| H | He | 1 | |||||
| B | C | N | O | F | Ne | 2 | |
| Si | P | S | Cl | Ar | 3 | ||
| Ge | As | Se | Br | Kr | 4 | ||
| Sb | Te | I | Xe | 5 | |||
| Rn | 6 | ||||||
Starting on the right side of the periodic table, three types of nonmetals can be recognized:
The elements in a fourth set are sometimes recognized as nonmetals:
While many of the early workers attempted to classify elements none of their classifications were satisfactory. They were divided into metals and nonmetals, but some were soon found to have properties of both. These were called metalloids. This only added to the confusion by making two indistinct divisions where one existed before.[128]
Whiteford & Coffin 1939, Essentials of College Chemistry
The boundaries between these types are not sharp.[v] Carbon, phosphorus, selenium, and iodine border the metalloids and show some metallic character, as does hydrogen.
The greatest discrepancy between authors occurs in metalloid "frontier territory".[130] Some consider metalloids distinct from both metals and nonmetals, while others classify them as nonmetals.[4] Some categorize certain metalloids as metals (e.g., arsenic and antimony due to their similarities to heavy metals).[131][w] Metalloids resemble the elements universally considered "nonmetals" in having relatively low densities, high electronegativity, and similar chemical behavior.[127][x]
For context, the metallic side of the periodic table also ranges widely in reactivity.[y] Highly reactive metals fill most of the s- and f-blocks on the left,[z] bleeding into the early part of the d-block. Thereafter, reactivity generally decreases closer to the p-block, whose metals are not particularly reactive.[aa] The very unreactive noble metals, such as platinum and gold, are clustered in an island within the d-block.[137]
Noble gases[edit]

Six nonmetals are classified as noble gases: helium, neon, argon, krypton, xenon, and the radioactive radon. In conventional periodic tables they occupy the rightmost column. They are called noble gases due to their exceptionally low chemical reactivity.[108]
These elements exhibit remarkably similar properties, characterized by their colorlessness, odorlessness, and nonflammability. Due to their closed outer electron shells, noble gases possess feeble interatomic forces of attraction, leading to exceptionally low melting and boiling points.[138] As a consequence, they all exist as gases under standard conditions, even those with atomic masses surpassing many typically solid elements.[139]
Chemically, the noble gases exhibit relatively high ionization energies, negligible or negative electron affinities, and high to very high electronegativities. The number of compounds formed by noble gases is in the hundreds and continues to expand,[140] with most of these compounds involving the combination of oxygen or fluorine with either krypton, xenon, or radon.[141]
Halogen nonmetals[edit]
While the halogen nonmetals are notably reactive and corrosive elements, they can also be found in everyday compounds like toothpaste (NaF); common table salt (NaCl); swimming pool disinfectant (NaBr); and food supplements (KI). The term "halogen" itself means "salt former".[142]
Physically, fluorine and chlorine exist as pale yellow and yellowish-green gases, respectively, while bromine is a reddish-brown liquid, typically covered by a layer of its fumes; iodine is a solid and under white light is metallic-looking.[143] Electrically, the first three elements function as insulators while iodine behaves as a semiconductor (along its planes).[144]
Chemically, the halogen nonmetals exhibit high ionization energies, electron affinities, and electronegativity values, and are mostly relatively strong oxidizing agents.[145] These characteristics contribute to their corrosive nature.[146] All four elements tend to form primarily ionic compounds with metals,[147] in contrast to the remaining nonmetals (except for oxygen) which tend to form primarily covalent compounds with metals.[ab] The highly reactive and strongly electronegative nature of the halogen nonmetals epitomizes nonmetallic character.[151]
Unclassified nonmetals[edit]

After classifying the nonmetallic elements into noble gases and halogens, but before encountering the metalloids, there are seven nonmetals: hydrogen, carbon, nitrogen, oxygen, phosphorus, sulfur, and selenium.
In their most stable forms, three of these are colorless gases (hydrogen, nitrogen, oxygen); three are metallic looking solids (carbon, phosphorus, selenium); and one is a yellow solid (sulfur). Electrically, graphitic carbon behaves as a semimetal along its planes[153] and a semiconductor perpendicular to its planes;[154] phosphorus and selenium are semiconductors;[155] while hydrogen, nitrogen, oxygen, and sulfur are insulators.[ac]
These elements are often considered too diverse to merit a collective name,[157] and have been referred to as other nonmetals,[158] or simply as nonmetals.[159] As a result, their chemistry is typically taught disparately, according to their respective periodic table groups:[160] hydrogen in group 1; the group 14 nonmetals (including carbon, and possibly silicon and germanium); the group 15 nonmetals (including nitrogen, phosphorus, and possibly arsenic and antimony); and the group 16 nonmetals (including oxygen, sulfur, selenium, and possibly tellurium). Authors may choose other subdivisions based on their preferences.[ad]
Hydrogen, in particular, behaves in some respects like a metal and in others like a nonmetal.[162] Like a metal it can, for example, form a solvated cation in aqueous solution;[163] it can substitute for alkali metals in compounds such as the chlorides (NaCl cf. HCl) and nitrates (KNO3 cf. HNO3), and in certain alkali metal organometallic structures;[164] and it can form alloy-like hydrides with some transition metals.[165] Conversely, it is an insulating diatomic gas, akin to the nonmetals nitrogen, oxygen, fluorine and chlorine. In chemical reactions, it tends to ultimately attain the electron configuration of helium (the following noble gas) behaving in this way as a nonmetal.[166] It attains this configuration by forming a covalent or ionic bond[167] or, if it has initially given up its electron, by attaching itself to a lone pair of electrons.[168]
Some or all of these nonmetals share several properties. Being generally less reactive than the halogens,[169] most of them can occur naturally in the environment.[170] They have significant roles in biology[171] and geochemistry.[157] Collectively, their physical and chemical characteristics can be described as "moderately non-metallic".[157] However, they all have corrosive aspects. Hydrogen can corrode metals. Carbon corrosion can occur in fuel cells.[172] Acid rain is caused by dissolved nitrogen or sulfur. Oxygen causes iron to corrode via rust. White phosphorus, the most unstable form, ignites in air and leaves behind phosphoric acid residue.[173] Untreated selenium in soils can lead to the formation of corrosive hydrogen selenide gas.[174] When combined with metals, the unclassified nonmetals can form high-hardness (interstitial or refractory) compounds[175] due to their relatively small atomic radii and sufficiently low ionization energies.[157] They also exhibit a tendency to bond to themselves, particularly in solid compounds.[176] Additionally, diagonal periodic table relationships among these nonmetals mirror similar relationships among the metalloids.[177]
Metalloids[edit]
The six elements more commonly recognized as metalloids are boron, silicon, germanium, arsenic, antimony, and tellurium, all of which have a metallic appearance. (Other elements appearing less commonly on lists of metalloids include carbon, aluminium, selenium and polonium; these have both metallic and nonmetallic properties, but one or the other predominates.) In the periodic table, metalloids occupy a diagonal region within the p-block extending from boron at the upper left to tellurium at the lower right, along the dividing line between metals and nonmetals shown on some tables.[3]
Metalloids are brittle and poor-to-good conductors of heat and electricity. Specifically, boron, silicon, germanium, and tellurium are semiconductors. Arsenic and antimony have the electronic band structures of semimetals, although both have less stable semiconducting allotropes: arsenic as arsenolamprite, an extremely rare naturally occurring form;[178] and antimony in its synthetic thin-film amorphous form.[3][179]
Chemically, metalloids generally behave like weak nonmetals. Among the nonmetallic elements they tend to have the lowest ionization energies, electron affinities, and electronegativity values, and are relatively weak oxidizing agents. Additionally, they tend to form alloys when combined with metals.[3]
Abundance, sources, and uses[edit]
Abundance[edit]
| Universe[180] | hydrogen 70.5%, helium 27.5% | oxygen 1% |
| Atmosphere[181] | nitrogen 78%, oxygen 21% | argon 0.5% |
| Hydrosphere[181] | oxygen 66.2%, hydrogen 33.2% | chlorine 0.3% |
| Biomass[182] | oxygen 63%, carbon 20%, hydrogen 10% |
nitrogen 3.0% |
| Crust[181] | oxygen 61%, silicon 20% | hydrogen 2.9% |
Hydrogen and helium dominate the observable universe, making up an estimated 98% of all ordinary matter by mass.[ae] Oxygen, the next most abundant element, accounts for about 1%.[184]
Five nonmetals—hydrogen, carbon, nitrogen, oxygen, and silicon—form the bulk of the directly observable structure of the earth: about 84% of the crust, 96% of the biomass, and over 99% of the atmosphere and hydrosphere, as shown in the accompanying table.[181][182]
The Earth's mantle and core, making up about 99% of the Earth's volume,[185] are estimated to be made up of oxygen (31% by weight) and silicon (16%), with the remainder largely composed of the metals iron (31%), magnesium (15%) and nickel (2%).[186][af]
Sources[edit]
| Group (1, 13−18) | Period | ||||||
| 13 | 14 | 15 | 16 | 1/17 | 18 | (1−6) | |
| H | He | 1 | |||||
| B | C | N | O | F | Ne | 2 | |
| Si | P | S | Cl | Ar | 3 | ||
| Ge | As | Se | Br | Kr | 4 | ||
| Sb | Te | I | Xe | 5 | |||
| Rn | 6 | ||||||
Nonmetals and metalloids are extracted from a variety of raw materials:[170]
Uses[edit]
| Nearly all nonmetals[ah] have uses in:[189][190] Household goods, lighting and lasers, and medicine and pharmaceuticals |
| Most nonmetals have uses in:[189][191] Agrochemicals, dyestuffs and smart phones |
| Some nonmetals have uses in or as:[189][192] Alloys, cryogenics and refrigerants, explosives, fire retardants, fuel cells, inert air replacements, insulation (thermal & electric), mineral acids, nuclear control rods, photography, plastics, plug-in hybrid vehicles, solar cells, water treatment, welding gases, and vulcanization |
| Metalloids have uses in:[193] Alloys, ceramics, oxide glasses, solar cells, and semiconductors |
The great variety of physical and chemical properties of nonmetals[194] enable a wide range of natural and technological uses, as shown in the accompanying table. In living organisms, hydrogen, oxygen, carbon, and nitrogen serve as the foundational building blocks of life.[195] Some key technological uses of nonmetallic elements are in lighting and lasers, medicine and pharmaceuticals, and ceramics and plastics.
Some specific uses of later-discovered or rarer nonmetallic elements include:
- Boron, first produced in a pure form in 1909,[196] is used in the form of high-strength fibers for aerospace components and certain sporting goods.[197] It is also added to steel alloys to improve hardenability.[198]
- Black phosphorus, first reported in 1916,[199] is employed mainly in high-performance electronic devices, including field-effect transistors (FETs), owing to its exceptional charge carrier mobility. It has potential applications in photodetectors, optoelectronic devices, advanced solar cells and thermoelectric materials.[200]
- Germanium, thought to be a metal up until the 1930s,[201] was historically used in electronics, particularly early transistors and diodes, and still has roles in specialized high-frequency electronics. It is used in the production of infrared optical components for thermal imaging and spectroscopy.[202]
- Xenon, one of the rarest elements on Earth,[203] finds use in high-intensity discharge lamps for bright white light in automotive headlights and marine lighting. Additionally, it serves as a contrast agent in medical imaging techniques like xenon computed tomography and xenon-enhanced magnetic resonance imaging. In space exploration, xenon is a propellant for ion thrusters, known for their efficiency.[204]
- Radon, the rarest noble gas,[205] was formerly used in radiography and radiation therapy. Usually, radium in either an aqueous solution or as a porous solid was stored in a glass vessel. The radium decayed to produce radon, which was pumped off, filtered, and compressed into a small tube every few days. The tube was then sealed and removed. It was a source of gamma rays, which came from bismuth-214, one of radon's decay products.[206] In radiotherapy, radon has now been replaced by 137cesium, 192iridium, and 103palladium.[207]
History, background, and taxonomy[edit]
Discovery[edit]

While most nonmetallic elements were identified during the 18th and 19th centuries, a few were recognized much earlier. Carbon, sulfur, and antimony were known in antiquity. Arsenic was discovered in the Middle Ages (credited to Albertus Magnus) and phosphorus in 1669 (isolated from urine by Hennig Brand). Helium, identified in 1868, is the only element not initially discovered on Earth itself.[ai] The most recently identified nonmetal is radon, detected at the end of the 19th century.[170]
Some nonmetals occur naturally as free elements, others required intricate extraction or isolation procedures. Such procedures included spectroscopy, fractional distillation, radiation detection, electrolysis, ore acidification, displacement reactions, combustion, and controlled heating processes.
The noble gases, renowned for their low reactivity, were first identified via spectroscopy, air fractionation, and radioactive decay studies. Helium was initially detected by its distinctive yellow line in the solar corona spectrum. Subsequently, it was observed escaping as bubbles when uranite UO2 was dissolved in acid. Neon, argon, krypton, and xenon were obtained through the fractional distillation of air. The discovery of radon occurred three years after Henri Becquerel's pioneering research on radiation in 1896.[209]
The isolation of the halogen nonmetals from their halides involved techniques including electrolysis, acid addition, or displacement. These efforts were not without peril, as some chemists died in their pursuit of isolating fluorine. [210][211]
The unclassified nonmetals have a diverse history. Hydrogen was discovered and first described in 1671 as the product of the reaction between iron filings and dilute acids. Carbon was found naturally in forms like charcoal, soot, graphite, and diamond. Nitrogen was discovered by examining air after carefully removing oxygen. Oxygen itself was obtained by heating mercurous oxide. Phosphorus was derived from the heating of ammonium sodium hydrogen phosphate (Na(NH4)HPO4), a compound found in urine.[212] Sulfur occurred naturally as a free element, simplifying its isolation. Selenium,[aj] was first identified as a residue in sulfuric acid.[214]
Most metalloids were first isolated by heating their oxides (boron, silicon, arsenic, tellurium) or a sulfide (germanium).[170] Antimony, first obtained by heating its sulfide, stibnite, was later discovered in native form.[215]
Origin and use of the term[edit]
Although a distinction had existed between metals and other mineral substances since ancient times, it was only towards the end of the 18th century that a basic classification of chemical elements as either metallic or nonmetallic substances began to emerge. It would take another nine decades before the term "nonmetal" was widely adopted.

