Aromatase
Aromatase (EC 1.14.14.14), also called estrogen synthetase or estrogen synthase, is an enzyme responsible for a key step in the biosynthesis of estrogens. It is CYP19A1, a member of the cytochrome P450 superfamily, which are monooxygenases that catalyze many reactions involved in steroidogenesis. In particular, aromatase is responsible for the aromatization of androgens into estrogens. The enzyme aromatase can be found in many tissues including gonads (granulosa cells), brain, adipose tissue, placenta, blood vessels, skin, and bone, as well as in tissue of endometriosis, uterine fibroids, breast cancer, and endometrial cancer.[citation needed] It is an important factor in sexual development.
Function[edit]
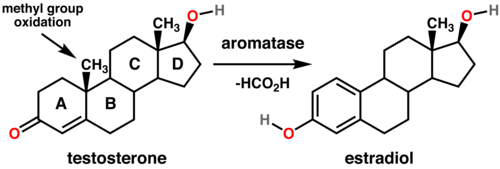
Aromatase is localized in the endoplasmic reticulum where it is regulated by tissue-specific promoters that are in turn controlled by hormones, cytokines, and other factors. It catalyzes the last steps of estrogen biosynthesis from androgens (specifically, it transforms androstenedione to estrone and testosterone to estradiol). These steps include three successive hydroxylations of the 19-methyl group of androgens, followed by simultaneous elimination of the methyl group as formate and aromatization of the A-ring.
- Androstenedione + 3O2 + 3NADPH + 3H+ Estrone + Formate + 4H2O + 3NADP+
- Testosterone + 3O2 + 3NADPH + 3H+ 17β-estradiol + Formate + 4H2O + 3NADP+
 |
 |
Expression[edit]
Aromatase is expressed in the gonads, placenta, brain, adipose tissue, bone, and other tissues.[citation needed] It is almost undetectable in adult human liver.[6]
Genomics[edit]
The gene expresses two transcript variants.[7] In humans, the gene CYP19, located on chromosome 15q21.1, encodes aromatase.[8] The gene has nine coding exons and a number of alternative non-coding first exons that regulate tissue specific expression.[9]
CYP19 is present in an early-diverging chordate, the cephalochordate amphioxus (the Florida lancelet, Branchiostoma floridae), but not in the earlier diverging tunicate Ciona intestinalis. Thus, the aromatase gene evolved early in chordate evolution and does not appear to be present in nonchordate invertebrates (e.g. insects, molluscs, echinoderms, sponges, corals). However, estrogens may be synthesized in some of these organisms, via other unknown pathways.
Activity[edit]
Aromatase activity is increased by age, obesity, insulin, gonadotropins, and alcohol.[10] It also appears to be enhanced in certain estrogen-dependent local tissue next to breast tissue, endometrial cancer, endometriosis, and uterine fibroids.[10]
Aromatase activity is decreased or antagonized by prolactin, anti-Müllerian hormone and glyphosate.[10]
Role in sex-determination[edit]
Aromatase is generally highly present during the differentiation of ovaries.[11][12] It is also susceptible to environmental influences, particularly temperature. In species with temperature-dependent sex determination, aromatase is expressed in higher quantities at temperatures that yield female offspring.[11] Despite the fact that data suggest temperature controls aromatase quantities, other studies have shown that aromatase can overpower the effects of temperature: if exposed to more aromatase at a male-producing temperature, the organism will develop female and conversely, if exposed to less aromatase at female-producing temperatures, the organism will develop male (see sex reversal).[11] In organisms that develop through genetic sex determination, temperature does not affect aromatase expression and function, suggesting that aromatase is the target molecule for temperature during TSD[11] (for challenges to this argument, see temperature-dependent sex determination). It varies from species to species whether it is the aromatase protein that has different activity at different temperatures or whether the amount of transcription undergone by the aromatase gene is what is temperature-sensitive, but in either case, differential development is observed at different temperatures.[13]
