User:Padibso/sandbox
Hydrogen is a chemical element; it has symbol H and atomic number 1. It is the lightest element and, at standard conditions, is a gas of diatomic molecules with the formula H2. It is colourless, odourless, tasteless,[1] non-toxic, and highly combustible. Hydrogen is the most abundant chemical substance in the universe, constituting roughly 75% of all normal matter.[2][note 1] Stars such as the Sun are mainly composed of hydrogen in the plasma state. Most of the hydrogen on Earth exists in molecular forms such as water and organic compounds. For the most common isotope of hydrogen (symbol 1H) each atom has one proton, one electron, and no neutrons.
Hydrogen is generally nonmetallic (except when it becomes metallic at extremely high pressures) and readily forms a single covalent bond with most nonmetallic elements, forming compounds such as water and nearly all organic compounds. Hydrogen plays a particularly important role in acid–base reactions because these reactions usually involve the exchange of protons between soluble molecules. In ionic compounds, hydrogen can take the form of a negative charge (i.e., anion) where it is known as a hydride, or as a positively charged (i.e., cation) species denoted by the symbol H+. The H+ cation is simply a proton (symbol p) but its behaviour in aqueous solutions and in ionic compounds involves screening of its electric charge by nearby polar molecules or anions. Because hydrogen is the only neutral atom for which the Schrödinger equation can be solved analytically,[3] the study of its energetics and chemical bonding has played a key role in the development of quantum mechanics.
Hydrogen gas was first artificially produced in the early 16th century by the reaction of acids on metals. In 1766–1781, Henry Cavendish was the first to recognize that hydrogen gas was a discrete substance[4] and that it produces water when burned, the property for which it was later named: in Greek, hydrogen means "water-former".
Industrial production is mainly from steam reforming of natural gas, oil reforming, or coal gasification. A small percentage is also produced using such as the electrolysis of water.[5][6] Most hydrogen is used near the site of its production, the two largest uses being fossil fuel processing (e.g., hydrocracking) and ammonia production. Hydrogen can be deployed as an energy source in fuel cells to produce electricity, or via combustion to generate heat.[7] When hydrogen is consumed in fuel cells, the only emission at the point of use is water vapour.[7] Combustion of hydrogen can lead to the thermal formation of nitrogen oxides.[7] Hydrogen atoms may embrittle metals.[8]
Properties[edit]
Combustion[edit]

Hydrogen gas (dihydrogen or molecular hydrogen)[9] is highly flammable:
- 2 H2(g) + O2(g) → 2 H2O(l) (572 kJ/2 mol = 286 kJ/mol = 141.865 MJ/kg)[note 2]
The enthalpy of combustion is −286 kJ/mol.[10]
Hydrogen gas forms explosive mixtures with air in concentrations from 4–74%[11] and with chlorine at 5–95%. The hydrogen autoignition temperature, the temperature of spontaneous ignition in air, is 500 °C (932 °F).[12]
Flame[edit]
Pure hydrogen-oxygen flames emit ultraviolet light and with high oxygen mix are nearly invisible to the naked eye, as illustrated by the faint plume of the Space Shuttle Main Engine, compared to the highly visible plume of a Space Shuttle Solid Rocket Booster, which uses an ammonium perchlorate composite. The detection of a burning hydrogen leak may require a flame detector; such leaks can be very dangerous. Hydrogen flames in other conditions are blue, resembling blue natural gas flames.[13] The destruction of the Hindenburg airship was a notorious example of hydrogen combustion and the cause is still debated. The visible flames in the photographs were the result of carbon compounds in the airship skin burning.[14]
Reactants[edit]
H2 is unreactive compared to diatomic elements such as halogens or oxygen. The thermodynamic basis of this low reactivity is the very strong H–H bond, with a bond dissociation energy of 435.7 kJ/mol.[15] The kinetic basis of the low reactivity is the nonpolar nature of H2 and its weak polarizability. It spontaneously reacts with chlorine and fluorine to form hydrogen chloride and hydrogen fluoride, respectively.[16] The reactivity of H2 is strongly affected by the presence of metal catalysts. Thus, while mixtures of H2 with O2 or air combust readily when heated to at least 500 °C by a spark or flame, they do not react at room temperature in the absence of a catalyst.
Electron energy levels[edit]

The ground state energy level of the electron in a hydrogen atom is −13.6 eV,[17] which is equivalent to an ultraviolet photon of roughly 91 nm wavelength.[18]
The energy levels of hydrogen can be calculated fairly accurately using the Bohr model of the atom, which conceptualizes the electron as "orbiting" the proton in analogy to the Earth's orbit of the Sun. However, the atomic electron and proton are held together by electromagnetic force, while planets and celestial objects are held by gravity. Because of the discretization of angular momentum postulated in early quantum mechanics by Bohr, the electron in the Bohr model can only occupy certain allowed distances from the proton, and therefore only certain allowed energies.[19]
A more accurate description of the hydrogen atom comes from a purely quantum mechanical treatment that uses the Schrödinger equation, Dirac equation or Feynman path integral formulation to calculate the probability density of the electron around the proton.[20] The most complicated treatments allow for the small effects of special relativity and vacuum polarization. In the quantum mechanical treatment, the electron in a ground state hydrogen atom has no angular momentum at all—illustrating how the "planetary orbit" differs from electron motion.