Around 340 BCE, in Book III of his treatise Meteorology, the ancient Greek philosopher Aristotle categorized substances found within the Earth into metals and "fossiles".[ak] The latter category included various minerals such as realgar, ochre, ruddle, sulfur, cinnabar, and other substances that he referred to as "stones which cannot be melted".[216]
Until the Middle Ages the classification of minerals remained largely unchanged, albeit with varying terminology. In the fourteenth century, the English alchemist Richardus Anglicus expanded upon the classification of minerals in his work Correctorium Alchemiae. In this text, he proposed the existence of two primary types of minerals. The first category, which he referred to as "major minerals", included well-known metals such as gold, silver, copper, tin, lead, and iron. The second category, labeled "minor minerals", encompassed substances like salts, atramenta (iron sulfate), alums, vitriol, arsenic, orpiment, sulfur, and similar substances that were not metallic bodies.[217]
The term "nonmetallic" dates back to at least the 16th century. In his 1566 medical treatise, French physician Loys de L'Aunay distinguished substances from plant sources based on whether they originated from metallic or non-metallic soils.[218]
Later, the French chemist Nicolas Lémery discussed metallic and nonmetallic minerals in his work Universal Treatise on Simple Drugs, Arranged Alphabetically published in 1699. In his writings, he contemplated whether the substance "cadmia" belonged to either the first category, akin to cobaltum (cobaltite), or the second category, exemplified by what was then known as calamine—a mixed ore containing zinc carbonate and silicate.[219]
The pivotal moment in the systematic classification of chemical elements into metallic and nonmetallic substances came in 1789 with the work of Antoine Lavoisier, a French chemist. He published the first modern list of chemical elements in his revolutionary[221] Traité élémentaire de chimie. The elements were categorized into distinct groups, including gases, metallic substances, nonmetallic substances, and earths (heat-resistant oxides).[222] Lavoisier's work gained widespread recognition and was republished in twenty-three editions across six languages within its first seventeen years, significantly advancing the understanding of chemistry in Europe and America.[223]
The widespread adoption of the term "nonmetal" followed a complex process spanning nearly nine decades. In 1811, the Swedish chemist Berzelius introduced the term "metalloids"[224] to describe nonmetallic elements, noting their ability to form negatively charged ions with oxygen in aqueous solutions.[225][226] While Berzelius' terminology gained significant acceptance,[227] it later faced criticism from some who found it counterintuitive,[226] misapplied,[228] or even invalid.[229][230] In 1864, reports indicated that the term "metalloids" was still endorsed by leading authorities,[231] but there were reservations about its appropriateness. The idea of designating elements like arsenic as metalloids had been considered.[231] By as early as 1866, some authors began preferring the term "nonmetal" over "metalloid" to describe nonmetallic elements.[232] In 1875, Kemshead[233] observed that elements were categorized into two groups: non-metals (or metalloids) and metals. He noted that the term "non-metal", despite its compound nature, was more precise and had become universally accepted as the nomenclature of choice.
Suggested distinguishing criteria[edit]
From the early 1800s, a variety of physical, chemical, and atomic properties have been suggested for distinguishing metals from nonmetals, as listed in the accompanying table. Some of the earliest recorded properties from 1803 are the (high) density and (good) electrical conductivity of metals.
In 1809, the British chemist and inventor Humphry Davy made a groundbreaking discovery that reshaped the understanding of metals and nonmetals.[259] When he isolated sodium and potassium, their low densities (floating on water!) contrasted with their metallic appearance, challenging the stereotype of metals as dense substances.[260][ar] Nevertheless, their classification as metals was firmly established by their distinct chemical properties.[262]
One of the most commonly recognized properties used in this context is the temperature coefficient of resistivity, the effect of heating on electrical resistance and conductivity. As temperature rises, the conductivity of metals decreases while that of nonmetals increases.[252] However, plutonium, carbon, arsenic, and antimony defy the norm. When plutonium (a metal) is heated within a temperature range of −175 to +125 °C its conductivity increases.[263] Similarly, despite its common classification as a nonmetal, when carbon (as graphite) is heated it experiences a decrease in electrical conductivity.[264] Arsenic and antimony, which are occasionally classified as nonmetals, show behavior similar to carbon, highlighting the complexity of the distinction between metals and nonmetals.[265]
Kneen and colleagues[266] proposed that the classification of nonmetals can be achieved by establishing a single criterion for metallicity. They acknowledged that various plausible classifications exist and emphasized that while these classifications may differ to some extent, they would generally agree on the categorization of nonmetals.
Emsley[267] pointed out the complexity of this task, asserting that no single property alone can unequivocally assign elements to either the metal or nonmetal category. Furthermore, Jones[268] emphasized that classification systems typically rely on more than two attributes to define distinct types.
Johnson[269] distinguished between metals and nonmetals on the basis of their physical states, electrical conductivity, mechanical properties, and the acid-base nature of their oxides:
- gaseous elements are nonmetals (hydrogen, nitrogen, oxygen, fluorine, chlorine and the noble gases);
- liquids (mercury, bromine) are either metallic or nonmetallic: mercury, as a good conductor, is a metal; bromine, with its poor conductivity, is a nonmetal;
- solids are either ductile and malleable, hard and brittle, or soft and crumbly:
- a. ductile and malleable elements are metals;
- b. hard and brittle elements include boron, silicon and germanium, which are semiconductors and therefore not metals; and
- c. soft and crumbly elements include carbon, phosphorus, sulfur, arsenic, antimony,[as] tellurium and iodine, which have acidic oxides indicative of nonmetallic character.[at]
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Several authors[274] have noted that nonmetals generally have low densities and high electronegativity. The accompanying table, using a threshold of 7 g/cm3 for density and 1.9 for electronegativity (revised Pauling), shows that all nonmetals have low density and high electronegativity. In contrast, all metals have either high density or low electronegativity (or both). Goldwhite and Spielman[275] added that, "... lighter elements tend to be more electronegative than heavier ones." The average electronegativity for the elements in the table with densities less than 7 gm/cm3 (metals and nonmetals) is 1.97 compared to 1.66 for the metals having densities of more than 7 gm/cm3.
Some authors divide elements into metals, metalloids, and nonmetals, but Oderberg[276] disagrees, arguing that by the principles of categorization, anything not classified as a metal should be considered a nonmetal.
Development of types[edit]

In 1844, Alphonse Dupasquier, a French doctor, pharmacist, and chemist,[277] established a basic taxonomy of nonmetals to aid in their study. He wrote:[278]
- They will be divided into four groups or sections, as in the following:
- Organogens—oxygen, nitrogen, hydrogen, carbon
- Sulphuroids—sulfur, selenium, phosphorus
- Chloroides—fluorine, chlorine, bromine, iodine
- Boroids—boron, silicon.
Dupasquier's quartet parallels the modern nonmetal types. The organogens and sulphuroids are akin to the unclassified nonmetals. The chloroides were later called halogens.[279] The boroids eventually evolved into the metalloids, with this classification beginning from as early as 1864.[231] The then unknown noble gases were recognized as a distinct nonmetal group after being discovered in the late 1800s.[280]
His taxonomy was noted for its natural basis.[281][av] That said, it was a significant departure from other contemporary classifications, since it grouped together oxygen, nitrogen, hydrogen, and carbon.[283]
In 1828 and 1859, the French chemist Dumas classified nonmetals as (1) hydrogen; (2) fluorine to iodine; (3) oxygen to sulfur; (4) nitrogen to arsenic; and (5) carbon, boron and silicon,[284] thereby anticipating the vertical groupings of Mendeleev's 1871 periodic table. Dumas' five classes fall into modern groups 1, 17, 16, 15, and 14 to 13 respectively.
Classification of metalloids[edit]

Boron and silicon were recognized early on as nonmetals[aw] but arsenic, antimony, tellurium, and germanium have a more complicated history. While the suitability of arsenic being counted as a metalloid had been considered in 1864,[231] Mendeleev, in 1897, counted it and antimony as metals.[286] Although tellurium likely acquired an "ium" suffix due to its metallic appearance,[287] Mendeleev said it represented a transition between metals and nonmetals.[288] The semiconductor germanium was first regarded as a poorly conducting metal due to the presence of impurities. The understanding of it as a semiconductor, and subsequently as a metalloid, emerged in the 1930s with the development of semiconductor physics.[201]
Since the 1940s, these six elements have been increasingly, but not universally, recognized as metalloids.[289] In 1947, Linus Pauling included a reference to them in his classic[290] and influential[291] textbook General chemistry: An introduction to descriptive chemistry and modern chemical theory. He described boron, silicon, germanium, arsenic, antimony (and polonium) as "elements with intermediate properties."[292] He said they were in the center of his electronegativity scale, with values close to 2.[ax] The emergence of the semiconductor industry and solid-state electronics in the 1950s and 1960s highlighted the semiconducting properties of germanium and silicon (and boron and tellurium), reinforcing the idea that metalloids were "in-between" or "half-way" elements.[294] Writing in 1982, Goldsmith[289] observed that, "The newest approach is to emphasize aspects of their physical and/or chemical nature such as electronegativity, crystallinity, overall electronic nature and the role of certain metalloids as semiconductors."
Comparison of selected properties[edit]
The two tables in this section list some of the properties of five types of elements (noble gases, halogen nonmetals, unclassified nonmetals, metalloids and, for comparison, metals) based on their most stable forms in ambient conditions.
The aim is to show that most properties display a left-to-right progression in metallic-to-nonmetallic character or average values.[295][296] Some overlap occurs as outlier elements of each type exhibit less-distinct, hybrid-like, or atypical properties.[297][ay] These overlaps or transitional points, along with horizontal, diagonal, and vertical relationships between the elements, form part of the "great deal of information" summarized by the periodic table.[299]
The dashed lines around the columns for metalloids signify that the treatment of these elements as a distinct type can vary depending on the author, or classification scheme in use.
Physical properties by element type[edit]
Physical properties are listed in loose order of ease of their determination.
| Property | Element type | ||||
|---|---|---|---|---|---|
| Metals | Metalloids | Unc. nonmetals | Halogen nonmetals | Noble gases | |
| General physical appearance | lustrous[19] | lustrous[300] | colorless[305] | ||
| Form and density[306] | solid (Hg liquid) |
solid | solid or gas | solid or gas (bromine liquid) |
gas |
| often high density such as iron, lead, tungsten | low to moderately high density | low density | low density | low density | |
| some light metals including beryllium, magnesium, aluminium | all lighter than iron | hydrogen, nitrogen lighter than air[307] | helium, neon lighter than air[308] | ||
| Elasticity | mostly malleable and ductile[19] | brittle[300] | carbon, phosphorus, sulfur, selenium, brittle[az] | iodine brittle[310] | not applicable |
| Electrical conductivity | good[ba] |
|
|
|
poor[be] |
| Electronic structure[41] | metallic (beryllium, strontium, α-tin, ytterbium, bismuth are semimetals) | semimetal (arsenic, antimony) or semiconductor |
|
semiconductor (I) or insulator | insulator |
Chemical properties by element type[edit]
Chemical properties are listed from general characteristics to more specific details.
| Property | Element type | ||||
|---|---|---|---|---|---|
| Metals | Metalloids | Unc. nonmetals | Halogen nonmetals | Noble gases | |
| General chemical behavior |
|
weakly nonmetallic[bf] | moderately nonmetallic[296] | strongly nonmetallic[315] | |
| Oxides | basic; some amphoteric or acidic[8] | amphoteric or weakly acidic[318][bg] | acidic[bh] or neutral[bi] | acidic[bj] | metastable XeO3 is acidic;[325] stable XeO4 strongly so[326] |
| few glass formers[bk] | all glass formers[328] | some glass formers[bl] | no glass formers reported | no glass formers reported | |
| ionic, polymeric, layer, chain, and molecular structures[330] | polymeric in structure[331] |
|
|||
| Compounds with metals | alloys[19] or intermetallic compounds[334] | tend to form alloys or intermetallic compounds[335] | mainly ionic[147] | simple compounds in ambient conditions not known[bm] | |
| Ionization energy (kJ mol−1)[64] ‡ | low to high | moderate | moderate to high | high | high to very high |
| 376 to 1,007 | 762 to 947 | 941 to 1,402 | 1,008 to 1,681 | 1,037 to 2,372 | |
| average 643 | average 833 | average 1,152 | average 1,270 | average 1,589 | |
| Electronegativity (Pauling)[bn][65] ‡ | low to high | moderate | moderate to high | high | high (radon) to very high |
| 0.7 to 2.54 | 1.9 to 2.18 | 2.19 to 3.44 | 2.66 to 3.98 | ca. 2.43 to 4.7 | |
| average 1.5 | average 2.05 | average 2.65 | average 3.19 | average 3.3 | |
† Hydrogen can also form alloy-like hydrides[165]
‡ The labels low, moderate, high, and very high are arbitrarily based on the value spans listed in the table
See also[edit]
- CHON (carbon, hydrogen, oxygen, nitrogen)
- List of nonmetal monographs
- Metallization pressure
- Nonmetal (astrophysics)
- Period 1 elements (hydrogen, helium)
- Properties of nonmetals (and metalloids) by group
Notes[edit]
- ^ These six (boron, silicon, germanium, arsenic, antimony, and tellurium) are the elements commonly recognized as "metalloids", a category sometimes considered to be a subcategory of nonmetals and sometimes considered to be a category separate from both metals and nonmetals.
- ^ The most stable forms are: diatomic hydrogen H2; β-rhombohedral boron; graphitic carbon; diatomic nitrogen N2; diatomic oxygen O2; tetrahedral silicon; black phosphorus; orthorhombic sulfur S8; α-germanium; gray arsenic; gray selenium; gray antimony; gray tellurium; and diatomic iodine I2. All other nonmetallic elements have only one stable form in ambient temperature and pressure.
- ^ At higher temperatures and pressures the numbers of nonmetals can be called into question. For example, when germanium melts it changes from a semiconducting metalloid to a metallic conductor with an electrical conductivity similar to that of liquid mercury.[12] At a high enough pressure, sodium (a metal) becomes a non-conducting insulator.[13]
- ^ The absorbed light may be converted to heat or re-emitted in all directions so that the emission spectrum is thousands of times weaker than the incident light radiation.[16]
- ^ Solid iodine has a silvery metallic appearance under white light at room temperature. At ordinary and higher temperatures it sublimes from the solid phase directly into a violet-colored vapor.[17]
- ^ The solid nonmetals have electrical conductivity values ranging from 10−18 S•cm−1 for sulfur[21] to 3 × 104 in graphite[22] or 3.9 × 104 for arsenic;[23] cf. 0.69 × 104 for manganese to 63 × 104 for silver, both metals.[21] The conductivity of graphite (a nonmetal) and arsenic (a metalloid nonmetal) exceeds that of manganese. Such overlaps show that it can be difficult to draw a clear line between metals and nonmetals.
- ^ Thermal conductivity values for metals range from 6.3 W m−1 K−1 for neptunium to 429 for silver; cf. antimony 24.3, arsenic 50, and carbon 2000.[21] Electrical conductivity values of metals range from 0.69 S•cm−1 × 104 for manganese to 63 × 104 for silver; cf. carbon 3 × 104,[22] arsenic 3.9 × 104 and antimony 2.3 × 104.[21]
- ^ While CO and NO are commonly referred to as being neutral, CO is a slightly acidic oxide, reacting with bases to produce formates (CO + OH− → HCOO−);[68] and in water, NO reacts with oxygen to form nitrous acid HNO2 (4NO + O2 + 2H2O → 4HNO2).[69]
- ^ Electronegativity values of fluorine to iodine are: 3.98 + 3.16 + 2.96 + 2.66 = 12.76/4 3.19.