Role in neuroprotection[edit]
Aromatase in the brain is usually only expressed in neurons. However, following penetrative brain injury of both mice and zebra finches, it has been shown to be expressed in astrocytes.[14] It has also been shown to decrease apoptosis following brain injury in zebra finches.[15] This is thought to be due to the neuroprotective actions of estrogens, including estradiol. Research has found that two pro-inflammatory cytokines, interleukin-1β (IL-1β) and interleukin-6 (IL-6), are responsible for the induction of aromatase expression in astrocytes following penetrative brain injury in the zebra finch.[16]
Disorders[edit]
Aromatase excess syndrome[edit]
A number of investigators have reported on a rather rare syndrome of excess aromatase activity. In boys, it creates gynecomastia, and in girls, precocious puberty and gigantomastia. In both sexes, early epiphyseal closure leads to short stature. This condition is due to mutations in the CYP19A1 gene which encodes aromatase.[17] It is inherited in an autosomal dominant fashion.[18] It has been suggested that the pharaoh Akhenaten and other members of his family may have had from this disorder,[19] but more recent genetic tests suggest otherwise.[20] It is one of the causes of familial precocious puberty—a condition first described in 1937.[21]
Aromatase deficiency syndrome[edit]
This syndrome is due to a mutation of gene CYP19 and inherited in an autosomal recessive way. Accumulations of androgens during pregnancy may lead to virilization of a female at birth (males are not affected). Females will have primary amenorrhea. Individuals of both sexes will be tall, as lack of estrogen does not bring the epiphyseal lines to closure.
Inhibition of aromatase[edit]
The inhibition of aromatase can cause hypoestrogenism (low estrogen levels). The following natural products have been found to have inhibiting effects on aromatase.
Extracts of certain (white button variety: Agaricus bisporus) mushrooms have been shown to inhibit aromatase in vitro.[32]
Pharmaceutical aromatase inhibitors[edit]
Aromatase inhibitors, which stop the production of estrogen in postmenopausal women, have become useful in the management of patients with breast cancer whose lesion was found to be estrogen receptor positive.[33] Inhibitors that are in current clinical use include anastrozole, exemestane, and letrozole. Aromatase inhibitors are also beginning to be prescribed to men on testosterone replacement therapy as a way to keep estrogen levels from spiking once doses of testosterone are introduced to their systems.
See also[edit]
References[edit]
- ^ a b c GRCh38: Ensembl release 89: ENSG00000137869 – Ensembl, May 2017
- ^ a b c GRCm38: Ensembl release 89: ENSMUSG00000032274 – Ensembl, May 2017
- ^ "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ "Mouse PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ Vaz AD (2003). "Chapter 1: Cytochrome activation by cytochromes P450: a role for multiple oxidants in the oxidation of substrates". In Fisher M, Lee JK, Obach RE (eds.). Drug metabolizing enzymes: cytochrome P450 and other enzymes in drug discovery and development. Lausanne, Switzerland: FontisMedia SA. ISBN 978-0-8247-4293-5.
- ^ Hata S, Miki Y, Saito R, Ishida K, Watanabe M, Sasano H (June 2013). "Aromatase in human liver and its diseases". Cancer Med. 2 (3): 305–15. doi:10.1002/cam4.85. PMC 3699842. PMID 23930207.
- ^ "Entrez Gene: CYP19A1 cytochrome P450, family 19, subfamily A, polypeptide 1".
- ^ Toda K, Shizuta Y (April 1993). "Molecular cloning of a cDNA showing alternative splicing of the 5'-untranslated sequence of mRNA for human aromatase P-450". European Journal of Biochemistry. 213 (1): 383–9. doi:10.1111/j.1432-1033.1993.tb17772.x. PMID 8477708.
- ^ Czajka-Oraniec I, Simpson ER (2010). "Aromatase research and its clinical significance". Endokrynologia Polska. 61 (1): 126–34. PMID 20205115.