Spin isomers[edit]
Molecular H2 exists as two spin isomers, i.e. compounds that differ only in the spin states of their nuclei.[21] In the orthohydrogen form, the spins of the two nuclei are parallel, forming a spin triplet state having a total molecular spin ; in the parahydrogen form the spins are antiparallel and form a spin singlet state having spin . The equilibrium ratio of ortho- to para-hydrogen depends on temperature. At room temperature or warmer, equilibrium hydrogen gas contains about 25% of the para form and 75% of the ortho form.[22] The ortho form is an excited state, having higher energy than the para form by 1.455 kJ/mol,[23] and it converts to the para form over the course of several minutes when cooled to low temperature.[24] The thermal properties of the forms differ because they differ in their allowed rotational quantum states, resulting in different thermal properties such as the heat capacity.[25]
The ortho-to-para ratio in H2 is an important consideration in the liquefaction and storage of liquid hydrogen: the conversion from ortho to para is exothermic and produces enough heat to evaporate most of the liquid if not converted first to parahydrogen during the cooling process.[26] Catalysts for the ortho-para interconversion, such as ferric oxide and activated carbon compounds, are used during hydrogen cooling to avoid this loss of liquid.[27]
Phases[edit]


- Gaseous hydrogen
- Liquid hydrogen
- Slush hydrogen
- Solid hydrogen
- Metallic hydrogen
- Plasma hydrogen
Compounds[edit]
Covalent and organic compounds[edit]
While H2 is not very reactive under standard conditions, it does form compounds with most elements. Hydrogen can form compounds with elements that are more electronegative, such as halogens (F, Cl, Br, I), or oxygen; in these compounds hydrogen takes on a partial positive charge.[28] When bonded to a more electronegative element, particularly fluorine, oxygen, or nitrogen, hydrogen can participate in a form of medium-strength noncovalent bonding with another electronegative element with a lone pair, a phenomenon called hydrogen bonding that is critical to the stability of many biological molecules.[29][30] Hydrogen also forms compounds with less electronegative elements, such as metals and metalloids, where it takes on a partial negative charge. These compounds are often known as hydrides.[31]
Hydrogen forms a vast array of compounds with carbon called the hydrocarbons, and an even vaster array with heteroatoms that, because of their general association with living things, are called organic compounds.[32] The study of their properties is known as organic chemistry[33] and their study in the context of living organisms is known as biochemistry.[34] By some definitions, "organic" compounds are only required to contain carbon. However, most of them also contain hydrogen, and because it is the carbon-hydrogen bond that gives this class of compounds most of its particular chemical characteristics, carbon-hydrogen bonds are required in some definitions of the word "organic" in chemistry.[32] Millions of hydrocarbons are known, and they are usually formed by complicated pathways that seldom involve elemental hydrogen.
Hydrogen is highly soluble in many rare earth and transition metals[35] and is soluble in both nanocrystalline and amorphous metals.[36] Hydrogen solubility in metals is influenced by local distortions or impurities in the crystal lattice.[37] These properties may be useful when hydrogen is purified by passage through hot palladium disks, but the gas's high solubility is a metallurgical problem, contributing to the embrittlement of many metals,[8] complicating the design of pipelines and storage tanks.[38]
Hydrides[edit]

Compounds of hydrogen are often called hydrides, a term that is used fairly loosely. The term "hydride" suggests that the H atom has acquired a negative or anionic character, denoted H−, and is used when hydrogen forms a compound with a more electropositive element. The existence of the hydride anion, suggested by Gilbert N. Lewis in 1916 for group 1 and 2 salt-like hydrides, was demonstrated by Moers in 1920 by the electrolysis of molten lithium hydride (LiH), producing a stoichiometric quantity of hydrogen at the anode.[39] For hydrides other than group 1 and 2 metals, the term is quite misleading, considering the low electronegativity of hydrogen. An exception in group 2 hydrides is BeH2, which is polymeric. In lithium aluminium hydride, the [AlH4]− anion carries hydridic centers firmly attached to the Al(III).