- ^ Electronegativity values of boron to tellurium are: 2.04 + 1.9 + 2.01 + 2.18 + 2.05 + 2.1 = 12.28/6 = 2.04.
- ^ Helium is shown above beryllium for electron configuration consistency purposes; as a noble gas it is usually placed above neon, in group 18.
- ^ The net result is an even-odd difference between periods (except in the s-block): elements in even periods have smaller atomic radii and prefer to lose fewer electrons, while elements in odd periods (except the first) differ in the opposite direction. Many properties in the p-block then show a zigzag rather than a smooth trend along the group. For example, phosphorus and antimony in odd periods of group 15 readily reach the +5 oxidation state, whereas nitrogen, arsenic, and bismuth in even periods prefer to stay at +3.[90]
- ^ Oxidation states, which denote hypothetical charges for conceptualizing electron distribution in chemical bonding, do not necessarily reflect the net charge of molecules or ions. This concept is illustrated by anions such as NO3−, where the nitrogen atom is considered to have an oxidation state of +5 due to the distribution of electrons. However, the net charge of the ion remains −1. Such observations underscore the role of oxidation states in describing electron loss or gain within bonding contexts, distinct from indicating the actual electrical charge, particularly in covalently bonded molecules.
- ^ Greenwood[97] commented that: "The extent to which metallic elements mimic boron (in having fewer electrons than orbitals available for bonding) has been a fruitful cohering concept in the development of metalloborane chemistry ... Indeed, metals have been referred to as "honorary boron atoms" or even as "flexiboron atoms". The converse of this relationship is clearly also valid."
- ^ For example, the conductivity of graphite is 3 × 104 S•cm−1.[98] whereas that of manganese is 6.9 × 103 S•cm−1.[99]
- ^ A homopolyatomic cation consists of two or more atoms of the same element bonded together and carrying a positive charge, for example, N5+, O2+ and Cl4+. This is unusual behavior for nonmetals since cation formation is normally associated with metals, and nonmetals are normally associated with anion formation. Homopolyatomic cations are further known for carbon, phosphorus, antimony, sulfur, selenium, tellurium, bromine, iodine and xenon.[101]
- ^ Of the twelve categories in the Royal Society periodic table, five only show up with the metal filter, three only with the nonmetal filter, and four with both filters. Interestingly, the six elements marked as metalloids (boron, silicon, germanium, arsenic, antimony, and tellurium) show under both filters. Six other elements (113–120: nihonium, flerovium, moscovium, livermorium, tennessine, and oganneson), whose status is unknown, also show up under both filters but are not included in any of the twelve color categories.
- ^ The quote marks are not found in the source; they are used here to make it clear that the source employs the word non-metals as a formal term for the subset of chemical elements in question, rather than applying to nonmetals generally.
- ^ Varying configurations of these nonmetals have been referred to as, for example, basic nonmetals,[110] bioelements,[111] central nonmetals,[112] CHNOPS,[113] essential elements,[114] "non-metals",[115][r] orphan nonmetals,[116] or redox nonmetals.[117]
- ^ Arsenic is stable in dry air. Extended exposure in moist air results in the formation of a black surface coating. "Arsenic is not readily attacked by water, alkaline solutions or non-oxidizing acids".[122] It can occasionally be found in nature in an uncombined form.[123] It has a positive standard reduction potential (As → As3+ + 3e = +0.30 V), corresponding to a classification of semi-noble metal.[124]
- ^ "Crystalline boron is relatively inert."[118] Silicon "is generally highly unreactive."[119] "Germanium is a relatively inert semimetal."[120] "Pure arsenic is also relatively inert."[121][t] "Metallic antimony is … inert at room temperature."[125] "Compared to S and Se, Te has relatively low chemical reactivity."[126]
- ^ Boundary fuzziness and overlaps often occur in classification schemes.[129]
- ^ Jones takes a philosophical or pragmatic view to these questions. He writes: "Though classification is an essential feature of all branches of science, there are always hard cases at the boundaries. The boundary of a class is rarely sharp ... Scientists should not lose sleep over the hard cases. As long as a classification system is beneficial to economy of description, to structuring knowledge and to our understanding, and hard cases constitute a small minority, then keep it. If the system becomes less than useful, then scrap it and replace it with a system based on different shared characteristics."[129]
- ^ For a related comparison of the properties of metals, metalloids, and nonmetals, see Rudakiya & Patel (2021), p. 36.
- ^ Thus, Weller at al.[132] write, "Those [elements] classified as metallic range from the highly reactive sodium and barium to the noble metals, such as gold and platinum. The nonmetals... encompass... the aggressive, highly-oxidizing fluorine and the unreactive gases such as helium." On a related note, Beiser[133] adds, "Across each period is a more or less steady transition from an active metal through less active metals and weakly active non-metals to highly active nonmetals and finally to an inert gas."
- ^ In a full-width periodic table the f-block is located between the s- and d-blocks.
- ^ For a p-block metal, aluminium can be quite reactive if its thin and transparent protective surface coating of Al2O3 is removed.[134] Aluminium is adjacent to the highly reactive s-block metal magnesium, as period 3 lacks f- or d-block elements. Magnesium too has "a very adherent thin film of oxide which protects the underlying metal from attack."[135] Thallium, a p-block metal, is unaffected by water or alkalis but is attacked by acids, and is slowly oxidized in room temperature air.[136]
- ^ Metal oxides are usually ionic.[148] On the other hand, oxides of metals with high oxidation states are usually either polymeric or covalent.[149] A polymeric oxide has a linked structure composed of multiple repeating units.[150]
- ^ Sulfur, an insulator, and selenium, a semiconductor, are each photoconductors—their electrical conductivities increase by up to six orders of magnitude when exposed to light.[156]
- ^ For example, Wulfsberg divides the nonmetals, based on their Pauling electronegativity, into very electronegative nonmetals (over 2.8: nitrogen, oxygen, fluorine, chlorine, and bromine) and electronegative nonmetals (1.9–2.8: hydrogen, boron, carbon, silicon, phosphorus, sulfur, germanium, arsenic, selenium, antimony, tellurium, iodine, and xenon). He susbequently compares the two types on the basis of their standard reduction potentials. The remaining noble gases (He, Ne, Ar, Kr and Rn) are not allocated as they lack standard reduction potentials and, on this basis, cannot be compared to the other very electronegative and electronegative nonmetals. However, on the basis of their listed electronegativity values (p. 37), helium, neon, argon and krypton would very electronegative nonmetals and radon would be an electronegative nonmetal. The nonmetals boron, silicon, germanium, arsenic, selenium, antimony, and tellurium are additionally recognized by him as metalloids.[161]
- ^ Ordinary baryonic matter – including the stars, planets, and all living creatures – constitutes less than 5% of the universe. The rest – dark energy and dark matter – is as yet poorly understood.[183]
- ^ In the Earth's core there may be around 1013 tons of xenon, in the form of stable XeFe3 and XeNi3 intermetallic compounds. This could explain why "studies of the Earth's atmosphere have shown that more than 90% of the expected amount of Xe is depleted."[187]
- ^ Exceptionally, a study reported in 2012 noted the presence of 0.04% native fluorine (F
2) by weight in antozonite, attributing these inclusions to radiation from tiny amounts of uranium.[188] - ^ or their compounds
- ^ How helium acquired the -ium suffix is explained in the following passage by its discoverer, William Lockyer: "I took upon myself the responsibility of coining the word helium ... I did not know whether the substance ... was a metal like calcium or a gas like hydrogen, but I did know that it behaved like hydrogen [being found in the sun] and that hydrogen, as Dumas had stated, behaved as a metal".[208]
- ^ Berzelius, who discovered selenium, thought it had the properties of a metal, combined with the properties of sulfur.[213]
- ^ The term "fossile" is not to be confused with the modern usage of fossil to refer to the preserved remains, impression, or trace of any once-living thing.
- ^ "... [metals'] specific gravity is greater than that of any other bodies yet discovered; they are better conductors of electricity, than any other body."
- ^ The Goldhammer-Herzfeld ratio is roughly equal to the cube of the atomic radius divided by the molar volume.[238] More specifically, it is the ratio of the force holding an individual atom's outer electrons in place with the forces on the same electrons from interactions between the atoms in the solid or liquid element. When the interatomic forces are greater than, or equal to, the atomic force, outer electron itinerancy is indicated and metallic behavior is predicted. Otherwise nonmetallic behavior is anticipated.
- ^ Sonorousness is making a ringing sound when struck.
- ^ Liquid range is the difference between melting point and boiling point.
- ^ Configuration energy is the average energy of the valence electrons in a free atom.
- ^ Atomic conductance is the electrical conductivity of one mole of a substance. It is equal to electrical conductivity divided by molar volume.
- ^ It was subsequently proposed, by Erman and Simon,[261] to refer to sodium and potassium as metalloids, meaning "resembling metals in form or appearance". Their suggestion was ignored; the two new elements were admitted to the metal club in cognizance of their physical properties (opacity, luster, malleability, conductivity) and "their qualities of chemical combination".
Hare and Bache[259] observed that the line of demarcation between metals and nonmetals had been "annihilated" by the discovery of alkaline metals having a density less than that of water:
- "Peculiar brilliance and opacity were in the next place appealed to as a means of discrimination; and likewise that superiority in the power of conducting heat and electricity ... Yet so difficult has it been to draw the line between metallic…and non-metallic ... that bodies which are by some authors placed in one class, are by others included in the other. Thus selenium, silicon, and zirconion [sic] have by some chemists been comprised among the metals, by others among non-metallic bodies."
- ^ While antimony trioxide is usually listed as being amphoteric its very weak acid properties dominate over those of a very weak base.[270]
- ^ Johnson counted boron as a nonmetal and silicon, germanium, arsenic, antimony, tellurium, polonium and astatine as "semimetals" i.e. metalloids.
- ^ (a) The table includes elements up to einsteinium (99) except for astatine (85) and francium (87), with densities and most electronegativities from Aylward and Findlay;[271] Electronegativities of noble gases are from Rahm, Zeng and Hoffmann.[272]
(b) A survey of definitions of the term "heavy metal" reported density criteria ranging from above 3.5 g/cm3 to above 7 g/cm3;[273]
(c) Vernon specified a minimum electronegativity of 1.9 for the metalloids, on the revised Pauling scale;[3] - ^ A natural classification was based on "all the characters of the substances to be classified as opposed to the 'artificial classifications' based on one single character" such as the affinity of metals for oxygen. "A natural classification in chemistry would consider the most numerous and most essential analogies."[282]
- ^ Both boron and silicon were initially isolated in their impure or amorphous forms; the pure crystalline, metallic-looking forms were isolated later.[285]
- ^ Pauling's electronegativity scale ran from 0.7 to 4, giving a 2.35 midpoint. The electronegativity values of his metalloids spanned 1.9 for silicon to 2.1 for tellurium. The unclassified nonmetals spanned 2.1 for hydrogen to 3.5 for oxygen.[293]
- ^ A similar phenomenon applies more generally to certain groups of the periodic table where, for example, the noble gases in group 18 act as bridge between the nonmetals of the p-block and the metals of the s-block (groups 1 and 2).[298]
- ^ All four have less stable non-brittle forms: carbon as exfoliated (expanded) graphite,[49][309] and as carbon nanotube wire;[51] phosphorus as white phosphorus (soft as wax, pliable and can be cut with a knife, at room temperature);[52] sulfur as plastic sulfur;[53] and selenium as selenium wires.[54]
- ^ Metals have electrical conductivity values of from 6.9×103 S•cm−1 for manganese to 6.3×105 for silver.[311]
- ^ Metalloids have electrical conductivity values of from 1.5×10−6 S•cm−1 for boron to 3.9×104 for arsenic.[312]
- ^ Unclassified nonmetals have electrical conductivity values of from ca. 1×10−18 S•cm−1 for the elemental gases to 3×104 in graphite.[98]
- ^ Halogen nonmetals have electrical conductivity values of from ca. 1×10−18 S•cm−1 for F and Cl to 1.7×10−8 S•cm−1 for iodine.[98][144]
- ^ Elemental gases have electrical conductivity values of ca. 1×10−18 S•cm−1.[98]
- ^ Metalloids always give "compounds less acidic in character than the corresponding compounds of the [typical] nonmetals."[300]
- ^ Arsenic trioxide reacts with sulfur trioxide, forming arsenic "sulfate" As2(SO4)3.[319] This substance is covalent in nature rather than ionic;[320] it is also given as As2O3·3SO3.[321]
- ^ NO
2, N
2O
5, SO
3, SeO
3 are strongly acidic.[322] - ^ H2O, CO, NO, N2O are neutral oxides; CO and N2O are "formally the anhydrides of formic and hyponitrous acid, respectively viz. CO + H2O → H2CO2 (HCOOH, formic acid); N2O + H2O → H2N2O2 (hyponitrous acid)."[323]
- ^ ClO
2, Cl
2O
7, I
2O
5 are strongly acidic.[324] - ^ Metals that form glasses are: vanadium; molybdenum, tungsten; alumnium, indium, thallium; tin, lead; and bismuth.[327]
- ^ Unclassified nonmetals that form glasses are phosphorus, sulfur, selenium;[327] CO2 forms a glass at 40 GPa.[329]
- ^ Disodium helide (Na2He) is a compound of helium and sodium that is stable at high pressures above 113 GPa. Argon forms an alloy with nickel, at 140 GPa and close to 1,500 K, however at this pressure argon is no longer a noble gas.[337]
- ^ Values for the noble gases are from Rahm, Zeng and Hoffmann.[272]
References[edit]
Citations[edit]
- ^ a b c Larrañaga, Lewis & Lewis 2016, p. 988
- ^ a b Steudel 2020, p. 43: Steudel's monograph is an updated translation of the fifth German edition of 2013, incorporating the literature up to Spring 2019.
- ^ a b c d e f g Vernon 2013
- ^ a b Goodrich 1844, p. 264; The Chemical News 1897, p. 189; Hampel & Hawley 1976, pp. 174, 191; Lewis 1993, p. 835; Hérold 2006, pp. 149–50
- ^ At: Restrepo et al. 2006, p. 411; Thornton & Burdette 2010, p. 86; Hermann, Hoffmann & Ashcroft 2013, pp. 11604‒1‒11604‒5; Cn: Mewes et al. 2019; Fl: Florez et al. 2022; Og: Smits et al. 2020
- ^ Pascoe 1982, p. 3
- ^ Malone & Dolter 2010, pp. 110–111
- ^ a b c Porterfield 1993, p. 336
- ^ Godovikov & Nenasheva 2020, p. 4; Sanderson 1957, p. 229; Morely & Muir 1892, p. 241
- ^ a b Vernon 2020, p. 220; Rochow 1966, p. 4
- ^ IUPAC Periodic Table of the Elements
- ^ Berger 1997, pp. 71–72
- ^ Gatti, Tokatly & Rubio 2010
- ^ Wibaut 1951, p. 33: "Many substances ...are colourless and therefore show no selective absorption in the visible part of the spectrum."