- ^ a b c Hussain A, Gilloteaux J (2020-09-01). "The human testes: Estrogen and ageing outlooks". Translational Research in Anatomy. 20: 100073. doi:10.1016/j.tria.2020.100073. ISSN 2214-854X. S2CID 219001284.
- ^ a b c d Duffy TA, Picha ME, Won ET, Borski RJ, McElroy AE, Conover DO (August 2010). "Ontogenesis of gonadal aromatase gene expression in atlantic silverside (Menidia menidia) populations with genetic and temperature-dependent sex determination". Journal of Experimental Zoology Part A. 313 (7): 421–31. Bibcode:2010JEZA..313..421D. doi:10.1002/jez.612. PMID 20623799.
- ^ Kohno S, Katsu Y, Urushitani H, Ohta Y, Iguchi T, Guillette LJ (2010). "Potential contributions of heat shock proteins to temperature-dependent sex determination in the American alligator". Sexual Development. 4 (1–2): 73–87. doi:10.1159/000260374. PMC 2855287. PMID 19940440.
- ^ Gilbert SF (2010). Developmental biology. Sunderland, Mass: Sinauer Associates. ISBN 978-0-87893-384-6.
- ^ Garcia-Segura LM, Wozniak A, Azcoitia I, Rodriguez JR, Hutchison RE, Hutchison JB (March 1999). "Aromatase expression by astrocytes after brain injury: implications for local estrogen formation in brain repair". Neuroscience. 89 (2): 567–78. doi:10.1016/s0306-4522(98)00340-6. PMID 10077336. S2CID 24689059.
- ^ Saldanha CJ, Rohmann KN, Coomaralingam L, Wynne RD (August 2005). "Estrogen provision by reactive glia decreases apoptosis in the zebra finch (Taeniopygia guttata)". Journal of Neurobiology. 64 (2): 192–201. doi:10.1002/neu.20147. PMID 15818556.
- ^ Duncan KA, Saldanha CJ (July 2011). "Neuroinflammation induces glial aromatase expression in the uninjured songbird brain". Journal of Neuroinflammation. 8 (81): 81. doi:10.1186/1742-2094-8-81. PMC 3158750. PMID 21767382.
- ^ Fukami M, Shozu M, Ogata T (2012). "Molecular bases and phenotypic determinants of aromatase excess syndrome". International Journal of Endocrinology. 2012: 584807. doi:10.1155/2012/584807. PMC 3272822. PMID 22319526.
- ^ Fukami M, Shozu M, Soneda S, Kato F, Inagaki A, Takagi H, Hanaki K, Kanzaki S, Ohyama K, Sano T, Nishigaki T, Yokoya S, Binder G, Horikawa R, Ogata T (June 2011). "Aromatase excess syndrome: identification of cryptic duplications and deletions leading to gain of function of CYP19A1 and assessment of phenotypic determinants". The Journal of Clinical Endocrinology and Metabolism. 96 (6): E1035-43. doi:10.1210/jc.2011-0145. PMID 21470988.
- ^ Braverman IM, Redford DB, Mackowiak PA (April 2009). "Akhenaten and the strange physiques of Egypt's 18th dynasty". Annals of Internal Medicine. 150 (8): 556–60. doi:10.7326/0003-4819-150-8-200904210-00010. PMID 19380856. S2CID 24766974.
- ^ Seshadri KG (May 2012). "The breasts of Tutankhamun". Indian Journal of Endocrinology and Metabolism. 16 (3): 429–30. doi:10.4103/2230-8210.95696. PMC 3354854. PMID 22629513.
- ^ Ziora K, Oświecimska J, Geisler G, Broll-Waśka K, Szalecki M, Dyduch A (2006). "[Familial precocious puberty -- a variant of norm or pathology?]". Endokrynologia, Diabetologia I Choroby Przemiany Materii Wieku Rozwojowego (in Polish). 12 (1): 53–8. PMID 16704862.