Although hydrides can be formed with almost all main-group elements, the number and combination of possible compounds varies widely; for example, more than 100 binary borane hydrides are known, but only one binary aluminium hydride.[40] Binary indium hydride has not yet been identified, although larger complexes exist.[41]
In inorganic chemistry, hydrides can also serve as bridging ligands that link two metal centres in a coordination complex. This function is particularly common in group 13 elements, especially in boranes (boron hydrides) and aluminium complexes, as well as in clustered carboranes.[42]
The most common metal hydrides for storage of Hydrogen in the clean energy industry is using Metal hydrides of the form FeTiH1.7.or LaNi5H6.[43] These hydrides store Hydrogen in the solid State.
Metal hydrides[edit]
Metal hydrides, such as MgH2, NaAlH4, LiAlH4, LiH, LaNi5H6, TiFeH21.7, TiFeH2, ammonia borane, and palladium hydride represent sources of stored hydrogen. Some are easy-to-fuel liquids at ambient temperature and pressure, whereas others are solids. These materials have very good volumetric energy density, although their specific energy is high due to the weight of the metal used. Aluminium, Magnesium and other lighter metal can also display a very high gravimetric energy density though currently the lighter hydrides are les commercial dues to the temperatures required and the system reversibility.[43]
The two commercial Metal Hydrides currently being used for storage of Hydrogen are FeTi[44] and [[LaNi, where hydrogen is stored a very safe solid of high energy density (15 times denser than lithium Batteries and 5 times denser than compressed hydrogen at 500 Bar) and low pressure; around 40 Bar compared with compressed Hydrogen up to 700Bar. These Hydrides store Hydrogen as a solid and have a density of around twice liquid hydrogen and 5 times compressed hydrogen at 500bar.[43]
Both FeTi and LaNi hydrides have been commercially used for decades and exhibit favourable temperature hysteresis curves around ambient temperature making them ideal Hydrogen storages. The cheapest of these hydrides, FeTi, when used for Hydrogen storage has a theoretically infinite lifecycle however in practice contaminants may reduce this to a finite time, say 20-30 years. These storages when coupled with electrolysers and fuel cells exhibit characteristics akin to an Advanced Very Long Life Battery.[43]
A company listed on the LSX, Dowlais, owns a division called GKN Hydrogen who have commercialised the use of FeTi Solid State Metal Hydride for the storage of Hydrogen.[43] GKN Hydrogen guarantee their commercial storages for over 20 years and have tested them to thirty year life with no degradation.[43] The FeTi powder is also 100% recyclable making it a long term sustainable storage medium for Hydrogen. FeTi has an adsorption temperature of around 20 Deg Celsius and a desorption temperature of around 60 Deg C making it ideal for storing Hydrogen. Storage pressure remain at around 40 Bar.[43]
Metal Hydrides with high gravimetric energy density, such as LiBH4 also retain a high volumetric energy density and can compete directly with Ammonia, however these have yet to be commercially developed due to a number of technical complexities including the high temperature required for Hydrogen recovery to feed to the fuel cell.[43]
LiNH2, LiBH4, and NaBH4[edit]
An alternative method for lowering dissociation temperatures is doping with activators (catalysts). This strategy has been used for aluminium hydride, but the complex synthesis makes the approach unattractive at present.[45]
Other proposed hydrides for use in a hydrogen economy include simple hydrides of magnesium[16] or transition metals and complex metal hydrides, typically containing sodium, lithium, or calcium and aluminium or boron. Hydrides chosen for storage applications require low reactivity (high safety) and high hydrogen storage densities. Leading candidates after FeTi and LaNi are lithium hydride, sodium borohydride, lithium aluminium hydride and ammonia borane. A French company McPhy Energy is developing the first industrial product, based on magnesium hydride, already sold to some major clients such as Iwatani and ENEL.[45]
Aluminium[edit]
Hydrogen can be produced using aluminium by reacting it with water.[20] It was previously believed that, to react with water, aluminium must be stripped of its natural oxide layer using caustic substances, alloys,[21] or mixing with gallium (which produces aluminium nanoparticles that allow 90% of the aluminium to react).[22] It has since been demonstrated that efficient reaction is possible by increasing the temperature and pressure of the reaction. The byproduct of the reaction to create hydrogen is aluminium oxide, which can be recycled back into aluminium with the Hall–Héroult process, making the reaction theoretically renewable. Although this requires electrolysis, which consumes a large amount of energy, the energy is then stored in the aluminium (and released when the aluminium is reacted with water).[46]
Magnesium[edit]
Mg-based hydrogen storage materials can generally fall into three categories, i.e., pure Mg, Mg-based alloys, and Mg-based composites. Particularly, more than 300 sorts of Mg-based hydrogen storage alloys have been receiving extensive attention because of the relatively better overall performance. Nonetheless, the inferior hydrogen absorption/desorption kinetics due to the thermodynamic instability of metal hydride make some of the Mg-based hydrogen storage alloys currently not appropriate for the real applications.