- ^ Elliot 1929, p. 629
- ^ Fox 2010, p. 31
- ^ Tidy 1887, pp. 107–108; Koenig 1962, p. 108
- ^ Wiberg 2001, p. 416; Wiberg is here referring to iodine.
- ^ a b c d e f Kneen, Rogers & Simpson 1972, pp. 261–264
- ^ Phillips 1973, p. 7
- ^ a b c d e Aylward & Findlay 2008, pp. 6–12
- ^ a b Jenkins & Kawamura 1976, p. 88
- ^ Carapella 1968, p. 30
- ^ Zumdahl & DeCoste 2010, pp. 455, 456, 469, A40; Earl & Wilford 2021, p. 3-24
- ^ Still 2016, p. 120
- ^ Wiberg 2001, pp. 780
- ^ Wiberg 2001, pp. 824, 785
- ^ Earl & Wilford 2021, p. 3-24
- ^ Siekierski & Burgess 2002, p. 86
- ^ Charlier, Gonze & Michenaud 1994
- ^ Taniguchi et al. 1984, p. 867: "... black phosphorus ... [is] characterized by the wide valence bands with rather delocalized nature."; Carmalt & Norman 1998, p. 7: "Phosphorus ... should therefore be expected to have some metalloid properties."; Du et al. 2010: Interlayer interactions in black phosphorus, which are attributed to van der Waals-Keesom forces, are thought to contribute to the smaller band gap of the bulk material (calculated 0.19 eV; observed 0.3 eV) as opposed to the larger band gap of a single layer (calculated ~0.75 eV).
- ^ Wiberg 2001, pp. 742
- ^ Evans 1966, pp. 124–25
- ^ Wiberg 2001, pp. 758
- ^ Stuke 1974, p. 178; Donohue 1982, pp. 386–87; Cotton et al. 1999, p. 501
- ^ Steudel 2020, p. 601: "... Considerable orbital overlap can be expected. Apparently, intermolecular multicenter bonds exist in crystalline iodine that extend throughout the layer and lead to the delocalization of electrons akin to that in metals. This explains certain physical properties of iodine: the dark color, the luster and a weak electric conductivity, which is 3400 times stronger within the layers then perpendicular to them. Crystalline iodine is thus a two-dimensional semiconductor."; Segal 1989, p. 481: "Iodine exhibits some metallic properties ..."
- ^ Taylor 1960, p. 207; Brannt 1919, p. 34
- ^ a b Green 2012, p. 14
- ^ Spencer, Bodner & Rickard 2012, p. 178
- ^ Redmer, Hensel & Holst 2010, preface
- ^ a b Keeler & Wothers 2013, p. 293
- ^ Cahn & Haasen 1996, p. 4; Boreskov 2003, p. 44
- ^ DeKock & Gray 1989, pp. 423, 426—427
- ^ Boreskov 2003, p. 45
- ^ Kneen, Rogers & Simpson 1972, pp. 85–86, 237
- ^ Salinas 2019, p. 379
- ^ Yang 2004, p. 9
- ^ Wiberg 2001, pp. 416, 574, 681, 824, 895, 930; Siekierski & Burgess 2002, p. 129
- ^ a b Chung 1987
- ^ Godfrin & Lauter 1995
- ^ a b Janas, Cabrero-Vilatela & Bulmer 2013
- ^ a b Faraday 1853, p. 42; Holderness & Berry 1979, p. 255
- ^ a b Partington 1944, p. 405
- ^ a b c Regnault 1853, p. 208
- ^ Edwards 2000, pp. 100, 102–103; Herzfeld 1927, pp. 701–705
- ^ Barton 2021, p. 200
- ^ Wiberg 2001, p. 796
- ^ Shang et al. 2021
- ^ Tang et al. 2021
- ^ Steudel 2020, passim; Carrasco et al. 2023; Shanabrook, Lannin & Hisatsune 1981, pp. 130–133
- ^ Weller et al. 2018, preface
- ^ a b Abbott 1966, p. 18
- ^ Ganguly 2012, p. 1-1
- ^ a b Aylward & Findlay 2008, p. 132
- ^ a b c d Aylward & Findlay 2008, p. 126
- ^ Eagleson 1994, 1169
- ^ Moody 1991, p. 365
- ^ House 2013, p. 427
- ^ Lewis & Deen 1994, p. 568
- ^ Smith 1990, pp. 177–189
- ^ Yoder, Suydam & Snavely 1975, p. 58
- ^ Young et al. 2018, p. 753
- ^ Brown et al. 2014, p. 227
- ^ Siekierski & Burgess 2002, pp. 21, 133, 177
- ^ Moore 2016; Burford, Passmore & Sanders 1989, p. 54
- ^ Brady & Senese 2009, p. 69
- ^ Chemical Abstracts Service 2021
- ^ Emsley 2011, pp. 81
- ^ Cockell 2019, p. 210
- ^ Scott 2014, p. 3
- ^ Emsley 2011, p. 184
- ^ Jensen 1986, p. 506
- ^ Lee 1996, p. 240
- ^ Greenwood & Earnshaw 2002, p. 43
- ^ Cressey 2010
- ^ Siekierski & Burgess 2002, pp. 24–25
- ^ Siekierski & Burgess 2002, p. 23
- ^ Petruševski & Cvetković 2018; Grochala 2018
- ^ Kneen, Rogers & Simpson 1972, pp. 226, 360; Siekierski & Burgess 2002, pp. 52, 101, 111, 124, 194
- ^ Scerri 2020, pp. 407–420
- ^ Greenwood & Earnshaw 2002, pp. 27, 1232, 1234
- ^ Shchukarev 1977, p. 229
- ^ a b Cox 2004, p. 146
- ^ Vij et al. 2001
- ^ Dorsey 2023, pp. 12–13
- ^ Humphrey 1908
- ^ Greenwood 2001, p. 2057
- ^ a b c d Bogoroditskii & Pasynkov 1967, p. 77; Jenkins & Kawamura 1976, p. 88
- ^ Desai, James & Ho 1984, p. 1160
- ^ Stein 1983, p. 165
- ^ Engesser & Krossing 2013, p. 947
- ^ Schweitzer & Pesterfield 2010, p. 305
- ^ Rieck 1967, p. 97: Tungsten trioxide dissolves in hydrofluoric acid to give an oxyfluoride complex.
- ^ Wiberg 2001, p. 1279
- ^ Power 2010; Crow 2013; Weetman & Inoue 2018
- ^ Encyclopaedia Britannica 2021
- ^ Royal Society of Chemistry 2021
- ^ a b Matson & Orbaek 2013, p. 203
- ^ Kernion & Mascetta 2019, p. 191; Cao et al. 2021, pp. 20–21; Hussain et al. 2023; also called "nonmetal halogens": Chambers & Holliday 1982, pp. 273–274; Bohlmann 1992, p. 213; Jentzsch & Matile 2015, p. 247 or "stable halogens": Vassilakis, Kalemos & Mavridis 2014, p. 1; Hanley & Koga 2018, p. 24; Kaiho 2017, ch. 2, p. 1
- ^ Williams 2007, pp. 1550–1561: H, C, N, P, O, S
- ^ Wächtershäuser 2014, p. 5: H, C, N, P, O, S, Se
- ^ Hengeveld & Fedonkin, pp. 181–226: C, N, P, O, S
- ^ Wakeman 1899, p. 562
- ^ Fraps 1913, p. 11: H, C, Si, N, P, O, S, Cl
- ^ Parameswaran at al. 2020, p. 210: H, C, N, P, O, S, Se
- ^ Knight 2002, p. 148: H, C, N, P, O, S, Se
- ^ Fraústo da Silva & Williams 2001, p. 500: H, C, N, O, S, Se
- ^ Zhu et al. 2022
- ^ Graves 2022
- ^ Rosenberg 2013, p. 847
- ^ Obodovskiy 2015, p. 151
- ^ Greenwood & Earnshaw 2002, p. 552
- ^ Eagleson 1994, p. 91
- ^ Huang 2018, pp. 30, 32
- ^ Orisakwe 2012, p. 000
- ^ Yin et al. 2018, p. 2
- ^ a b Moeller et al. 1989, p. 742
- ^ Whiteford & Coffin 1939, p. 239
- ^ a b Jones 2010, pp. 169–71
- ^ Russell & Lee 2005, p. 419
- ^ Tyler 1948, p. 105; Reilly 2002, pp. 5–6
- ^ Weller et al. 2018, preface
- ^ Beiser 1987, p. 249
- ^ Whitten & Davis 1996, p. 853
- ^ Parish 1977, p. 37
- ^ Parish 1977, p. 183; Russell & Lee 2005, p. 419
- ^ Parish 1977, pp. 37, 52–53, 112, 115, 145, 163, 182
- ^ Jolly 1966, p. 20
- ^ Clugston & Flemming 2000, pp. 100–101, 104–105, 302
- ^ Maosheng 2020, p. 962
- ^ Mazej 2020
- ^ Wiberg 2001, p. 402
- ^ Vernon 2013, p. 1706
- ^ a b Greenwood & Earnshaw 2002, p. 804
- ^ Rudolph 1973, p. 133: "Oxygen and the halogens in particular ... are therefore strong oxidizing agents."
- ^ Daniel & Rapp 1976, p. 55
- ^ a b Cotton et al. 1999, p. 554
- ^ Woodward et al. 1999, pp. 133–194
- ^ Phillips & Williams 1965, pp. 478–479
- ^ Moeller et al. 1989, p. 314
- ^ Lanford 1959, p. 176
- ^ Emsley 2011, p. 478
- ^ Greenwood & Earnshaw 2002, p. 277
- ^ Atkins et al. 2006, p. 320
- ^ Greenwood & Earnshaw 2002, p. 482; Berger 1997, p. 86
- ^ Moss 1952, pp. 180, 202
- ^ a b c d Cao et al. 2021, p. 20
- ^ Challoner 2014, p. 5; Government of Canada 2015; Gargaud et al. 2006, p. 447
- ^ Crichton 2012, p. 6; Scerri 2013; Los Alamos National Laboratory 2021
- ^ Vernon 2020, p. 218
- ^ Wulfsberg 2000, 37, 273–274, 620
- ^ Seese & Daub 1985, p. 65
- ^ MacKay, MacKay & Henderson 2002, pp. 209, 211
- ^ Cousins, Davidson & García-Vivó 2013, pp. 11809–11811
- ^ a b Cao et al. 2021, p. 4
- ^ Liptrot 1983, p. 161; Malone & Dolter 2008, p. 255
- ^ Wiberg 2001, pp. 255–257
- ^ Scott & Kanda 1962, p. 153
- ^ Taylor 1960, p. 316
- ^ a b c d Emsley 2011, passim
- ^ Crawford 1968, p. 540; Benner, Ricardo & Carrigan 2018, pp. 167–168: "The stability of the carbon-carbon bond ... has made it the first choice element to scaffold biomolecules. Hydrogen is needed for many reasons; at the very least, it terminates C-C chains. Heteroatoms (atoms that are neither carbon nor hydrogen) determine the reactivity of carbon-scaffolded biomolecules. In ... life, these are oxygen, nitrogen and, to a lesser extent, sulfur, phosphorus, selenium, and an occasional halogen."
- ^ Zhao, Tu & Chan 2021
- ^ Kosanke et al. 2012, p. 841
- ^ Wasewar 2021, pp. 322–323
- ^ Messler 2011, p. 10
- ^ King 1994, p. 1344; Powell & Tims 1974, pp. 189–191; Cao et al. 2021, pp. 20–21
- ^ Vernon 2020, pp. 221–223; Rayner-Canham 2020, p. 216
- ^ Ramdohr 1969, p. 371
- ^ Gillham 1956, p. 338
- ^ Chandra X-ray Center 2018
- ^ a b c d Nelson 1987, p. 732
- ^ a b Fortescue 2012, pp. 56, 65
- ^ Ostriker & Steinhardt 2001, pp. 46‒53; Zhu 2020, p. 27
- ^ Cox 1997, pp. 17–19
- ^ World Economic Forum 2021
- ^ Wang, Lineweaver & Ireland 2018, p. 462
- ^ Zhu et al. 2014, pp. 644–648
- ^ Schmedt, Mangstl & Kraus 2012, p. 7847‒7849
- ^ a b c Allcock 2020, pp. 61–63; Emsley 2011, passim; Harbison, Bourgeois & Johnson 2015, p. 364; USGS Mineral Commodity Summaries 2023
- ^ Burke 2020, p. 262; Csele 2016, pp. 7–11; Imberti & Sadler 2020, p. 8
- ^ Kiiski et al. 2016; King 2019, p. 408
- ^ Beard et al. 2021; Bhuwalka et al. 2021, pp. 10097–10107; Bolin 2017, p. 2-1; Reinhardt et al. 2015
- ^ Allcock 2020, pp. 61–63; Emsley 2011, passim; Gaffney & Marley 2017, p. 23; USGS Mineral Commodity Summaries 2023
- ^ Whitten et al. 2014, p. 133
- ^ Ward 2010, p. 250
- ^ Weeks & Leicester 1968, p. 550
- ^ Zhong & Nsengiyumva, p. 19
- ^ Angelo & Ravisankar p. 56–57
- ^ Greenwood & Earnshaw 2002, p. 482
- ^ Sultana et al. 2022
- ^ a b Haller 2006, p. 3
- ^ Shanks et al. 2017, pp. I2–I3
- ^ Emsley 2011, p. 611
- ^ Bajaj, Cascella & Borger 2022; Webb-Mack 2019
- ^ Rodgers 2012, p. 571
- ^ Gregersen 2008
- ^ Pawlicki, Scanderbeg & Starkschall 2016, p. 228
- ^ Labinger 2019, p. 305
- ^ Emsley 2011, pp. 42–43, 219–220, 263–264, 341, 441–442, 596, 609
- ^ Toon 2011
- ^ Emsley 2011, pp. 84, 128, 180–181, 247
- ^ Cook 1923, p. 124
- ^ Weeks ME & Leicester 1968, p. 309
- ^ Emsley 2011, pp. 113, 363, 378, 477, 514–515
- ^ Weeks & Leicester 1968, pp. 95, 97, 103
- ^ Jordan 2016
- ^ Stillman 1924, p. 213
- ^ de L'Aunay 1566, p. 7
- ^ Lémery 1699, p. 118; Dejonghe 1998, p. 329
- ^ Lavoisier 1790, p. 175
- ^ Strathern 2000, p. 239
- ^ Criswell 2007, p. 1140
- ^ Salzberg 1991, p. 204
- ^ Berzelius 1811, p. 258
- ^ Partington 1964, p. 168
- ^ a b Bache 1832, p. 250
- ^ Goldsmith 1982, p. 526
- ^ Roscoe & Schormlemmer 1894, p. 4
- ^ Glinka 1960, p. 76
- ^ Hérold 2006, pp. 149–150
- ^ a b c d The Chemical News and Journal of Physical Science 1864
- ^ Oxford English Dictionary 1989
- ^ Kemshead 1875, p. 13
- ^ Harris 1803, p. 274
- ^ Brande 1821, p. 5
- ^ Smith 1906, pp. 646–647
- ^ Beach 1911
- ^ Edwards & Sienko 1983, p. 693
- ^ Herzfeld 1927; Edwards 2000, pp. 100–103
- ^ Kubaschewski 1949, pp. 931–940
- ^ Remy 1956, p. 9
- ^ Stott 1956, pp. 100–102
- ^ Sanderson 1957, p. 229
- ^ White 1962, p. 106
- ^ Johnson 1966, pp. 3–4
- ^ Martin 1969, p. 6
- ^ Horvath 1973, pp. 335–336
- ^ Parish 1977, p. 178
- ^ Myers 1979, p. 712
- ^ Rao & Ganguly 1986
- ^ Smith & Dwyer 1991, p. 65
- ^ a b Herman 1999, p. 702
- ^ Scott 2001, p. 1781
- ^ Mann et al. 2000, p. 5136
- ^ Suresh & Koga 2001, pp. 5940–5944
- ^ a b Edwards 2010, pp. 941–965
- ^ Povh & Rosin 2017, p. 131
- ^ Hill, Holman & Hulme 2017, p. 182
- ^ a b Hare & Bache 1836, p. 310
- ^ Chambers 1743: "That which distinguishes metals from all other bodies ... is their heaviness ..."