- ^ a b c d Balunas MJ, Su B, Brueggemeier RW, Kinghorn AD (August 2008). "Natural products as aromatase inhibitors". Anti-Cancer Agents in Medicinal Chemistry. 8 (6): 646–82. doi:10.2174/1871520610808060646. PMC 3074486. PMID 18690828.
- ^ Satoh K, Sakamoto Y, Ogata A, Nagai F, Mikuriya H, Numazawa M, Yamada K, Aoki N (July 2002). "Inhibition of aromatase activity by green tea extract catechins and their endocrinological effects of oral administration in rats". Food and Chemical Toxicology. 40 (7): 925–33. doi:10.1016/S0278-6915(02)00066-2. PMID 12065214.
- ^ Kapiszewska M, Miskiewicz M, Ellison PT, Thune I, Jasienska G (May 2006). "High tea consumption diminishes salivary 17beta-estradiol concentration in Polish women". The British Journal of Nutrition. 95 (5): 989–95. doi:10.1079/BJN20061755. hdl:11315/483. PMID 16611391.
- ^ Le Bail JC, Pouget C, Fagnere C, Basly JP, Chulia AJ, Habrioux G (January 2001). "Chalcones are potent inhibitors of aromatase and 17beta-hydroxysteroid dehydrogenase activities". Life Sciences. 68 (7): 751–61. doi:10.1016/S0024-3205(00)00974-7. PMID 11205867.
- ^ Ye L, Chan FL, Chen S, Leung LK (October 2012). "The citrus flavonone hesperetin inhibits growth of aromatase-expressing MCF-7 tumor in ovariectomized athymic mice". The Journal of Nutritional Biochemistry. 23 (10): 1230–7. doi:10.1016/j.jnutbio.2011.07.003. PMID 22209285.
- ^ Doering IL, Richter E (April 2009). "Inhibition of human aromatase by myosmine". Drug Metabolism Letters. 3 (2): 83–6. doi:10.2174/187231209788654045. PMID 19601869.
- ^ Biegon A, Kim SW, Logan J, Hooker JM, Muench L, Fowler JS (April 2010). "Nicotine blocks brain estrogen synthase (aromatase): in vivo positron emission tomography studies in female baboons". Biological Psychiatry. 67 (8): 774–7. doi:10.1016/j.biopsych.2010.01.004. PMC 2904480. PMID 20188349.
- ^ Wang Y, Lee KW, Chan FL, Chen S, Leung LK (July 2006). "The red wine polyphenol resveratrol displays bilevel inhibition on aromatase in breast cancer cells". Toxicological Sciences. 92 (1): 71–7. doi:10.1093/toxsci/kfj190. PMID 16611627.
- ^ Siler U, Barella L, Spitzer V, Schnorr J, Lein M, Goralczyk R, Wertz K (June 2004). "Lycopene and vitamin E interfere with autocrine/paracrine loops in the Dunning prostate cancer model". FASEB Journal. 18 (9): 1019–21. doi:10.1096/fj.03-1116fje. PMID 15084515. S2CID 26424745.
- ^ Om AS, Chung KW (April 1996). "Dietary zinc deficiency alters 5 alpha-reduction and aromatization of testosterone and androgen and estrogen receptors in rat liver". The Journal of Nutrition. 126 (4): 842–8. doi:10.1093/jn/126.4.842. PMID 8613886.
- ^ Chen S, Oh SR, Phung S, Hur G, Ye JJ, Kwok SL, Shrode GE, Belury M, Adams LS, Williams D (December 2006). "Anti-aromatase activity of phytochemicals in white button mushrooms (Agaricus bisporus)". Cancer Research. 66 (24): 12026–34. doi:10.1158/0008-5472.CAN-06-2206. PMID 17178902.
- ^ "Aromatase Inhibitors". Breastcancer.org. 29 October 2020.