Some sample preparation methods, such as smelting, powder sintering, diffusion, mechanical alloying, hydriding combustion synthesis method, surface treatment, and heat treatment, etc., have been broadly employed for altering the dynamic performance and cycle life of Mg-based hydrogen storage alloys. Besides, some intrinsic modification strategies, including alloying, nanostructuring, doping by catalytic additives,[32][33] and acquiring nanocomposites with other hydrides, etc., have been mainly explored for intrinsically boosting the performance of Mg-based hydrogen storage alloys. Like aluminium, magnesium also reacts with water to produce hydrogen.[47]
Of the primary hydrogen storage alloys progressed formally, Mg and Mg-based hydrogen storage materials are believed to provide the possibility of practical application, on account of the advantages as following: 1) the resource of Mg is plentiful and economical. Mg element exists abundantly and accounts for ~2.35% of the earth's crust with the rank of the eighth; 2) low density of merely 1.74 g cm-3; 3) superior hydrogen storage capacity. The theoretical hydrogen storage amounts of the pure Mg is 7.6 wt % (weight percent), and the Mg2Ni is 3.6 wt%, respectively.[47] The main disadvantage is that Mg is highly combustible at room temperature.
Alanates-based systems[edit]
Sodium alanate (NaAlH4) is a complex hydride for H2 storage. The crystal structure was first determined through a single crystal X-ray diffracrion study in 1979. The atomic structure consisted of isolated [AlH4]− tetrahedra in which the Na atoms are surrounded by eight [AlH4]−tetrahedra in a distorted square.[47]
Hydrogen release from NaAlH4 is known since the 1950s. In 1997, Bogdanovic discovered that TiO2 doping of materials makes the process reversible at modest temperature and pressure. TiO2-doped materials are reversible in hydrogen storage, NaAlH4 is currently the state of the art reversible solid state hydrogen storage material which can be used in low temperature and has 5.6 wt.% hydrogen contained. The chemical reaction is, 3NaAlH4 ← catalyst → Na3AlH6 + 2Al + 3H2 ← catalyst → 3NaH + Al + 3/2H2. The heat required to change from NaAlH4 to Na3AlH6 is 37 kJ/mol. The heat required to change from Na3AlH6 to NaH is 47 kJ/mol. In principle, the first step of NaAlH4 releases 3.7 wt.% hydrogen at about 190 °C and the second step releases 1.8 wt.% hydrogen at about 225 °C upon heating. Further dehydrogenation of NaH occurs only at temperature higher than 400 °C. This temperature is too high for technical applications, therefore, can not be used in a fuel cell vehicle.[47]
Lithium alanate (LiAlH4) was synthesized for the first time in 1947 by dissolution of lithium hydride in an ether solution of aluminium chloride.[41] LiAlH4 has a theoretical gravimetric capacity of 10.5 wt %H2 and dehydrogenates in the following three steps: 3LiAlH4 ↔ Li3AlH6 + 3H2 + 2Al (423–448 K; 5.3 wt %H2; ∆H = −10 kJ•mol−1 H2); Li3AlH6 ↔ 3LiH + Al + 1.5H2 (453–493 K; 2.6 wt %H2; ∆H = 25 kJ•mol−1 H2); 3LiH + 3Al ↔ 3LiAl + 3/2H2 (>673 K; 2.6 wt %H2; ∆H = 140 kJ•mol−1 H2).[47]
The first two steps lead to a total amount of hydrogen released equal to 7.9 wt %, which could be attractive for practical applications, but the working temperatures and the desorption kinetics are still far from the practical targets. Several strategies have been applied in the last few years to overcome these limits, such as ball-milling and catalysts additions.[47]
Potassium Alanate (KAlH4) was first prepared by Ashby et al. by one-step synthesis in toluene, tetrahydrofuran, and diglyme. Concerning the hydrogen absorption and desorption properties, this alanate was only scarcely studied. Morioka et al.,[52] by temperature programmed desorption (TPD) analyses, proposed the following dehydrogenation mechanism: 3KAlH4 →K3AlH6 + 2Al + 3H2 (573 K, ∆H = 55 kJ•mol−1 H2; 2.9 wt %H2), K3AlH6 → 3KH + Al + 3/2H2 (613 K, ∆H = 70 kJ•mol−1 H2; 1.4 wt %H2), 3KH → 3K + 3/2H2 (703 K, 1.4 wt %H2). These reactions were demonstrated reversible without catalysts addition at relatively low hydrogen pressure and temperatures. The addition of TiCl3 was found to decrease the working temperature of the first dehydrogenation step of 50 K, but no variations were recorded for the last two reaction steps.[47]
Protons and acids[edit]
Oxidation of hydrogen removes its electron and gives H+, which contains no electrons and a nucleus which is usually composed of one proton. That is why H+ is often called a proton. This species is central to discussion of acids. Under the Brønsted–Lowry acid–base theory, acids are proton donors, while bases are proton acceptors.