- ^ Erman and Simon 1808
- ^ Edwards 2000, p. 85
- ^ Russell & Lee 2005, p. 466
- ^ Atkins et al. 2006, pp. 320–21
- ^ Zhigal'skii & Jones 2003, p. 66
- ^ Kneen, Rogers & Simpson 1972, pp. 218–219
- ^ Emsley 1971, p. 1
- ^ Jones 2010, p. 169
- ^ Johnson 1966, pp. 3–6, 15
- ^ Shkol'nikov 2010, p. 2127
- ^ Aylward & Findlay 2008, pp. 6–13; 126
- ^ a b Rahm, Zeng & Hoffmann 2019, p. 345
- ^ Duffus 2002, p. 798
- ^ Hein & Arena 2011, pp. 228, 523; Timberlake 1996, pp. 88, 142; Kneen, Rogers & Simpson 1972, p. 263; Baker 1962, pp. 21, 194; Moeller 1958, pp. 11, 178
- ^ Goldwhite & Spielman 1984, p. 130
- ^ Oderberg 2007, p. 97
- ^ Bertomeu-Sánchez et al. 2002, pp. 248–249
- ^ Dupasquier 1844, pp. 66–67
- ^ Bache 1832, pp. 248–276
- ^ Renouf 1901, pp. 268
- ^ Bertomeu-Sánchez et al. 2002, p. 248
- ^ Bertomeu-Sánchez et al. 2002, p. 236
- ^ Hoefer 1845, p. 85
- ^ Dumas 1828; Dumas 1859
- ^ Emsley 2011, pp. 80, 485
- ^ Mendeléeff 1897, pp. 180, 186–187
- ^ Emsley 2011, p. 530
- ^ Mendeléeff 1897, p. 274
- ^ a b Goldsmith 1982
- ^ Lundgren & Bensaude-Vincent 2000, p. 409
- ^ Greenberg 2007, p. 562
- ^ Pauling 1947, pp. 65, 160
- ^ Pauling 1947, p. 160
- ^ Chedd 1969
- ^ Vernon 2020, pp. 217–225
- ^ a b Welcher 2009, p. 3–32: "The elements change from ... metalloids, to moderately active nonmetals, to very active nonmetals, and to a noble gas."
- ^ Vernon 2020, pp. 224
- ^ MacKay, MacKay & Henderson 2002, pp. 195–196
- ^ Bynum, Browne & Porter 1981, p. 318
- ^ a b c Rochow 1966, p. 4
- ^ Wiberg 2001, p. 780; Emsley 2011, p. 397; Rochow 1966, pp. 23, 84
- ^ Kneen, Rogers & Simpson 1972, p. 439
- ^ Kneen, Rogers & Simpson 1972, pp. 321, 404, 436
- ^ Kneen, Rogers & Simpson 1972, p. 465
- ^ Kneen, Rogers & Simpson 1972, p. 308
- ^ Tregarthen 2003, p. 10
- ^ Lewis 1993, pp. 28, 827
- ^ Lewis 1993, pp. 28, 813
- ^ Godfrin & Lauter 1995, pp. 216‒218
- ^ Wiberg 2001, p. 416
- ^ Desai, James & Ho 1984, p. 1160; Matula 1979, p. 1260
- ^ Schaefer 1968, p. 76; Carapella 1968, pp. 29‒32
- ^ Kneen, Rogers & Simpson 1972, p. 264
- ^ Rayner-Canham 2018, p. 203
- ^ Mackin 2014, p. 80
- ^ Johnson 1966, pp. 105–108
- ^ Stein 1969, pp. 5396‒5397; Pitzer 1975, pp. 760‒761
- ^ Rochow 1966, p. 4; Atkins et al. 2006, pp. 8, 122–123
- ^ Wiberg 2001, p. 750.
- ^ Douglade & Mercier 1982, p. 723
- ^ Gillespie & Robinson 1959, p. 418
- ^ Sanderson 1967, p. 172; Mingos 2019, p. 27
- ^ House 2008, p. 441
- ^ Mingos 2019, p. 27; Sanderson 1967, p. 172
- ^ Wiberg 2001, p. 399
- ^ Kläning & Appelman 1988, p. 3760
- ^ a b Rao 2002, p. 22
- ^ Sidorov 1960, pp. 599–603
- ^ McMillan 2006, p. 823
- ^ Wells 1984, p. 534
- ^ a b Puddephatt & Monaghan 1989, p. 59
- ^ King 1995, p. 182
- ^ Ritter 2011, p. 10
- ^ Yamaguchi & Shirai 1996, p. 3
- ^ Vernon 2020, p. 223
- ^ Woodward et al. 1999, p. 134
- ^ Dalton 2019
Bibliography[edit]
- Abbott D 1966, An Introduction to the Periodic Table, J. M. Dent & Sons, London
- Allcock HR 2020, Introduction to Materials Chemistry, 2nd ed., John Wiley & Sons, Hoboken, ISBN 978-1-119-34119-2
- Angelo PC & Ravisankar B 2019, Introduction to Steels: Processing, Properties, and Applications, CRC Press, Boca Raton, ISBN 9781138389991
- Atkins PA et al. 2006, Shriver & Atkins' Inorganic Chemistry, 4th ed., Oxford University Press, Oxford, ISBN 978-0-7167-4878-6
- Aylward G and Findlay T 2008, SI Chemical Data, 6th ed., John Wiley & Sons Australia, Milton, ISBN 978-0-470-81638-7
- Bache AD 1832, "An essay on chemical nomenclature, prefixed to the treatise on chemistry; by J. J. Berzelius", American Journal of Science, vol. 22, pp. 248–277
- Bajaj T, Cascella M & Borger J 2022, "Xenon", in StatPearls, StatPearls Publishing, Treasure Island, Florida, PMID 31082041, accessed 4 October 2023
- Baker et al. PS 1962, Chemistry and You, Lyons and Carnahan, Chicago
- Barton AFM 2021, States of Matter, States of Mind, CRC Press, Boca Raton, ISBN 978-0-7503-0418-4
- Beach FC (ed.) 1911, The Americana: A universal reference library, vol. XIII, Mel–New, Metalloid, Scientific American Compiling Department, New York
- Beard A, Battenberg, C & Sutker BJ 2021, "Flame retardants", in Ullmann's Encyclopedia of Industrial Chemistry, doi:10.1002/14356007.a11_123.pub2
- Beiser A 1987, Concepts of modern physics, 4th ed., McGraw-Hill, New York, ISBN 978-0-07-004473-9
- Benner SA, Ricardo A & Carrigan MA 2018, "Is there a common chemical model for life in the universe?", in Cleland CE & Bedau MA (eds.), The Nature of Life: Classical and Contemporary Perspectives from Philosophy and Science, Cambridge University Press, Cambridge, ISBN 978-1-108-72206-3
- Berger LI 1997, Semiconductor Materials, CRC Press, Boca Raton, ISBN 978-0-8493-8912-2
- Bertomeu-Sánchez JR, Garcia-Belmar A & Bensaude-Vincent B 2002, "Looking for an order of things: Textbooks and chemical classifications in nineteenth century France", Ambix, vol. 49, no. 3, doi:10.1179/amb.2002.49.3.227
- Berzelius JJ 1811, 'Essai sur la nomenclature chimique', Journal de Physique, de Chimie, d'Histoire Naturelle, vol. LXXIII, pp. 253‒286
- Bhuwalka et al. 2021, "Characterizing the changes in material use due to vehicle electrification", Environmental Science & Technology, vol. 55, no. 14, pp. 10097–10107, doi:10.1021/acs.est.1c00970
- Bogoroditskii NP & Pasynkov VV 1967, Radio and Electronic Materials, Iliffe Books, London
- Bohlmann R 1992, "Synthesis of halides", in Winterfeldt E (ed.), Heteroatom manipulation, Pergamon Press, Oxford, ISBN 978-0-08-091249-3
- Bolin P 2017, "Gas-insulated substations", in McDonald JD (ed.), Electric Power Substations Engineering, 3rd, ed., CRC Press, Boca Raton, FL, ISBN 978-1-4398-5638-3
- Boreskov GK 2003, Heterogeneous Catalysis, Nova Science, New York, ISBN 978-1-59033-864-3
- Brady JE & Senese F 2009, Chemistry: The study of Matter and its Changes, 5th ed., John Wiley & Sons, New York, ISBN 978-0-470-57642-7
- Brande WT 1821, A Manual of Chemistry, vol. II, John Murray, London
- Brannt WT 1919, Metal Worker's Handy-book of Receipts and Processes, HC Baird & Company, Philadelphia
- Brown TL et al. 2014, Chemistry: The Central Science, 3rd ed., Pearson Australia: Sydney, ISBN 978-1-4425-5460-3
- Burford N, Passmore J & Sanders JCP 1989, "The preparation, structure, and energetics of homopolyatomic cations of groups 16 (the chalcogens) and 17 (the halogens)", in Liebman JF & Greenberg A (eds.), From atoms to polymers: isoelectronic analogies, VCH, New York, ISBN 978-0-89573-711-3
- Burke RA 2020, Hazmatology: The Science of Hazardous Materials, Vol. 3: Applied Chemistry and Physics, CRC Press, Boca Raton, ISBN 978-1-138-31652-2
- Bynum WF, Browne J & Porter R 1981 (eds), Dictionary of the History of Science, Princeton University Press, Princeton, ISBN 978-0-691-08287-5
- Cahn RW & Haasen P, Physical Metallurgy: Vol. 1, 4th ed., Elsevier Science, Amsterdam,ISBN 978-0-444-89875-3
- Cao C et al. 2021, "Understanding periodic and non-periodic chemistry in periodic tables", Frontiers in Chemistry, vol. 8, no. 813, doi:10.3389/fchem.2020.00813
- Carapella SC 1968, "Arsenic" in Hampel CA (ed.), The Encyclopedia of the Chemical Elements, Reinhold, New York
- Carmalt CJ & Norman NC 1998, "Arsenic, antimony and bismuth: Some general properties and aspects of periodicity", in Norman NC (ed.), Chemistry of Arsenic, Antimony and Bismuth, Blackie Academic & Professional, London, pp. 1–38, ISBN 0-7514-0389-X
- Carrasco et al. 2023, "Antimonene: a tuneable post-graphene material for advanced applications in optoelectronics, catalysis, energy and biomedicine", Chemical Society Reviews, vol. 52, no. 4, p. 1288–1330, doi:10.1039/d2cs00570k
- Challoner J 2014, The Elements: The New Guide to the Building Blocks of our Universe, Carlton Publishing Group, ISBN 978-0-233-00436-5
- Chambers E 1743, in "Metal", Cyclopedia: Or an Universal Dictionary of Arts and Sciences (etc.), vol. 2, D Midwinter, London
- Chambers C & Holliday AK 1982, Inorganic Chemistry, Butterworth & Co., London, ISBN 978-0-408-10822-5
- Chandra X-ray Observatory 2018, Abundance Pie Chart, accessed 26 October 2023
- Charlier J-C, Gonze X, Michenaud J-P 1994, "First-principles study of the stacking effect on the electronic properties of graphite(s)", Carbon, vol. 32, no. 2, pp. 289–99, doi:10.1016/0008-6223(94)90192-9
- Chedd G 1969, Half-way elements: The technology of metalloids, Double Day, Garden City, NY
- Chemical Abstracts Service 2021, CAS REGISTRY database as of November 2, Case #01271182
- Chung DD 1987, "Review of exfoliated graphite", Journal of Materials Science, vol. 22, doi:10.1007/BF01132008
- Clugston MJ & Flemming R 2000, Advanced Chemistry, Oxford University Press, Oxford, ISBN 978-0-19-914633-8
- Cockell C 2019, The Equations of Life: How Physics Shapes Evolution, Atlantic Books, London, ISBN 978-1-78649-304-0
- Cook CG 1923, Chemistry in Everyday Life: With Laboratory Manual, D Appleton, New York
- Cotton A et al. 1999, Advanced Inorganic Chemistry, 6th ed., Wiley, New York, ISBN 978-0-471-19957-1
- Cousins DM, Davidson MG & García-Vivó D 2013, "Unprecedented participation of a four-coordinate hydrogen atom in the cubane core of lithium and sodium phenolates", Chemical Communications, vol. 49, doi:10.1039/C3CC47393G
- Cox PA 1997, The Elements: Their Origins, Abundance, and Distribution, Oxford University Press, Oxford, ISBN 978-0-19-855298-7
- Cox T 2004, Inorganic Chemistry, 2nd ed., BIOS Scientific Publishers, London, ISBN 978-1-85996-289-3
- Crawford FH 1968, Introduction to the Science of Physics, Harcourt, Brace & World, New York
- Cressey D 2010, "Chemists re-define hydrogen bond", Nature newsblog, accessed August 23, 2017
- Crichton R 2012, Biological Inorganic Chemistry: A New Introduction to Molecular Structure and Function, 2nd ed., Elsevier, Amsterdam, ISBN 978-0-444-53783-6
- Criswell B 2007, "Mistake of having students be Mendeleev for just a day", Journal of Chemical Education, vol. 84, no. 7, pp. 1140–1144, doi:10.1021/ed084p1140
- Crow JM 2013, Main group renaissance, Chemistry World, 31 May, accessed 26 December 2023
- Csele M 2016, Lasers, in Ullmann's Encyclopedia of Industrial Chemistry, doi:10.1002/14356007.a15_165.pub2
- Dalton L 2019, "Argon reacts with nickel under pressure-cooker conditions", Chemical & Engineering News, accessed November 6, 2019
- Daniel PL & Rapp RA 1976, "Halogen corrosion of metals", in Fontana MG & Staehle RW (eds.), Advances in Corrosion Science and Technology, Springer, Boston, doi:10.1007/978-1-4615-9062-0_2
- de L'Aunay L 1566, Responce au discours de maistre Iacques Grevin, docteur de Paris, qu'il a escript contre le livre de maistre Loys de l'Aunay, medecin en la Rochelle, touchant la faculté de l'antimoine (Response to the Speech of Master Jacques Grévin,... Which He Wrote Against the Book of Master Loys de L'Aunay,... Touching the Faculty of Antimony), De l'Imprimerie de Barthelemi Berton, La Rochelle
- DeKock RL & Gray HB 1989, Chemical structure and bonding, University Science Books, Mill Valley, CA, ISBN 978-0-935702-61-3
- Dejonghe L 1998, "Zinc–lead deposits of Belgium", Ore Geology Reviews, vol. 12, no. 5, 329–354, doi:10.1016/s0169-1368(98)00007-9
- Desai PD, James HM & Ho CY 1984, "Electrical resistivity of aluminum and manganese", Journal of Physical and Chemical Reference Data, vol. 13, no. 4, doi:10.1063/1.555725
- Donohue J 1982, The Structures of the Elements, Robert E. Krieger, Malabar, Florida, ISBN 978-0-89874-230-5
- Dorsey MG 2023, Holding Their Breath: How the Allies Confronted the Threat of Chemical Warfare in World War II, Cornell University Press, Ithaca, New York, pp. 12-13, ISBN 978-1-5017-6837-8