Further reading[edit]
- Attar E, Bulun SE (May 2006). "Aromatase inhibitors: the next generation of therapeutics for endometriosis?". Fertility and Sterility. 85 (5): 1307–18. doi:10.1016/j.fertnstert.2005.09.064. PMID 16647373.
- Chen S (August 1998). "Aromatase and breast cancer". Frontiers in Bioscience. 3 (4): d922–33. doi:10.2741/A333. PMID 9696881.
- Strobel HW, Thompson CM, Antonovic L (June 2001). "Cytochromes P450 in brain: function and significance". Current Drug Metabolism. 2 (2): 199–214. doi:10.2174/1389200013338577. PMID 11469726.
- Simpson ER, Clyne C, Rubin G, Boon WC, Robertson K, Britt K, Speed C, Jones M (2002). "Aromatase--a brief overview". Annual Review of Physiology. 64: 93–127. doi:10.1146/annurev.physiol.64.081601.142703. PMID 11826265.
- Bulun SE, Yang S, Fang Z, Gurates B, Tamura M, Zhou J, Sebastian S (December 2001). "Role of aromatase in endometrial disease". The Journal of Steroid Biochemistry and Molecular Biology. 79 (1–5): 19–25. doi:10.1016/S0960-0760(01)00134-0. PMID 11850203. S2CID 7642211.
- Balthazart J, Baillien M, Ball GF (December 2001). "Phosphorylation processes mediate rapid changes of brain aromatase activity". The Journal of Steroid Biochemistry and Molecular Biology. 79 (1–5): 261–77. doi:10.1016/S0960-0760(01)00143-1. PMID 11850233. S2CID 34269540.
- Richards JA, Petrel TA, Brueggemeier RW (February 2002). "Signaling pathways regulating aromatase and cyclooxygenases in normal and malignant breast cells". The Journal of Steroid Biochemistry and Molecular Biology. 80 (2): 203–12. doi:10.1016/S0960-0760(01)00187-X. PMID 11897504. S2CID 12728545.
- Balthazart J, Baillien M, Ball GF (May 2002). "Interactions between aromatase (estrogen synthase) and dopamine in the control of male sexual behavior in quail". Comparative Biochemistry and Physiology. Part B, Biochemistry & Molecular Biology. 132 (1): 37–55. doi:10.1016/S1096-4959(01)00531-0. PMID 11997208.
- Meinhardt U, Mullis PE (2002). "The aromatase cytochrome P-450 and its clinical impact". Hormone Research. 57 (5–6): 145–52. doi:10.1159/000058374. PMID 12053085. S2CID 24115725.
- Carreau S, Bourguiba S, Lambard S, Galeraud-Denis I, Genissel C, Levallet J (July 2002). "Reproductive system: aromatase and estrogens". Molecular and Cellular Endocrinology. 193 (1–2): 137–43. doi:10.1016/S0303-7207(02)00107-7. PMID 12161013. S2CID 34446206.
- Meinhardt U, Mullis PE (August 2002). "The essential role of the aromatase/p450arom". Seminars in Reproductive Medicine. 20 (3): 277–84. doi:10.1055/s-2002-35374. PMID 12428207. S2CID 25407830.
- Carreau S, Bourguiba S, Lambard S, Galeraud-Denis I (2003). "[Testicular aromatase]". Journal de la Société de Biologie. 196 (3): 241–4. doi:10.1051/jbio/2002196030241. PMID 12462076.
- Carani C, Fabbi M, Zirilli L, Sgarbi I (2003). "[Estrogen resistance and aromatase deficiency in humans]". Journal de la Société de Biologie. 196 (3): 245–8. doi:10.1051/jbio/2002196030245. PMID 12462077.
- Kragie L (August 2002). "Aromatase in primate pregnancy: a review". Endocrine Research. 28 (3): 121–8. doi:10.1081/ERC-120015041. PMID 12489562. S2CID 8509882.
