SRD5A1
3-Oxo-5α-steroid 4-dehydrogenase 1 is an enzyme that in humans is encoded by the SRD5A1 gene.[5] It is one of three forms of steroid 5α-reductase.
Steroid 5α-reductase (EC 1.3.99.5) catalyzes, among other reactions, the conversion of testosterone into the more potent androgen, 5α-dihydrotestosterone (DHT). The SRD5A1, SRD5A2, and SRD5A3 genes in humans all encode 5α-reductase isozymes.[6]
Function[edit]
The 3-oxo-5α-steroid 4-dehydrogenase 1 enzyme is involved in bile acid biosynthesis, androgen and estrogen metabolism. For instance, the enzyme catalyzes the conversion of testosterone into the more potent androgen, 5α-dihydrotestosterone. It can also catalyze the conversion of progesterone, corticosterone or other steroids, to its corresponding 5α-3-oxo-steroids. This chemical reaction is called 5α-reduction, i.e. the reduction of the Δ5-4 double bond in steroids by catalyzing direct hydride transfer from NADPH to the carbon 5 position of the steroid substrate.[7][8][9]
Regulation[edit]
ETV4 family members bind to ETS DNA-binding sites and both regulate their own expression and the transcription of a subset of genes that are dependent upon testicular luminal fluid factors, including Ggt_pr4, SRD5A1, and Gpx5.[10]
Six-month dietary vitamin E deficiency in rats resulted in a twofold increase in the mRNA level of SRD5A1 gene and a twofold decrease in the mRNA level of GCLM gene but is not directly mediated by changes in promoter DNA methylation.[11]
Insulin increases the expression of 5α-reductase type 1 mRNA via Akt signalling suggest that elevated levels of 5α-reduced androgens seen in hyperinsulinemic conditions might be explained on the basis of a stimulatory effect of insulin on 5α-reductase in granulosa cells leading to impaired follicle growth and ovulation.[12]
Clinical significance[edit]
Hyperinsulinemia acutely enhances ACTH effects on both the androgen and glucocorticoid pathways leading to changes in steroid metabolites molar ratios that suggest insulin stimulation of 5α-reductase activity.[13]
PCOS is associated with enhanced androgen and cortisol metabolite excretion and increased 5α-reductase activity that cannot be explained by obesity alone. Increased adrenal corticosteroid production represents an important pathogenic pathway in PCOS.[14]
Progression to castration-resistant prostate cancer (CRPC) is accompanied by increased expression of SRD5A1 over SRD5A2, which is otherwise the dominant isoenzyme expressed in the prostate. The dominant route of DHT synthesis in human CRPC bypasses testosterone, and instead requires 5α-reduction of androstenedione by SRD5A1 to 5α-androstanedione, which is then converted to DHT fuelling cancer growth.[15]
Expression[edit]

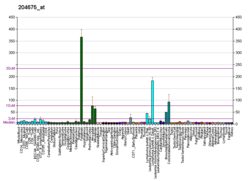
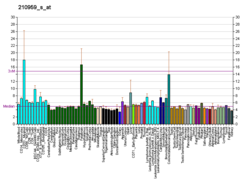
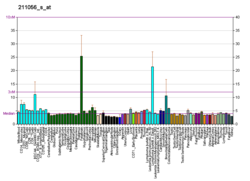
In humans, the protein isozyme encoded by the SRD5A1 gene is expressed in esophagus, liver, skin and 24 other tissues.[18][17]
See also[edit]
References[edit]
- ^ a b c GRCh38: Ensembl release 89: ENSG00000145545 – Ensembl, May 2017
- ^ a b c GRCm38: Ensembl release 89: ENSMUSG00000021594 – Ensembl, May 2017
- ^ "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ "Mouse PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ Jenkins EP, Hsieh CL, Milatovich A, Normington K, Berman DM, Francke U, Russell DW (December 1991). "Characterization and chromosomal mapping of a human steroid 5 alpha-reductase gene and pseudogene and mapping of the mouse homologue". Genomics. 11 (4): 1102–1112. doi:10.1016/0888-7543(91)90038-G. PMID 1686016.
- ^ "Entrez Gene: SRD5A1 steroid-5-alpha-reductase, alpha polypeptide 1 (3-oxo-5 alpha-steroid delta 4-dehydrogenase alpha 1)".
- ^ Nixon M, Upreti R, Andrew R (February 2012). "5α-Reduced glucocorticoids: a story of natural selection". The Journal of Endocrinology. 212 (2): 111–127. doi:10.1530/JOE-11-0318. PMID 21903862.
- ^ Azzouni F, Godoy A, Li Y, Mohler J (2012). "The 5 alpha-reductase isozyme family: a review of basic biology and their role in human diseases". Advances in Urology. 2012: 530121. doi:10.1155/2012/530121. PMC 3253436. PMID 22235201.