A bare proton, H+, cannot exist in solution or in ionic crystals because of its unstoppable attraction to other atoms or molecules with electrons. Except at the high temperatures associated with plasmas, such protons cannot be removed from the electron clouds of atoms and molecules, and will remain attached to them. However, the term 'proton' is sometimes used loosely and metaphorically to refer to positively charged or cationic hydrogen attached to other species in this fashion, and as such is denoted "H+" without any implication that any single protons exist freely as a species.
To avoid the implication of the naked "solvated proton" in solution, acidic aqueous solutions are sometimes considered to contain a less unlikely fictitious species, termed the "hydronium ion" ([H3O]+). However, even in this case, such solvated hydrogen cations are more realistically conceived as being organized into clusters that form species closer to [H9O4]+.[48] Other oxonium ions are found when water is in acidic solution with other solvents.[49]
Although exotic on Earth, one of the most common ions in the universe is the H+3 ion, known as protonated molecular hydrogen or the trihydrogen cation.[50]
Isotopes[edit]
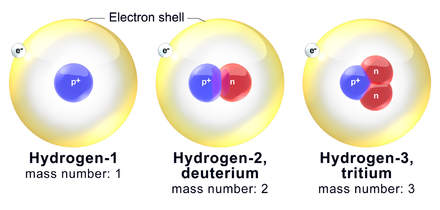


Hydrogen has three naturally occurring isotopes, denoted 1
H, 2
H and 3
H. Other, highly unstable nuclei (4
H to 7
H) have been synthesized in the laboratory but not observed in nature.[51][52]
- 1
H is the most common hydrogen isotope, with an abundance of more than 99.98%. Because the nucleus of this isotope consists of only a single proton, it is given the descriptive but rarely used formal name protium.[53] It is unique among all stable isotopes in having no neutrons; see diproton for a discussion of why others do not exist. - 2
H, the other stable hydrogen isotope, is known as deuterium and contains one proton and one neutron in the nucleus. Deuterium is not radioactive, and does not represent a significant toxicity hazard. Water enriched in molecules that include deuterium instead of normal hydrogen is called heavy water. Deuterium and its compounds are used as a non-radioactive label in chemical experiments and in solvents for 1
H-NMR spectroscopy.[54] Heavy water is used as a neutron moderator and coolant for nuclear reactors. Deuterium is also a potential fuel for commercial nuclear fusion.[55] - 3
H is known as tritium and contains one proton and two neutrons in its nucleus. It is radioactive, decaying into helium-3 through beta decay with a half-life of 12.32 years.[42] It is radioactive enough to be used in luminous paint to enhance the visibility of data displays, such as for painting the hands and dial-markers of watches. The watch glass prevents the small amount of radiation involved from escaping the case.[56] Small amounts of tritium are produced naturally by the interaction of cosmic rays with atmospheric gases; tritium has also been released during nuclear weapons tests.[57] It is used in nuclear fusion reactions,[58] as a tracer in isotope geochemistry,[59] and in specialized self-powered lighting devices.[60] Tritium has also been used in chemical and biological labelling experiments as a radiolabel.[61]
Unique among the elements, distinct names are assigned to its isotopes in common use today. During the early study of radioactivity, various heavy radioactive isotopes were given their own names, but such names are no longer used, except for deuterium and tritium. The symbols D and T (instead of 2
H and 3
H) are sometimes used for deuterium and tritium, but the symbol P is already in use for phosphorus and thus is not available for protium.[62] In its nomenclatural guidelines, the International Union of Pure and Applied Chemistry (IUPAC) allows any of D, T, 2
H, and 3
H to be used, although 2
H and 3
H are preferred.[63]