- Simpson ER (July 2000). "Biology of aromatase in the mammary gland". Journal of Mammary Gland Biology and Neoplasia. 5 (3): 251–8. doi:10.1023/A:1009590626450. PMID 14973387. S2CID 13296474.
- Bulun SE, Takayama K, Suzuki T, Sasano H, Yilmaz B, Sebastian S (February 2004). "Organization of the human aromatase p450 (CYP19) gene". Seminars in Reproductive Medicine. 22 (1): 5–9. doi:10.1055/s-2004-823022. PMID 15083376. S2CID 260316748.
- Simpson ER (February 2004). "Aromatase: biologic relevance of tissue-specific expression". Seminars in Reproductive Medicine. 22 (1): 11–23. doi:10.1055/s-2004-823023. PMID 15083377. S2CID 260319521.
- Bulun SE, Fang Z, Imir G, Gurates B, Tamura M, Yilmaz B, Langoi D, Amin S, Yang S, Deb S (February 2004). "Aromatase and endometriosis". Seminars in Reproductive Medicine. 22 (1): 45–50. doi:10.1055/s-2004-823026. PMID 15083380. S2CID 260319473.
- Shozu M, Murakami K, Inoue M (February 2004). "Aromatase and leiomyoma of the uterus". Seminars in Reproductive Medicine. 22 (1): 51–60. doi:10.1055/s-2004-823027. PMID 15083381. S2CID 260319833.
- Chen S, Ye J, Kijima I, Kinoshita Y, Zhou D (May 2005). "Positive and negative transcriptional regulation of aromatase expression in human breast cancer tissue". The Journal of Steroid Biochemistry and Molecular Biology. 95 (1–5): 17–23. doi:10.1016/j.jsbmb.2005.04.002. PMID 15955695. S2CID 22138523.
- Lambard S, Silandre D, Delalande C, Denis-Galeraud I, Bourguiba S, Carreau S (May 2005). "Aromatase in testis: expression and role in male reproduction". The Journal of Steroid Biochemistry and Molecular Biology. 95 (1–5): 63–9. doi:10.1016/j.jsbmb.2005.04.020. PMID 16019206. S2CID 40087589.
- Bulun SE, Imir G, Utsunomiya H, Thung S, Gurates B, Tamura M, Lin Z (May 2005). "Aromatase in endometriosis and uterine leiomyomata". The Journal of Steroid Biochemistry and Molecular Biology. 95 (1–5): 57–62. doi:10.1016/j.jsbmb.2005.04.012. PMID 16024248. S2CID 37228186.
- Lambard S, Carreau S (October 2005). "Aromatase and oestrogens in human male germ cells". International Journal of Andrology. 28 (5): 254–9. doi:10.1111/j.1365-2605.2005.00546.x. PMID 16128984.
- Ellem SJ, Risbridger GP (March 2006). "Aromatase and prostate cancer". Minerva Endocrinologica. 31 (1): 1–12. PMID 16498360.
- Brueggemeier RW, Díaz-Cruz ES (March 2006). "Relationship between aromatase and cyclooxygenases in breast cancer: potential for new therapeutic approaches". Minerva Endocrinologica. 31 (1): 13–26. PMID 16498361.
- Jongen VH, Hollema H, Van Der Zee AG, Heineman MJ (March 2006). "Aromatase in the context of breast and endometrial cancer. A review". Minerva Endocrinologica. 31 (1): 47–60. PMID 16498363.
- Hiltunen M, Iivonen S, Soininen H (March 2006). "Aromatase enzyme and Alzheimer's disease". Minerva Endocrinologica. 31 (1): 61–73. PMID 16498364.
External links[edit]
- "U.S. study of gay sheep may shed light on sexuality", via WikiNews, 15 August 2005.
- Human CYP19A1 genome location and CYP19A1 gene details page in the UCSC Genome Browser.
- Overview of all the structural information available in the PDB for UniProt: P11511 (Aromatase) at the PDBe-KB.