- ^ Han Y, Zhuang Q, Sun B, Lv W, Wang S, Xiao Q, et al. (January 2021). "Crystal structure of steroid reductase SRD5A reveals conserved steroid reduction mechanism". Nature Communications. 12 (1): 449. Bibcode:2021NatCo..12..449H. doi:10.1038/s41467-020-20675-2. PMC 7815742. PMID 33469028.
- ^ Yang L, Fox SA, Kirby JL, Troan BV, Hinton BT (April 2006). "Putative regulation of expression of members of the Ets variant 4 transcription factor family and their downstream targets in the rat epididymis". Biology of Reproduction. 74 (4): 714–720. doi:10.1095/biolreprod.105.044354. PMID 16394217.
- ^ Fischer A, Gaedicke S, Frank J, Döring F, Rimbach G (October 2010). "Dietary vitamin E deficiency does not affect global and specific DNA methylation patterns in rat liver". The British Journal of Nutrition. 104 (7): 935–940. doi:10.1017/S0007114510001649. PMID 20447326.
- ^ Kayampilly PP, Wanamaker BL, Stewart JA, Wagner CL, Menon KM (October 2010). "Stimulatory effect of insulin on 5alpha-reductase type 1 (SRD5A1) expression through an Akt-dependent pathway in ovarian granulosa cells". Endocrinology. 151 (10): 5030–5037. doi:10.1210/en.2010-0444. PMC 2946143. PMID 20810561.
- ^ Tosi F, Negri C, Brun E, Castello R, Faccini G, Bonora E, et al. (February 2011). "Insulin enhances ACTH-stimulated androgen and glucocorticoid metabolism in hyperandrogenic women". European Journal of Endocrinology. 164 (2): 197–203. doi:10.1530/EJE-10-0782. PMID 21059865.
- ^ Vassiliadi DA, Barber TM, Hughes BA, McCarthy MI, Wass JA, Franks S, et al. (September 2009). "Increased 5 alpha-reductase activity and adrenocortical drive in women with polycystic ovary syndrome". The Journal of Clinical Endocrinology and Metabolism. 94 (9): 3558–3566. doi:10.1210/jc.2009-0837. PMID 19567518.
- ^ Chang KH, Li R, Papari-Zareei M, Watumull L, Zhao YD, Auchus RJ, Sharifi N (August 2011). "Dihydrotestosterone synthesis bypasses testosterone to drive castration-resistant prostate cancer". Proceedings of the National Academy of Sciences of the United States of America. 108 (33): 13728–13733. Bibcode:2011PNAS..10813728C. doi:10.1073/pnas.1107898108. PMC 3158152. PMID 21795608.
- ^ "What the FPKM? A review of RNA-Seq expression units". The farrago. 2 May 2014. Archived from the original on 30 December 2019. Retrieved 2 November 2020.
- ^ a b Fagerberg L, Hallström BM, Oksvold P, Kampf C, Djureinovic D, Odeberg J, et al. (February 2014). "Analysis of the human tissue-specific expression by genome-wide integration of transcriptomics and antibody-based proteomics". Molecular & Cellular Proteomics. 13 (2): 397–406. doi:10.1074/mcp.M113.035600. PMC 3916642. PMID 24309898.
- ^ "SRD5A1 steroid 5 alpha-reductase 1 - Homo sapiens (human) - expression". Archived from the original on 28 January 2024. Retrieved 8 March 2024.
Further reading[edit]
- Jenkins EP, Andersson S, Imperato-McGinley J, Wilson JD, Russell DW (January 1992). "Genetic and pharmacological evidence for more than one human steroid 5 alpha-reductase". The Journal of Clinical Investigation. 89 (1): 293–300. doi:10.1172/JCI115574. PMC 442847. PMID 1345916.
- Andersson S, Russell DW (May 1990). "Structural and biochemical properties of cloned and expressed human and rat steroid 5 alpha-reductases". Proceedings of the National Academy of Sciences of the United States of America. 87 (10): 3640–3644. Bibcode:1990PNAS...87.3640A. doi:10.1073/pnas.87.10.3640. PMC 53958. PMID 2339109.
- Moore RJ, Wilson JD (February 1972). "Localization of the reduced nicotinamide adenine dinucleotide phosphate: 4 -3-ketosteroid 5 -oxidoreductase in the nuclear membrane of the rat ventral prostate". The Journal of Biological Chemistry. 247 (3): 958–967. doi:10.1016/S0021-9258(19)45700-1. PMID 4400474.
- Milewich L, Kaimal V, Toews GB (May 1983). "Androstenedione metabolism in human alveolar macrophages". The Journal of Clinical Endocrinology and Metabolism. 56 (5): 920–924. doi:10.1210/jcem-56-5-920. PMID 6833469.