- Douglade J, Mercier R 1982, Structure cristalline et covalence des liaisons dans le sulfate d’arsenic(III), As2(SO4)3, Acta Crystallographica Section B Structural Crystallography and Crystal Chemistry, vol. 38, no, 3, 720–723, doi:10.1107/s056774088200394x
- Du Y, Ouyang C, Shi S & Lei M 2010, Ab initio studies on atomic and electronic structures of black phosphorus, Journal of Applied Physics, vol. 107, no. 9, pp. 093718–1–4, doi:10.1063/1.3386509
- Duffus JH 2002, " 'Heavy metals'—A meaningless term?", Pure and Applied Chemistry, vol. 74, no. 5, pp. 793–807, doi:10.1351/pac200274050793
- Dumas JBA 1828, Traité de Chimie Appliquée aux Arts, Béchet Jeune, Paris
- Dumas JBA 1859, Mémoire sur les Équivalents des Corps Simples, Mallet-Bachelier, Paris
- Dupasquier A 1844, Traité élémentaire de chimie industrielle, Charles Savy Juene, Lyon
- Eagleson M 1994, Concise Encyclopedia Chemistry, Walter de Gruyter, Berlin, ISBN 3-11-011451-8
- Earl B & Wilford D 2021, Cambridge O Level Chemistry, Hodder Education, London, ISBN 978-1-3983-1059-9
- Edwards PP 2000, "What, why and when is a metal?", in Hall N (ed.), The New Chemistry, Cambridge University, Cambridge, pp. 85–114, ISBN 978-0-521-45224-3
- Edwards PP et al. 2010, "... a metal conducts and a non-metal doesn't", Philosophical Transactions of the Royal Society A, 2010, vol, 368, no. 1914, doi:10.1098/rsta.2009.0282
- Edwards PP & Sienko MJ 1983, "On the occurrence of metallic character in the periodic table of the elements", Journal of Chemical Education, vol. 60, no. 9, doi:10.1021/ed060p691, PMID 25666074
- Elliot A 1929, "The absorption band spectrum of chlorine", Proceedings of the Royal Society A, vol. 123, no. 792, pp. 629–644, doi:10.1098/rspa.1929.0088
- Emsley J 1971, The Inorganic Chemistry of the Non-metals, Methuen Educational, London, ISBN 978-0-423-86120-4
- Emsley J 2011, Nature's Building Blocks: An A–Z Guide to the Elements, Oxford University Press, Oxford, ISBN 978-0-19-850341-5
- Encyclopaedia Britannica, 2021, Periodic table, accessed September 21, 2021
- Engesser TA & Krossing I 2013, "Recent advances in the syntheses of homopolyatomic cations of the non metallic elements C, N, P, S, Cl, Br, I and Xe", Coordination Chemistry Reviews, vol. 257, nos. 5-6, pp. 946–955, doi:10.1016/j.ccr.2012.07.025
- Erman P & Simon P 1808, "Third report of Prof. Erman and State Architect Simon on their joint experiments", Annalen der Physik, vol. 28, no. 3, pp. 347–367
- Evans RC 1966, An Introduction to Crystal Chemistry, 2nd ed., Cambridge University, Cambridge
- Faraday M 1853, The Subject Matter of a Course of Six Lectures on the Non-metallic Elements, (arranged by John Scoffern), Longman, Brown, Green, and Longmans, London
- Florez et al. 2022, "From the gas phase to the solid state: The chemical bonding in the superheavy element flerovium", The Journal of Chemical Physics, vol. 157, 064304, doi:10.1063/5.0097642
- Fortescue JAC 2012, Environmental Geochemistry: A Holistic Approach, Springer-Verlag, New York, ISBN 978-1-4612-6047-9
- Fox M 2010, Optical Properties of Solids, 2nd ed., Oxford University Press, New York, ISBN 978-0-19-957336-3
- Fraps GS 1913, Principles of Agricultural Chemistry, The Chemical Publishing Company, Easton, PA
- Fraústo da Silva JJR & Williams RJP 2001, The Biological Chemistry of the Elements: The Inorganic Chemistry of Life, 2nd ed., Oxford University Press, Oxford, ISBN 978-0-19-850848-9
- Gaffney J & Marley N 2017, General Chemistry for Engineers, Elsevier, Amsterdam, ISBN 978-0-12-810444-6
- Ganguly A 2012, Fundamentals of Inorganic Chemistry, 2nd ed., Dorling-Kindersly (India), New Dehli ISBN 978-81-317-6649-1
- Gargaud M et al. (eds.) 2006, Lectures in Astrobiology, vol. 1, part 1: The Early Earth and Other Cosmic Habitats for Life, Springer, Berlin, ISBN 978-3-540-29005-6
- Gatti M, Tokatly IV & Rubio A, 2010, Sodium: a charge-transfer insulator at high pressures, Physical Review Letters, vol. 104, no. 21, doi:10.1103/PhysRevLett.104.216404
- Gillespie RJ, Robinson EA 1959, The sulfuric acid solvent system, in Emeléus HJ, Sharpe AG (eds), Advances in Inorganic Chemistry and Radiochemistry, vol. 1, pp. 386–424, Academic Press, New York
- Gillham EJ 1956, A semi-conducting antimony bolometer, Journal of Scientific Instruments, vol. 33, no. 9, doi:10.1088/0950-7671/33/9/303
- Glinka N 1960, General chemistry, Sobolev D (trans.), Foreign Languages Publishing House, Moscow
- Godfrin H & Lauter HJ 1995, "Experimental properties of 3He adsorbed on graphite", in Halperin WP (ed.), Progress in Low Temperature Physics, volume 14, Elsevier Science B.V., Amsterdam, ISBN 978-0-08-053993-5
- Godovikov AA & Nenasheva N 2020, Structural-chemical Systematics of Minerals, 3rd ed., Springer, Cham, Switzerland, ISBN 978-3-319-72877-3
- Goldsmith RH 1982, "Metalloids", Journal of Chemical Education, vol. 59, no. 6, pp. 526–527, doi:10.1021/ed059p526
- Goldwhite H & Spielman JR 1984, College Chemistry, Harcourt Brace Jovanovich, San Diego, ISBN 978-0-15-601561-5
- Goodrich BG 1844, A Glance at the Physical Sciences, Bradbury, Soden & Co., Boston
- Government of Canada 2015, Periodic table of the elements, accessed August 30, 2015
- Graves Jr JL 2022, A Voice in the Wilderness: A Pioneering Biologist Explains How Evolution Can Help Us Solve Our Biggest Problems, Basic Books, New York, ISBN 978-1-6686-1610-9,
- Green D 2012, The Elements, Scholastic, Southam, Warwickshire, ISBN 978-1-4071-3155-9
- Greenberg A 2007, From alchemy to chemistry in picture and story, John Wiley & Sons, Hoboken, NJ, 978-0-471-75154-0
- Greenwood NN 2001, Main group element chemistry at the millennium, Journal of the Chemical Society, Dalton Transactions, no. 14, pp. 2055–66, doi:10.1039/b103917m
- Greenwood NN & Earnshaw A 2002, Chemistry of the Elements, 2nd ed., Butterworth-Heinemann, ISBN 978-0-7506-3365-9
- Gregersen E 2008, "Radon", in Encyclopedia Britannica, accessed 5 October 2023
- Grochala W 2018, "On the position of helium and neon in the Periodic Table of Elements", Foundations of Chemistry, vol. 20, pp. 191–207, doi:10.1007/s10698-017-9302-7
- Haller EE 2006, "Germanium: From its discovery to SiGe devices", Materials Science in Semiconductor Processing, vol. 9, nos 4–5, accessed 9 October 2013
- Hampel CA & Hawley GG 1976, Glossary of Chemical Terms, Van Nostrand Reinhold, New York, ISBN 978-0-442-23238-2
- Hanley JJ & Koga KT 2018, "Halogens in terrestrial and cosmic geochemical systems: Abundances, geochemical behaviors, and analytical methods" in The Role of Halogens in Terrestrial and Extraterrestrial Geochemical Processes: Surface, Crust, and Mantle, Harlov DE & Aranovich L (eds.), Springer, Cham, ISBN 978-3-319-61667-4
- Harbison RD, Bourgeois MM & Johnson GT 2015, Hamilton and Hardy's Industrial Toxicology, 6th ed., John Wiley & Sons, Hoboken, ISBN 978-0-470-92973-5
- Hare RA & Bache F 1836, Compendium of the Course of Chemical Instruction in the Medical Department of the University of Pennsylvania, 3rd ed., JG Auner, Philadelphia
- Harris TM 1803, The Minor Encyclopedia, vol. III, West & Greenleaf, Boston
- Hein M & Arena S 2011, Foundations of College Chemistry, 13th ed., John Wiley & Sons, Hoboken, New Jersey, ISBN 978-0470-46061-0
- Hengeveld R & Fedonkin MA 2007, "Bootstrapping the energy flow in the beginning of life", Acta Biotheoretica, vol. 55, doi:10.1007/s10441-007-9019-4
- Herman ZS 1999, "The nature of the chemical bond in metals, alloys, and intermetallic compounds, according to Linus Pauling", in Maksić, ZB, Orville-Thomas WJ (eds.), 1999, Pauling's Legacy: Modern Modelling of the Chemical Bond, Elsevier, Amsterdam, doi:10.1016/S1380-7323(99)80030-2
- Hermann A, Hoffmann R & Ashcroft NW 2013, "Condensed astatine: Monatomic and metallic", Physical Review Letters, vol. 111, doi:10.1103/PhysRevLett.111.116404
- Hérold A 2006, "An arrangement of the chemical elements in several classes inside the periodic table according to their common properties", Comptes Rendus Chimie, vol. 9, no. 1, doi:10.1016/j.crci.2005.10.002
- Herzfeld K 1927, "On atomic properties which make an element a metal", Physical Review, vol. 29, no. 5, doi:10.1103/PhysRev.29.701
- Hill G, Holman J & Hulme PG 2017, Chemistry in Context, 7th ed., Oxford University Press, Oxford, ISBN 978-0-19-839618-5
- Hoefer F 1845, Nomenclature et Classifications Chimiques, J.-B. Baillière, Paris
- Holderness A & Berry M 1979, Advanced Level Inorganic Chemistry, 3rd ed., Heinemann Educational Books, London, ISBN 978-0-435-65435-1
- Horvath AL 1973, "Critical temperature of elements and the periodic system", Journal of Chemical Education, vol. 50, no. 5, doi:10.1021/ed050p335
- House JE 2008, Inorganic Chemistry, Elsevier, Amsterdam, ISBN 978-0-12-356786-4
- House JE 2013, Inorganic Chemistry, 2nd ed., Elsevier, Kidlington, ISBN 978-0-12-385110-9
- Huang Y 2018, Thermodynamics of materials corrosion, in Huang Y & Zhang J (eds), Materials Corrosion and Protection, De Gruyter, Boston, pp. 25-58, doi:10.1515/9783110310054-002
- Humphrey TPJ 1908, "Systematic course of study, Chemisty and physics", Pharmaceutical Journal, vol. 80, p. 58
- Hussain et al. 2023, "Tuning the electronic properties of molybdenum di-sulphide monolayers via doping using first-principles calculations", Physica Scripta, vol. 98, no. 2, doi:10.1088/1402-4896/acacd1
- Imberti C & Sadler PJ, 2020, "150 years of the periodic table: New medicines and diagnostic agents", in Sadler PJ & van Eldik R 2020, Advances in Inorganic Chemistry, vol. 75, Academic Press, ISBN 978-0-12-819196-5
- IUPAC Periodic Table of the Elements, accessed October 11, 2021
- Janas D, Cabrero-Vilatela, A & Bulmer J 2013, "Carbon nanotube wires for high-temperature performance", Carbon, vol. 64, pp. 305–314, doi:10.1016/j.carbon.2013.07.067
- Jenkins GM & Kawamura K 1976, Polymeric Carbons—Carbon Fibre, Glass and Char, Cambridge University Press, Cambridge, ISBN 978-0-521-20693-8
- Jentzsch AV & Matile S 2015, "Anion transport with halogen bonds", in Metrangolo P & Resnati G (eds.), Halogen Bonding I: Impact on Materials Chemistry and Life Sciences, Springer, Cham, ISBN 978-3-319-14057-5
- Jensen WB 1986, Classification, symmetry and the periodic table, Computers & Mathematics with Applications, vol. 12B, nos. 1/2, pp. 487−510, doi:10.1016/0898-1221(86)90167-7
- Johnson RC 1966, Introductory Descriptive Chemistry, WA Benjamin, New York
- Jolly WL 1966, The Chemistry of the Non-metals, Prentice-Hall, Englewood Cliffs, New Jersey
- Jones BW 2010, Pluto: Sentinel of the Outer Solar System, Cambridge University, Cambridge, ISBN 978-0-521-19436-5
- Jordan JM 2016 " 'Ancient episteme' and the nature of fossils: a correction of a modern scholarly error", History and Philosophy of the Life Sciences, vol. 38, no, 1, pp. 90–116, doi:10.1007/s40656-015-0094-6
- Kaiho T 2017, Iodine Made Simple, CRC Press, e-book, doi:10.1201/9781315158310
- Keeler J & Wothers P 2013, Chemical Structure and Reactivity: An Integrated Approach, Oxford University Press, Oxford, ISBN 978-0-19-960413-5
- Kemshead WB 1875, Inorganic chemistry, William Collins, Sons, & Company, London
- Kernion MC & Mascetta JA 2019, Chemistry: The Easy Way, 6th ed., Kaplan, New York, ISBN 978-1-4380-1210-0
- Kiiski et al. 2016, "Fertilizers, 1. General", in Ullmann's Encyclopedia of Industrial Chemistry, doi:10.1002/14356007.a10_323.pub4
- King AH 2019, "Our elemental footprint", Nature Materials, vol. 18, doi:10.1038/s41563-019-0334-3
- King RB 1994, Encyclopedia of Inorganic Chemistry, vol. 3, John Wiley & Sons, New York, ISBN 978-0-471-93620-6
- King RB 1995, Inorganic Chemistry of Main Group Elements, VCH, New York, ISBN 978-1-56081-679-9
- Kläning UK & Appelman EH 1988, "Protolytic properties of perxenic acid", Inorganic Chemistry, vol. 27, no. 21, doi:10.1021/ic00294a018
