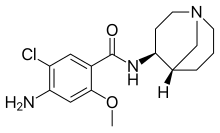
Renzapride
 | |
| Clinical data | |
|---|---|
| ATC code |
|
| Identifiers | |
| |
| CAS Number |
|
| PubChem CID | |
| IUPHAR/BPS | |
| ChemSpider | |
| UNII |
|
| ChEMBL | |
| Chemical and physical data | |
| Formula | C16H22ClN3O2 |
| Molar mass | 323.82 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Renzapride is a prokinetic agent and antiemetic which acts as a full 5-HT4 agonist and partial 5-HT3 antagonist.[1][2] It also functions as a 5-HT2B antagonist and has some affinity for the 5-HT2A and 5-HT2C receptors.[1]
Renzapride was being developed by Alizyme plc of the United Kingdom. In May 2016, EndoLogic LLC, a US-based pharmaceutical and medical device company, acquired the US and worldwide patent rights to Renzapride.
Endologic confirmed the cardiac safety of renzapride through a “Thorough QTc” study [3] and sold the rights to Atlantic Healthcare plc in 2019,[4] a specialist pharmaceutical company.
Atlantic Healthcare is focusing on the development of renzapride for the management of gastrointestinal (GI) motility in a number of rare diseases, including systemic scleroderma and cystic fibrosis, both of which are associated with chronic GI motility problems and for which there are no approved therapies.
Clinical trials[edit]
In nine diabetic patients with autonomic neuropathy, renzapride reduced the mean lag phase of gastric emptying by 20–26 min at all doses (P < 0.01) [5]
In Phase 2a studies on subjects with constipation Renzapride was shown to accelerate colonic transit (p=0.016 vs placebo P=0.009) (Ref: ATL 1251/001/CL) as well as increase daily stool frequency (p<0.005) (Ref: ATL 1251/025/CL)
Renzapride has been assessed in Phase II clinical trials with a total of 578 patients with constipation-predominant irritable bowel syndrome (IBS-C). As compared with placebo, the treatment groups reported better relief of their overall symptoms, namely abdominal pain and discomfort, increase in the number of pain free days, improved stool frequency, consistency and ease of passage of bowel movements. There were no significant differences in the reported Serious Adverse Events between treatment and placebo groups.[2][6][7]
In the largest of these Phase II trials, 510 subjects with IBS-C received either 1, 2 or 4 mg QD renzapride, or placebo QD for 12 weeks. The Weekly responder rate based on subject's assessment of whether they had relief from abdominal pain and/or discomfort associated with IBS during weeks 5-12 was 56% (renzapride 4 mg) vs 49% (placebo). For females the treatment effect was larger, 61% (renzapride 4 mg) vs 49% (placebo). Statistically significant effects in favour of renzapride were observed for improvements in stool consistency and increased bowel movements.[7]
In the Phase III clinical trial in IBS-C, 1798 female patients received either 2 or 4 mg Renzapride, or placebo once daily, for 12 weeks. The mean number of months with relief of overall symptoms was 0.6, 0.55 and 0.44 for renzapride 2 mg twice a day, renzapride 4 mg once a day and placebo, respectively, with both renzapride doses being statistically superior to placebo (p=0.004 and p=0.027, respectively). On responder analysis, the proportion of responders was 33.2%, 29.8%, and 24.3% for renzapride 2 mg twice a day, renzapride 4 mg once a day and placebo, respectively.[8]
The 8.9% delta between renzapride 2 mg twice daily and placebo compares favourably with other FDA approved therapies (Ford [9]).
References[edit]
- ^ a b Meyers NL, Hickling RI (2008). "Pharmacology and metabolism of renzapride : a novel therapeutic agent for the potential treatment of irritable bowel syndrome". Drugs in R&D. 9 (1): 37–63. doi:10.2165/00126839-200809010-00004. PMC 7044400. PMID 18095752.
- ^ a b Camilleri M, McKinzie S, Fox J, Foxx-Orenstein A, Burton D, Thomforde G, et al. (October 2004). "Effect of renzapride on transit in constipation-predominant irritable bowel syndrome". Clinical Gastroenterology and Hepatology. 2 (10): 895–904. doi:10.1016/s1542-3565(04)00391-x. PMID 15476153.
- ^ "FDA accepts cardiac safety trial for gastroparesis drug". Healio. 21 May 2018.
- ^ Quested T (26 June 2019). "Atlantic Healthcare pounces on big bucks US opportunity". Business Weekly.
- ^ Mackie AD, Ferrington C, Cowan S, Merrick MV, Baird JD, Palmer KR (April 1991). "The effects of renzapride, a novel prokinetic agent, in diabetic gastroparesis". Alimentary Pharmacology & Therapeutics. 5 (2): 135–142. doi:10.1111/j.1365-2036.1991.tb00014.x. PMID 1888816. S2CID 20428833.
- ^ Tack J, Middleton SJ, Horne MC, Piessevaux H, Bloor JS, Meyers NL, Palmer RM (June 2006). "Pilot study of the efficacy of renzapride on gastrointestinal motility and symptoms in patients with constipation-predominant irritable bowel syndrome". Alimentary Pharmacology & Therapeutics. 23 (11): 1655–1665. doi:10.1111/j.1365-2036.2006.02940.x. PMID 16696817. S2CID 25481560.
- ^ a b George AM, Meyers NL, Hickling RI (May 2008). "Clinical trial: renzapride therapy for constipation-predominant irritable bowel syndrome--multicentre, randomized, placebo-controlled, double-blind study in primary healthcare setting". Alimentary Pharmacology & Therapeutics. 27 (9): 830–837. doi:10.1111/j.1365-2036.2008.03649.x. PMID 18284648. S2CID 38167889.
- ^ Lembo AJ, Cremonini F, Meyers N, Hickling R (May 2010). "Clinical trial: renzapride treatment of women with irritable bowel syndrome and constipation - a double-blind, randomized, placebo-controlled, study". Alimentary Pharmacology & Therapeutics. 31 (9): 979–990. doi:10.1111/j.1365-2036.2010.04265.x. PMID 20163375.
- ^ Ford AC (July 2010). "Renzapride in IBS: is efficacy in the eye of the beholder?". Alimentary Pharmacology & Therapeutics. 32 (1): 113–114. doi:10.1111/j.1365-2036.2010.04275.x. PMID 20597877. S2CID 28565520.