- Thigpen AE, Silver RI, Guileyardo JM, Casey ML, McConnell JD, Russell DW (August 1993). "Tissue distribution and ontogeny of steroid 5 alpha-reductase isozyme expression". The Journal of Clinical Investigation. 92 (2): 903–910. doi:10.1172/JCI116665. PMC 294929. PMID 7688765.
- Antonipillai I, Wahe M, Yamamoto J, Horton R (January 1995). "Activin and inhibin have opposite effects on steroid 5 alpha-reductase activity in genital skin fibroblasts". Molecular and Cellular Endocrinology. 107 (1): 99–104. doi:10.1016/0303-7207(94)03430-2. PMID 7796940. S2CID 21654090.
- Hirsch KS, Jones CD, Audia JE, Andersson S, McQuaid L, Stamm NB, et al. (June 1993). "LY191704: a selective, nonsteroidal inhibitor of human steroid 5 alpha-reductase type 1". Proceedings of the National Academy of Sciences of the United States of America. 90 (11): 5277–5281. Bibcode:1993PNAS...90.5277H. doi:10.1073/pnas.90.11.5277. PMC 46699. PMID 8389478.
- Andersson B, Wentland MA, Ricafrente JY, Liu W, Gibbs RA (April 1996). "A "double adaptor" method for improved shotgun library construction". Analytical Biochemistry. 236 (1): 107–113. doi:10.1006/abio.1996.0138. PMID 8619474.
- Bonkhoff H, Stein U, Aumüller G, Remberger K (October 1996). "Differential expression of 5 alpha-reductase isoenzymes in the human prostate and prostatic carcinomas". The Prostate. 29 (4): 261–267. doi:10.1002/(SICI)1097-0045(199610)29:4<261::AID-PROS7>3.0.CO;2-E. PMID 8876709. S2CID 29826751.
- Yu W, Andersson B, Worley KC, Muzny DM, Ding Y, Liu W, et al. (April 1997). "Large-scale concatenation cDNA sequencing". Genome Research. 7 (4): 353–358. doi:10.1101/gr.7.4.353. PMC 139146. PMID 9110174.
- Ellis JA, Stebbing M, Harrap SB (June 1998). "Genetic analysis of male pattern baldness and the 5alpha-reductase genes". The Journal of Investigative Dermatology. 110 (6): 849–853. doi:10.1046/j.1523-1747.1998.00224.x. PMID 9620288.
- Zhou Z, Shackleton CH, Pahwa S, White PC, Speiser PW (March 1998). "Prominent sex steroid metabolism in human lymphocytes". Molecular and Cellular Endocrinology. 138 (1–2): 61–69. doi:10.1016/S0303-7207(98)00052-5. PMID 9685215. S2CID 20490901.
- Hu RM, Han ZG, Song HD, Peng YD, Huang QH, Ren SX, et al. (August 2000). "Gene expression profiling in the human hypothalamus-pituitary-adrenal axis and full-length cDNA cloning". Proceedings of the National Academy of Sciences of the United States of America. 97 (17): 9543–9548. Bibcode:2000PNAS...97.9543H. doi:10.1073/pnas.160270997. PMC 16901. PMID 10931946.
- Eminović I, Liović M, Prezelj J, Kocijancic A, Rozman D, Komel R (2002). "New steroid 5alpha-reductase type I (SRD5A1) homologous sequences on human chromosomes 6 and 8". Pflügers Archiv. 442 (6 Suppl 1): R187–R189. doi:10.1007/s004240100019. PMID 11678334. S2CID 21081236.
- Ellis JA, Wong ZY, Stebbing M, Harrap SB (December 2001). "Sex, genes and blood pressure". Clinical and Experimental Pharmacology & Physiology. 28 (12): 1053–1055. doi:10.1046/j.1440-1681.2001.03579.x. PMID 11903314. S2CID 23923031.
- Rodríguez-Dorantes M, Lizano-Soberón M, Camacho-Arroyo I, Calzada-León R, Morimoto S, Téllez-Ascencio N, Cerbón MA (March 2002). "Evidence that steroid 5alpha-reductase isozyme genes are differentially methylated in human lymphocytes". The Journal of Steroid Biochemistry and Molecular Biology. 80 (3): 323–330. doi:10.1016/S0960-0760(02)00023-7. PMID 11948017. S2CID 20292300.
- Ha SJ, Kim JS, Myung JW, Lee HJ, Kim JW (April 2003). "Analysis of genetic polymorphisms of steroid 5alpha-reductase type 1 and 2 genes in Korean men with androgenetic alopecia". Journal of Dermatological Science. 31 (2): 135–141. doi:10.1016/S0923-1811(02)00145-7. PMID 12670724.
- Habib FK, Ross M, Bayne CW, Bollina P, Grigor K, Chapman K (May 2003). "The loss of 5alpha-reductase type I and type II mRNA expression in metastatic prostate cancer to bone and lymph node metastasis". Clinical Cancer Research. 9 (5): 1815–1819. PMID 12738739.