- Kneen WR, Rogers MJW & Simpson P 1972, Chemistry: Facts, Patterns, and Principles, Addison-Wesley, London, ISBN 978-0-201-03779-1
- Knight J 2002, Science of Everyday Things: Real-life chemistry, Gale Group, Detroit, ISBN 9780787656324
- Koenig SH 1962, in Proceedings of the International Conference on the Physics of Semiconductors, held at Exeter, July 16–20, 1962, The Institute of Physics and the Physical Society, London
- Kosanke et al. 2012, Encyclopedic Dictionary of Pyrotechnics (and Related Subjects), Part 3 – P to Z, Pyrotechnic Reference Series No. 5, Journal of Pyrotechnics, Whitewater, Colorado, ISBN 978-1-889526-21-8
- Kubaschewski O 1949, "The change of entropy, volume and binding state of the elements on melting", Transactions of the Faraday Society, vol. 45, doi:10.1039/TF9494500931
- Labinger JA 2019, "The history (and pre-history) of the discovery and chemistry of the noble gases", in Giunta CJ, Mainz VV & Girolami GS (eds.), 150 Years of the Periodic Table: A Commemorative Symposium, Springer Nature, Cham, Switzerland, ISBN 978-3-030-67910-1
- Lanford OE 1959, Using Chemistry, McGraw-Hill, New York
- Larrañaga MD, Lewis RJ & Lewis RA 2016, Hawley's Condensed Chemical Dictionary, 16th ed., Wiley, Hoboken, New York, ISBN 978-1-118-13515-0
- Lavoisier A 1790, Elements of Chemistry, R Kerr (trans.), William Creech, Edinburgh
- Lee JD 1996, Concise Inorganic Chemistry, 5th ed., Blackwell Science, Oxford, ISBN 978-0-632-05293-6
- Lémery N 1699, Traité universel des drogues simples, mises en ordre alphabetique, L d'Houry, Paris, p. 118
- Lewis RJ 1993, Hawley's Condensed Chemical Dictionary, 12th ed., Van Nostrand Reinhold, New York, ISBN 978-0-442-01131-4
- Lewis RS & Deen WM 1994, "Kinetics of the reaction of nitric oxide with oxygen in aqueous solutions", Chemical Research in Toxicology, vol. 7, no. 4, pp. 568–574, doi:10.1021/tx00040a013
- Liptrot GF 1983, Modern Inorganic Chemistry, 4th ed., Bell & Hyman, ISBN 978-0-7135-1357-8
- Los Alamos National Laboratory 2021, Periodic Table of Elements: A Resource for Elementary, Middle School, and High School Students, accessed September 19, 2021
- Lundgren A & Bensaude-Vincent B 2000, Communicating chemistry: textbooks and their audiences, 1789–1939, Science History, Canton, MA, ISBN 0-88135-274-8
- MacKay KM, MacKay RA & Henderson W 2002, Introduction to Modern Inorganic Chemistry, 6th ed., Nelson Thornes, Cheltenham, ISBN 978-0-7487-6420-4
- Mackin M 2014, Study Guide to Accompany Basics for Chemistry, Elsevier Science, Saint Louis, ISBN 978-0-323-14652-4
- Malone LJ & Dolter T 2008, Basic Concepts of Chemistry, 8th ed., John Wiley & Sons, Hoboken, ISBN 978-0-471-74154-1
- Mann et al. 2000, Configuration energies of the d-block elements, Journal of the American Chemical Society, vol. 122, no. 21, pp. 5132–5137, doi:10.1021/ja9928677
- Maosheng M 2020, "Noble gases in solid compounds show a rich display of chemistry with enough pressure", Frontiers in Chemistry, vol. 8, doi:10.3389/fchem.2020.570492
- Martin JW 1969, Elementary Science of Metals, Wykeham Publications, London
- Matson M & Orbaek AW 2013, Inorganic Chemistry for Dummies, John Wiley & Sons: Hoboken, ISBN 978-1-118-21794-8
- Matula RA 1979, "Electrical resistivity of copper, gold, palladium, and silver", Journal of Physical and Chemical Reference Data, vol. 8, no. 4, doi:10.1063/1.555614
- Mazej Z 2020, "Noble-gas chemistry more than half a century after the first report of the noble-gas compound", Molecules, vol. 25, no. 13, doi:10.3390/molecules25133014, PMID 32630333, PMC 7412050
- McMillan P 2006, "A glass of carbon dioxide", Nature, vol. 441, doi:10.1038/441823a
- Mendeléeff DI 1897, The Principles of Chemistry, vol. 2, 5th ed., trans. G Kamensky, AJ Greenaway (ed.), Longmans, Green & Co., London
- Messler Jr RW 2011, The Essence of Materials for Engineers, Jones and Bartlett Learning, Sudbury, Massachusetts, ISBN 978-0-7637-7833-0
- Mewes et al. 2019, "Copernicium: A relativistic noble liquid", Angewandte Chemie International Edition, vol. 58, pp. 17964–17968, doi:10.1002/anie.201906966
- Mingos DMP 2019, "The discovery of the elements in the Periodic Table", in Mingos DMP (ed.), The Periodic Table I. Structure and Bonding, Springer Nature, Cham, doi:10.1007/978-3-030-40025-5
- Moeller T 1958, Qualitative Analysis: An Introduction to Equilibrium and Solution Chemistry, McGraw-Hill, New York
- Moeller T et al. 1989, Chemistry: With Inorganic Qualitative Analysis, 3rd ed., Academic Press, New York, ISBN 978-0-12-503350-3
- Moody B 1991, Comparative Inorganic Chemistry, 3rd ed., Edward Arnold, London, ISBN 978-0-7131-3679-1
- Moore JT 2016, Chemistry for Dummies, 2nd ed., ch. 16, Tracking periodic trends, John Wiley & Sons: Hoboken, ISBN 978-1-119-29728-4
- Morely HF & Muir MM 1892, Watt's Dictionary of Chemistry, vol. 3, Longman's Green, and Co., London
- Moss, TS 1952, Photoconductivity in the Elements, Butterworths Scientific, London
- Myers RT 1979, "Physical and chemical properties and bonding of metallic elements", Journal of Chemical Education, vol. 56, no. 11, pp. 712–73, doi:10.1021/ed056p71
- Nelson PG 1987, "Important elements", Journal of Chemical Education, vol. 68, no. 9, doi:10.1021/ed068p732
- Obodovskiy I 2015, Fundamentals of Radiation and Chemical Safety, Elsevier, Amsterdam, ISBN 978-0-12-802026-5
- Oderberg DS 2007, Real Essentialism, Routledge, New York, ISBN 978-1-134-34885-5
- Ostriker JP & Steinhardt PJ 2001, "The quintessential universe", Scientific American, vol. 284, no. 1, pp. 46–53 PMID 11132422, doi:10.1038/scientificamerican0101-46
- Oxford English Dictionary 1989, "nonmetal"
- Orisakwe OE 2012, Other heavy metals: antimony, cadmium, chromium and mercury, in Pacheco-Torgal F, Jalali S & Fucic A (eds), Toxicity of Building Materials, Woodhead Publishing, Oxford, pp. 297–333, doi:10.1533/9780857096357.297
- Parameswaran P et al. 2020, "Phase evolution and characterization of mechanically alloyed hexanary Al16.6Mg16.6Ni16.6Cr16.6Ti16.6Mn16.6 high entropy alloy", Metal Powder Report, vol. 75, no. 4, doi:10.1016/j.mprp.2019.08.001
- Parish RV 1977, The Metallic Elements, Longman, London, ISBN 978-0-582-44278-8
- Partington JR 1944, A Text-book of Inorganic Chemistry, 5th ed., Macmillan & Co., London
- Partington JR 1964, A history of chemistry, vol. 4, Macmillan, London
- Pascoe KJ 1982, An Introduction to the Properties of Engineering Materials, 3rd ed., Von Nostrand Reinhold (UK), Wokingham, Berkshire, ISBN 978-0-442-30233-7
- Pauling L 1947, General chemistry: An introduction to descriptive chemistry and modern chemical theory, WH Freeman, San Francisco
- Pawlicki T, Scanderbeg DJ & Starkschall G 2016, Hendee's Radiation Therapy Physics, 4th ed., John Wiley & Sons, Hoboken, NJ, p. 228, ISBN 978-0-470-37651-5
- Petruševski VM & Cvetković J 2018, "On the 'true position' of hydrogen in the Periodic Table", Foundations of Chemistry, vol. 20, pp. 251–260, doi:10.1007/s10698-018-9306-y
- Phillips CSG & Williams RJP 1965, Inorganic Chemistry, vol. 1, Principles and non-metals, Clarendon Press, Oxford
- Phillips JC 1973, "The chemical structure of solids", in Hannay NB (ed.), Treatise on Solid State Chemistry, vol. 1, Plenum Press, New York, pp. 1–42, ISBN 978-1-4684-2663-2
- Pitzer K 1975, "Fluorides of radon and elements 118", Journal of the Chemical Society, Chemical Communications, no. 18, doi:10.1039/C3975000760B
- Porterfield WW 1993, Inorganic Chemistry, Academic Press, San Diego, ISBN 978-0-12-562980-5
- Povh B & Rosina M 2017, Scattering and Structures: Essentials and Analogies in Quantum Physics, 2nd ed., Springer, Berlin, doi:10.1007/978-3-662-54515-7
- Powell P & Timms P 1974, The Chemistry of the Non-Metals, Chapman and Hall, London, ISBN 978-0-412-12200-2
- Power PP 2010, Main-group elements as transition metals, Nature, vol. 463, 14 January 2010, pp. 171–177, doi:10.1038/nature08634
- Puddephatt RJ & Monaghan PK 1989, The Periodic Table of the Elements, 2nd ed., Clarendon Press, Oxford, ISBN 978-0-19-855516-2
- Rahm M, Zeng T & Hoffmann R 2019, "Electronegativity seen as the ground-state average valence electron binding energy", Journal of the American Chemical Society, vol. 141, no. 1, pp. 342–351, doi:10.1021/jacs.8b10246
- Ramdohr P 1969, The Ore Minerals and Their Intergrowths, Pergamon Press, Oxford
- Rao CNR & Ganguly PA 1986, "New criterion for the metallicity of elements", Solid State Communications, vol. 57, no. 1, pp. 5–6, doi:10.1016/0038-1098(86)90659-9
- Rao KY 2002, Structural chemistry of glasses, Elsevier, Oxford, ISBN 0-08-043958-6
- Rayner-Canham G 2018, "Organizing the transition metals", in Scerri E & Restrepo G (Ed's.) Mendeleev to Oganesson: A multidisciplinary perspective on the periodic table, Oxford University, New York, ISBN 978-0-190-668532
- Rayner-Canham G 2020, The Periodic Table: Past, Present and Future, World Scientific, New Jersey, ISBN 978-981-121-850-7
- Redmer R, Hensel F & Holst B (eds) 2010, "Metal-to-Nonmetal Transitions", Springer, Berlin, ISBN 978-3-642-03952-2
- Regnault MV 1853, Elements of Chemistry, vol. 1, 2nd ed., Clark & Hesser, Philadelphia
- Reilly C 2002, Metal Contamination of Food, Blackwell Science, Oxford, ISBN 978-0-632-05927-0
- Reinhardt et al. 2015, Inerting in the chemical industry, Linde, Pullach, Germany, accessed October 19 2021
- Remy H 1956, Treatise on Inorganic Chemistry, Anderson JS (trans.), Kleinberg J (ed.), vol. II, Elsevier, Amsterdam
- Renouf E 1901, "Lehrbuch der Anorganischen Chemie", Science, vol. 13, no. 320, doi:10.1126/science.13.320.268
- Restrepo G, Llanos EJ & Mesa H 2006, "Topological space of the chemical elements and its properties", Journal of Mathematical Chemistry, vol. 39, doi:10.1007/s10910-005-9041-1
- Rieck GD 1967, Tungsten and its Compounds, Pergamon Press, Oxford
- Ritter SK 2011, "The case of the missing xenon", Chemical & Engineering News, vol. 89, no. 9, ISSN 0009-2347
- Rochow EG 1966, The Metalloids, DC Heath and Company, Boston
- Rodgers GE 2012, Descriptive Inorganic, Coordination, and Solid State Chemistry, 3rd ed., Brooks/Cole, Belmont, California, ISBN 978-0-8400-6846-0
- Rosenberg E 2013, Germanium-containing compounds, current knowledge and applications, in Kretsinger RH, Uversky VN & Permyakov EA (eds), Encyclopedia of Metalloproteins, Springer, New York, doi:10.1007/978-1-4614-1533-6_582
- Roscoe HE & Schorlemmer FRS 1894, A Treatise on Chemistry: Volume II: The Metals, D Appleton, New York
- Royal Society of Chemistry 2021, Periodic Table: Non-metal, accessed September 3, 2021
- Rudakiya DM & Patel Y 2021, Bioremediation of metals, metalloids, and nonmetals, in Panpatte DG & Jhala YK (eds), Microbial Rejuvenation of Polluted Environment, vol. 2, Springer Nature, Singapore, pp. 33–49, doi:10.1007/978-981-15-7455-9_2
- Rudolph J 1973, Chemistry for the Modern Mind, Macmillan, New York
- Russell AM & Lee KL 2005, Structure-Property Relations in Nonferrous Metals, Wiley-Interscience, New York, ISBN 0-471-64952-X
- Salinas JT 2019 Exploring Physical Science in the Laboratory, Moreton Publishing, Englewood, Colorado, ISBN 978-1-61731-753-8
- Salzberg HW 1991, From Caveman to Chemist: Circumstances and Achievements, American Chemical Society, Washington, DC, ISBN 0-8412-1786-6
- Sanderson RT 1957, "An electronic distinction between metals and nonmetals", Journal of Chemical Education, vol. 34, no. 5, doi:10.1021/ed034p229
- Sanderson RT 1967, Inorganic Chemistry, Reinhold, New York
- Scerri E (ed.) 2013, 30-Second Elements: The 50 Most Significant Elements, Each Explained In Half a Minute, Ivy Press, London, ISBN 978-1-84831-616-4
- Scerri E 2020, The Periodic Table: Its Story and Its Significance, Oxford University Press, New York, ISBN 978-0-19091-436-3