The exotic atom muonium (symbol Mu), composed of an antimuon and an electron, can also be considered a light radioisotope of hydrogen.[64] Because muons decay with lifetime 2.2 µs, muonium is too unstable to exhibit observable chemistry.[65] Nevertheless, muonium compounds are important test cases for quantum simulation, due to the mass difference between the antimuon and the proton,[66] and IUPAC nomenclature incorporates such hypothetical compounds as muonium chloride (MuCl) and sodium muonide (NaMu), analogous to hydrogen chloride and sodium hydride respectively.[67]
Thermal and physical properties[edit]
Table of thermal and physical properties of hydrogen (H2) at atmospheric pressure:[68][69]
| Temperature (K) | Density (kg/m^3) | Specific heat (kJ/kg °C) | Dynamic viscosity (kg/m s) | Kinematic viscosity (m^2/s) | Thermal conductivity (W/m °C) | Thermal diffusivity (m^2/s) | Prandtl Number |
| 100 | 0.24255 | 11.23 | 4.21E-06 | 1.74E-05 | 6.70E-02 | 2.46E-05 | 0.707 |
| 150 | 0.16371 | 12.602 | 5.60E-06 | 3.42E-05 | 0.0981 | 4.75E-05 | 0.718 |
| 200 | 0.1227 | 13.54 | 6.81E-06 | 5.55E-05 | 0.1282 | 7.72E-05 | 0.719 |
| 250 | 0.09819 | 14.059 | 7.92E-06 | 8.06E-05 | 0.1561 | 1.13E-04 | 0.713 |
| 300 | 0.08185 | 14.314 | 8.96E-06 | 1.10E-04 | 0.182 | 1.55E-04 | 0.706 |
| 350 | 0.07016 | 14.436 | 9.95E-06 | 1.42E-04 | 0.206 | 2.03E-04 | 0.697 |
| 400 | 0.06135 | 14.491 | 1.09E-05 | 1.77E-04 | 0.228 | 2.57E-04 | 0.69 |
| 450 | 0.05462 | 14.499 | 1.18E-05 | 2.16E-04 | 0.251 | 3.16E-04 | 0.682 |
| 500 | 0.04918 | 14.507 | 1.26E-05 | 2.57E-04 | 0.272 | 3.82E-04 | 0.675 |
| 550 | 0.04469 | 14.532 | 1.35E-05 | 3.02E-04 | 0.292 | 4.52E-04 | 0.668 |
| 600 | 0.04085 | 14.537 | 1.43E-05 | 3.50E-04 | 0.315 | 5.31E-04 | 0.664 |
| 700 | 0.03492 | 14.574 | 1.59E-05 | 4.55E-04 | 0.351 | 6.90E-04 | 0.659 |
| 800 | 0.0306 | 14.675 | 1.74E-05 | 5.69E-04 | 0.384 | 8.56E-04 | 0.664 |
| 900 | 0.02723 | 14.821 | 1.88E-05 | 6.90E-04 | 0.412 | 1.02E-03 | 0.676 |
| 1000 | 0.02424 | 14.99 | 2.01E-05 | 8.30E-04 | 0.448 | 1.23E-03 | 0.673 |
| 1100 | 0.02204 | 15.17 | 2.13E-05 | 9.66E-04 | 0.488 | 1.46E-03 | 0.662 |
| 1200 | 0.0202 | 15.37 | 2.26E-05 | 1.12E-03 | 0.528 | 1.70E-03 | 0.659 |
| 1300 | 0.01865 | 15.59 | 2.39E-05 | 1.28E-03 | 0.568 | 1.96E-03 | 0.655 |
| 1400 | 0.01732 | 15.81 | 2.51E-05 | 1.45E-03 | 0.61 | 2.23E-03 | 0.65 |
| 1500 | 0.01616 | 16.02 | 2.63E-05 | 1.63E-03 | 0.655 | 2.53E-03 | 0.643 |
| 1600 | 0.0152 | 16.28 | 2.74E-05 | 1.80E-03 | 0.697 | 2.82E-03 | 0.639 |
| 1700 | 0.0143 | 16.58 | 2.85E-05 | 1.99E-03 | 0.742 | 3.13E-03 | 0.637 |
| 1800 | 0.0135 | 16.96 | 2.96E-05 | 2.19E-03 | 0.786 | 3.44E-03 | 0.639 |
| 1900 | 0.0128 | 17.49 | 3.07E-05 | 2.40E-03 | 0.835 | 3.73E-03 | 0.643 |
| 2000 | 0.0121 | 18.25 | 3.18E-05 | 2.63E-03 | 0.878 | 3.98E-03 | 0.661 |
History[edit]
Discovery and use[edit]
Robert Boyle[edit]

In 1671, the Irish scientist Robert Boyle discovered and described the reaction between iron filings and dilute acids, which results in the production of hydrogen gas.[70][71]
Having provided a saline spirit [hydrochloric acid], which by an uncommon way of preparation was made exceeding sharp and piercing, we put into a vial, capable of containing three or four ounces of water, a convenient quantity of filings of steel, which were not such as are commonly sold in shops to Chemists and Apothecaries, (those being usually not free enough from rust) but such as I had a while before caus'd to be purposely fil'd off from a piece of good steel. This metalline powder being moistn'd in the viol with a little of the menstruum, was afterwards drench'd with more; whereupon the mixture grew very hot, and belch'd up copious and stinking fumes; which whether they consisted altogether of the volatile sulfur of the Mars [iron?], or of metalline steams participating of a sulfureous nature, and join'd with the saline exhalations of the menstruum, is not necessary to be here discuss'd. But whencesoever this stinking smoak proceeded, so inflammable it was, that upon the approach of a lighted candle to it, it would readily enough take fire, and burn with a blewish and somewhat greenish flame at the mouth of the viol for a good while together; and that, though with little light, yet with more strength than one would easily suspect.
— Robert Boyle, Tracts written by the Honourable Robert Boyle containing new experiments, touching the relation betwixt flame and air...
The word "sulfureous" may be somewhat confusing, especially since Boyle did a similar experiment with iron and sulfuric acid.[72] However, in all likelihood, "sulfureous" should here be understood to mean combustible.[73]
Henry Cavendish[edit]
In 1766, Henry Cavendish was the first to recognize hydrogen gas as a discrete substance, by naming the gas from a metal-acid reaction "inflammable air". He speculated that "inflammable air" was in fact identical to the hypothetical substance called "phlogiston"[74][75] and further finding in 1781 that the gas produces water when burned. He is usually given credit for the discovery of hydrogen as an element.[76][77]
Antoine Lavoisier[edit]

In 1783, Antoine Lavoisier identified the element that came to be known as hydrogen (from the Greek ὑδρο- hydro meaning "water" and -γενής genes meaning "former")[78] when he and Laplace reproduced Cavendish's finding that water is produced when hydrogen is burned.[77] Lavoisier produced hydrogen for his experiments on mass conservation by reacting a flux of steam with metallic iron through an incandescent iron tube heated in a fire. Anaerobic oxidation of iron by the protons of water at high temperature can be schematically represented by the set of following reactions:
- 1) Fe + H2O → FeO + H2
- 2) Fe + 3 H2O → Fe2O3 + 3 H2
- 3) Fe + 4 H2O → Fe3O4 + 4 H2
Many metals such as zirconium undergo a similar reaction with water leading to the production of hydrogen.
19th century[edit]
François Isaac de Rivaz built the first de Rivaz engine, an internal combustion engine powered by a mixture of hydrogen and oxygen in 1806. Edward Daniel Clarke invented the hydrogen gas blowpipe in 1819. The Döbereiner's lamp and limelight were invented in 1823.[77]
Hydrogen was liquefied for the first time by James Dewar in 1898 by using regenerative cooling and his invention, the vacuum flask.[77] He produced solid hydrogen the next year.[77]
Hydrogen-lifted airship[edit]
The first hydrogen-filled balloon was invented by Jacques Charles in 1783.[77] Hydrogen provided the lift for the first reliable form of air-travel following the 1852 invention of the first hydrogen-lifted airship by Henri Giffard.[77] German count Ferdinand von Zeppelin promoted the idea of rigid airships lifted by hydrogen that later were called Zeppelins; the first of which had its maiden flight in 1900.[77] Regularly scheduled flights started in 1910 and by the outbreak of World War I in August 1914, they had carried 35,000 passengers without a serious incident. Hydrogen-lifted airships were used as observation platforms and bombers during the war.

The first non-stop transatlantic crossing was made by the British airship R34 in 1919. Regular passenger service resumed in the 1920s and the discovery of helium reserves in the United States promised increased safety, but the U.S. government refused to sell the gas for this purpose. Therefore, H2 was used in the Hindenburg airship, which was destroyed in a midair fire over New Jersey on 6 May 1937.[77] The incident was broadcast live on radio and filmed. Ignition of leaking hydrogen is widely assumed to be the cause, but later investigations pointed to the ignition of the aluminized fabric coating by static electricity. But the damage to hydrogen's reputation as a lifting gas was already done and commercial hydrogen airship travel ceased. Hydrogen is still used, in preference to non-flammable but more expensive helium, as a lifting gas for weather balloons.
Deuterium and tritium[edit]
Deuterium was discovered in December 1931 by Harold Urey, and tritium was prepared in 1934 by Ernest Rutherford, Mark Oliphant, and Paul Harteck.[76] Heavy water, which consists of deuterium in the place of regular hydrogen, was discovered by Urey's group in 1932.[77]
Hydrogen-cooled turbogenerator[edit]
The first hydrogen-cooled turbogenerator went into service with gaseous hydrogen as a coolant in the rotor and the stator in 1937 at Dayton, Ohio, by the Dayton Power & Light Co.;[79] because of the thermal conductivity and very low viscosity of hydrogen gas, thus lower drag than air, this is the most common type in its field today for large generators (typically 60 MW and bigger; smaller generators are usually air-cooled).
Nickel–hydrogen battery[edit]
The nickel–hydrogen battery was used for the first time in 1977 aboard the U.S. Navy's Navigation technology satellite-2 (NTS-2).[80] The International Space Station,[81] Mars Odyssey[82] and the Mars Global Surveyor[83] are equipped with nickel-hydrogen batteries. In the dark part of its orbit, the Hubble Space Telescope is also powered by nickel-hydrogen batteries, which were finally replaced in May 2009,[84] more than 19 years after launch and 13 years beyond their design life.[85]
Role in quantum theory[edit]

Because of its simple atomic structure, consisting only of a proton and an electron, the hydrogen atom, together with the spectrum of light produced from it or absorbed by it, has been central to the development of the theory of atomic structure.[86] Furthermore, study of the corresponding simplicity of the hydrogen molecule and the corresponding cation H+2 brought understanding of the nature of the chemical bond, which followed shortly after the quantum mechanical treatment of the hydrogen atom had been developed in the mid-1920s.