- Schaefer JC 1968, "Boron" in Hampel CA (ed.), The Encyclopedia of the Chemical Elements, Reinhold, New York
- Schmedt auf der Günne J, Mangstl M & Kraus F 2012, "Occurrence of difluorine F2 in nature—In situ proof and quantification by NMR spectroscopy", Angewandte Chemie International Edition, vol. 51, no. 31, doi:10.1002/anie.201203515
- Schweitzer GK & Pesterfield LL 2010, Aqueous Chemistry of the Elements, Oxford University Press, Oxford, ISBN 978-0-19-539335-4
- Scott D 2014, Around the World in 18 Elements, Royal Society of Chemistry, e-book, ISBN 978-1-78262-509-4
- Scott EC & Kanda FA 1962, The Nature of Atoms and Molecules: A General Chemistry, Harper & Row, New York
- Scott WAH 2001, Chemistry Basic Facts, 5th ed., HarperCollins, Glasgow, ISBN 978-0-00-710321-8
- Seese WS & Daub GH 1985, Basic Chemistry, 4th ed., Prentice-Hall, Englewood Cliffs, NJ, ISBN 978-0-13-057811-2
- Segal BG 1989, Chemistry: Experiment and Theory, 2nd ed., John Wiley & Sons, New York, ISBN 0-471-84929-4
- Shanabrook BV, Lannin JS & Hisatsune IC 1981, "Inelastic light scattering in a onefold-coordinated amorphous semiconductor", Physical Review Letters, vol. 46, no. 2, 12 January, doi:10.1103/PhysRevLett.46.130
- Shang et al. 2021, "Ultrahard bulk amorphous carbon from collapsed fullerene", Nature, vol. 599, pp. 599–604, doi:10.1038/s41586-021-03882-9
- Shanks III WCP et al. 2017, "Germanium and indium", in Schulz et al. (eds), Critical Mineral Resources of the United States: Economic and Environmental Geology and Prospects for Future Supply, U.S. Geological Survey, Reston, Virginia, ISBN 978-1-4113-3991-0
- Shchukarev SA 1977, New views of D. I. Mendeleev's system. I. Periodicity of the stratigraphy of atomic electronic shells in the system, and the concept of Kainosymmetry", Zhurnal Obshchei Kimii, vol. 47, no. 2, pp. 246–259
- Shkol’nikov EV 2010, "Thermodynamic characterization of the amphoterism of oxides M2O3 (M = AS, Sb, Bi) and their hydrates in aqueous media, Russian Journal of Applied Chemistry, vol. 83, no. 12, pp. 2121–2127, doi:10.1134/S1070427210120104
- Sidorov TA 1960, "The connection between structural oxides and their tendency to glass formation", Glass and Ceramics, vol. 17, no. 11, doi:10.1007BF00670116
- Siekierski S & Burgess J 2002, Concise Chemistry of the Elements, Horwood Press, Chichester, ISBN 978-1-898563-71-6
- Smith A 1906, Introduction to Inorganic Chemistry, The Century Co., New York
- Smith A & Dwyer C 1991, Key Chemistry: Investigating Chemistry in the Contemporary World: Book 1: Materials and Everyday Life, Melbourne University Press, Carlton, Victoria, ISBN 978-0-522-84450-4
- Smith DW 1990, Inorganic Substances: A Prelude to the Study of Descriptive Chemistry, Cambridge University Press, Cambridge, ISBN 978-0-521-33136-4
- Smits et al. 2020, "Oganesson: A noble gas element that is neither noble nor a gas", Angewandte Chemie International Edition, vol. 59, pp. 23636–23640, doi:10.1002/anie.202011976
- Spencer JN, Bodner GM, Rickard LY 2012, Chemistry: Structure & Dynamics, 5th ed., John Wiley & Sons, Hoboken, ISBN 978-0-470-58711-9
- Stein L 1969, "Oxidized radon in halogen fluoride solutions", Journal of the American Chemical Society, vol. 19, no. 19, doi:10.1021/ja01047a042
- Stein L 1983, "The chemistry of radon", Radiochimica Acta, vol. 32, doi:10.1524/ract.1983.32.13.163
- Steudel R 2020, Chemistry of the Non-metals: Syntheses – Structures – Bonding – Applications, in collaboration with D Scheschkewitz, Berlin, Walter de Gruyter, doi:10.1515/9783110578065
- Still B 2016 The Secret Life of the Periodic Table, Cassell, London, ISBN 978-1-84403-885-5
- Stillman JM 1924, The Story of Early Chemistry, D. Appleton, New York
- Stott RWA 1956, Companion to Physical and Inorganic Chemistry, Longmans, Green and Co, London
- Stuke J 1974, "Optical and electrical properties of selenium", in Zingaro RA & Cooper WC (eds.), Selenium, Van Nostrand Reinhold, New York, pp. 174
- Strathern P 2000, Mendeleyev's dream: The Quest for the Elements, Hamish Hamilton, London, ISBN 978-0-8412-1786-7
- Sultana et al. 2022, "Synthesis, modification, and application of black phosphorus, few-layer black phosphorus (FLBP), and phosphorene: a detailed review", Materials Advances, vol. 3, no. 14, pp. 5557–5574, doi:10.1039/D1MA01101D
- Suresh CH & Koga NA 2001, "A consistent approach toward atomic radii”, Journal of Physical Chemistry A, vol. 105, no. 24. doi:10.1021/jp010432b
- Tang et al. 2021, "Synthesis of paracrystalline diamond", Nature, vol. 599, pp. 605–610, doi:10.1038/s41586-021-04122-w
- Taniguchi M, Suga S, Seki M, Sakamoto H, Kanzaki H, Akahama Y, Endo S, Terada S & Narita S 1984, "Core-exciton induced resonant photoemission in the covalent semiconductor black phosphorus", Solid State Communications, vo1. 49, no. 9, pp. 867–7, doi:10.1016/0038-1098(84)90441-1
- Taylor MD 1960, First Principles of Chemistry, Van Nostrand, Princeton
- The Chemical News and Journal of Physical Science 1864, "Notices of books: Manual of the Metalloids", vol. 9, p. 22
- The Chemical News and Journal of Physical Science 1897, "Notices of books: A Manual of Chemistry, Theoretical and Practical", by WA Tilden", vol. 75, pp. 188–189
- Thornton BF & Burdette SC 2010, "Finding eka-iodine: Discovery priority in modern times", Bulletin for the History of Chemistry, vol. 35, no. 2, accessed September 14, 2021
- Tidy CM 1887, Handbook of Modern Chemistry, 2nd ed., Smith, Elder & Co., London
- Timberlake KC 1996, Chemistry: An Introduction to General, Organic, and Biological Chemistry, 6th ed., HarperCollinsCollege, ISBN 978-0-673-99054-9
- Toon R 2011, "The discovery of fluorine", Education in Chemistry, Royal Society of Chemistry, accessed 7 October 2023
- Tregarthen L 2003, Preliminary Chemistry, Macmillan Education: Melbourne, ISBN 978-0-7329-9011-4
- Tyler PM 1948, From the Ground Up: Facts and Figures of the Mineral Industries of the United States, McGraw-Hill, New York
- U.S. Geological Survey 2023, Mineral Commodity Summaries, U.S. Geological Survey, accessed 3 October 2023
- Vassilakis AA, Kalemos A & Mavridis A 2014, "Accurate first principles calculations on chlorine fluoride ClF and its ions ClF±", Theoretical Chemistry Accounts, vol. 133, no. 1436, doi:10.1007/s00214-013-1436-7
- Vernon R 2013, "Which elements are metalloids?", Journal of Chemical Education, vol. 90, no. 12, pp. 1703‒1707, doi:10.1021/ed3008457
- Vernon R 2020, "Organising the metals and nonmetals", Foundations of Chemistry, vol. 22, pp. 217‒233doi:10.1007/s10698-020-09356-6 (open access)
- Vij et al. 2001, Polynitrogen chemistry. Synthesis, characterization, and crystal structure of surprisingly stable fluoroantimonate salts of N5+. Journal of the American Chemical Society, vol. 123, no. 26, pp. 6308−6313, doi:10.1021/ja010141g
- Wächtershäuser G 2014, "From chemical invariance to genetic variability", in Weigand W and Schollhammer P (eds.), Bioinspired Catalysis: Metal Sulfur Complexes, Wiley-VCH, Weinheim, doi:10.1002/9783527664160.ch1
- Wakeman TH 1899, "Free thought—Past, present and future", Free Thought Magazine, vol. 17
- Wang HS, Lineweaver CH & Ireland TR 2018, The elemental abundances (with uncertainties) of the most Earth-like planet, Icarus, vol. 299, pp. 460–474, doi:10.1016/j.icarus.2017.08.024
- Ward D 2010, "What Is the diversity of life in the cosmos?" in Lynden-Bell et al. (eds), Water and Life: The Unique Properties of H2O, CRC Press, Boca Raton, ISBN 978-0-429-19103-9
- Wasewar KL 2021, "Intensifying approaches for removal of selenium", in Devi et al. (eds.), Selenium contamination in water, John Wiley & Sons, Hoboken, pp. 319–355, ISBN 978-1-119-69354-3
- Webb-Mack 2019, A Brief History of Ion Propulsion, NASA, accessed 5 October 2023
- Weeks ME & Leicester HM 1968, Discovery of the Elements, 7th ed., Journal of Chemical Education, Easton, Pennsylvania
- Weetman C & Inoue S 2018, The road travelled: After main-group elements as transition metals, ChemCatChem, vol. 10, no. 19, pp. 4213–4228, doi:10.1002/cctc.201800963
- Welcher SH 2009, High marks: Regents Chemistry Made Easy, 2nd ed., High Marks Made Easy, New York, ISBN 978-0-9714662-0-3
- Weller et al. 2018, Inorganic Chemistry, 7th ed., Oxford University Press, Oxford, ISBN 978-0-19-252295-5
- Wells AF 1984, Structural Inorganic Chemistry, 5th ed., Clarendon Press, Oxford, ISBN 978-0-19-855370-0
- White JH 1962, Inorganic Chemistry: Advanced and Scholarship Levels, University of London Press, London
- Whiteford GH & and Coffin RG 1939, Essentials of College Chemistry, 2nd ed., Mosby Co., St Louis
- Whitten KW & Davis RE 1996, General Chemistry, 5th ed., Saunders College Publishing, Philadelphia, ISBN 978-0-03-006188-2
- Whitten et al. 2014, Chemistry, 10th ed., Brooks Cole, Belmont CA, ISBN 978-1-133-61066-3
- Wibaut P 1951, Organic Chemistry, Elsevier Publishing Company, New York
- Wiberg N 2001, Inorganic Chemistry, Academic Press, San Diego, ISBN 978-0-12-352651-9
- Williams RPJ 2007, "Life, the environment and our ecosystem", Journal of Inorganic Biochemistry, vol. 101, nos. 11–12, doi:10.1016/j.jinorgbio.2007.07.006
- Woodward et al. 1999, "The electronic structure of metal oxides", In Fierro JLG (ed.), Metal Oxides: Chemistry and Applications, CRC Press, Boca Raton, ISBN 1-4200-2812-X
- World Economic Forum 2021, Visualizing the abundance of elements in the Earth's crust, accessed 21 March 2024
- Wulfsberg G 2000, Inorganic Chemistry, University Science Books, Sausalito, California, ISBN 978-1-891389-01-6
- Yamaguchi M & Shirai Y 1996, "Defect structures", in Stoloff NS & Sikka VK (eds.), Physical Metallurgy and Processing of Intermetallic Compounds, Chapman & Hall, New York, ISBN 978-1-4613-1215-4
- Yang J 2004, "Theory of thermal conductivity", in Tritt TM (ed.), Thermal Conductivity: Theory, Properties, and Applications, Kluwer Academic/Plenum Publishers, New York, pp. 1–20, ISBN 978-0-306-48327-1
- Yin et al. 2018, Hydrogen-assisted post-growth substitution of tellurium into molybdenum disulfide monolayers with tunable compositions, Nanotechnology, vol. 29, no 14, doi:10.1088/1361-6528/aaabe8
- Yoder CH, Suydam FH & Snavely FA 1975, Chemistry, 2nd ed, Harcourt Brace Jovanovich, New York, ISBN 978-0-15-506470-6
- Young JA 2006, "Iodine", Journal of Chemical Education, vol. 83, no. 9, doi:10.1021/ed083p1285
- Young et al. 2018, General Chemistry: Atoms First, Cengage Learning: Boston, ISBN 978-1-337-61229-6
- Zhao J, Tu Z & Chan SH 2021, "Carbon corrosion mechanism and mitigation strategies in a proton exchange membrane fuel cell (PEMFC): A review", Journal of Power Sources, vol. 488, #229434, doi:10.1016/j.jpowsour.2020.229434
- Zhigal'skii GP & Jones BK 2003, The Physical Properties of Thin Metal Films, Taylor & Francis, London, ISBN 978-0-415-28390-8
- Zhong S & Nsengiyumva W 2022, "Nondestructive Testing and Evaluation of Fiber-Reinforced Composite Structures", Science Press, Singapore, ISBN 978-981-19-0848-4
- Zhu W 2020, Chemical Elements In Life, World Scientific, Singapore, ISBN 978-981-121-032-7
- Zhu et al. 2014, "Reactions of xenon with iron and nickel are predicted in the Earth's inner core", Nature Chemistry, vol. 6, doi:10.1038/nchem.1925, PMID 24950336
- Zhu et al. 2022, Introduction: basic concept of boron and its physical and chemical properties, in Fundamentals and Applications of Boron Chemistry, vol. 2, Zhu Y (ed.), Elsevier, Amsterdam, ISBN 978-0-12-822127-3
- Zumdahl SS & DeCoste DJ 2010, Introductory Chemistry: A Foundation, 7th ed., Cengage Learning, Mason, Ohio, ISBN 978-1-111-29601-8
External links[edit]
 Media related to Nonmetals at Wikimedia Commons
Media related to Nonmetals at Wikimedia Commons
