One of the first quantum effects to be explicitly noticed (but not understood at the time) was a Maxwell observation involving hydrogen, half a century before full quantum mechanical theory arrived. Maxwell observed that the specific heat capacity of H2 unaccountably departs from that of a diatomic gas below room temperature and begins to increasingly resemble that of a monatomic gas at cryogenic temperatures. According to quantum theory, this behavior arises from the spacing of the (quantized) rotational energy levels, which are particularly wide-spaced in H2 because of its low mass. These widely spaced levels inhibit equal partition of heat energy into rotational motion in hydrogen at low temperatures. Diatomic gases composed of heavier atoms do not have such widely spaced levels and do not exhibit the same effect.[87]
Antihydrogen (
H
) is the antimatter counterpart to hydrogen. It consists of an antiproton with a positron. Antihydrogen is the only type of antimatter atom to have been produced as of 2015[update].[88][89]
Cosmic prevalence and distribution[edit]

Hydrogen, as atomic H, is the most abundant chemical element in the universe, making up 75 percent of normal matter by mass and more than 90 percent by number of atoms. (Most of the mass of the universe, however, is not in the form of chemical-element type matter, but rather is postulated to occur as yet-undetected forms of mass such as dark matter and dark energy.[90])
Hydrogen is found in great abundance in stars and gas giant planets. Molecular clouds of H2 are associated with star formation. Hydrogen plays a vital role in powering stars through the proton-proton reaction in case of stars with very low to approximately 1 mass of the Sun and the CNO cycle of nuclear fusion in case of stars more massive than the Sun.[91]
States[edit]
Throughout the universe, hydrogen is mostly found in the atomic and plasma states, with properties quite distinct from those of molecular hydrogen. As a plasma, hydrogen's electron and proton are not bound together, resulting in very high electrical conductivity and high emissivity (producing the light from the Sun and other stars). The charged particles are highly influenced by magnetic and electric fields. For example, in the solar wind they interact with the Earth's magnetosphere giving rise to Birkeland currents and the aurora.
Hydrogen is found in the neutral atomic state in the interstellar medium because the atoms seldom collide and combine. They are the source of the 21-cm hydrogen line at 1420 MHz that is detected in order to probe primordial hydrogen.[92] The large amount of neutral hydrogen found in the damped Lyman-alpha systems is thought to dominate the cosmological baryonic density of the universe up to a redshift of z = 4.[93]
Under ordinary conditions on Earth, elemental hydrogen exists as the diatomic gas, H2. Hydrogen gas is very rare in the Earth's atmosphere (around 0.53 ppm on a molar basis[94]) because of its light weight, which enables it to escape from the atmosphere more rapidly than heavier gases. However, hydrogen is the third most abundant element on the Earth's surface,[95] mostly in the form of chemical compounds such as hydrocarbons and water.[42]
A molecular form called protonated molecular hydrogen (H+3) is found in the interstellar medium, where it is generated by ionization of molecular hydrogen from cosmic rays. This ion has also been observed in the upper atmosphere of the planet Jupiter. The ion is relatively stable in the environment of outer space due to the low temperature and density. H+3 is one of the most abundant ions in the universe, and it plays a notable role in the chemistry of the interstellar medium.[96] Neutral triatomic hydrogen H3 can exist only in an excited form and is unstable.[97] By contrast, the positive hydrogen molecular ion (H+2) is a rare molecule in the universe.
Production[edit]
Many methods exist for producing H2, but three dominate commercially: steam reforming often coupled to water-gas shift, partial oxidation of hydrocarbons, and water electrolysis.[98]
Steam reforming[edit]
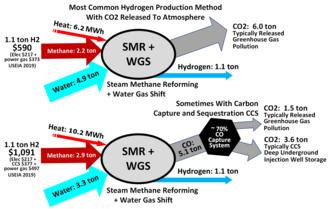
Hydrogen is mainly produced by steam reforming, the reaction of water and methane.[99][100] [101] Thus, at high temperatures (1000–1400 K, 700–1100 °C or 1300–2000 °F), steam (water vapor) reacts with methane to yield carbon monoxide and H2.
- CH4 + H2O → CO + 3 H2
Steam reforming is also used for the industrial preparation of ammonia.
This reaction is favored at low pressures, Nonetheless conducted at high pressures (2.0 MPa, 20 atm or 600 inHg) because high-pressure H2 is the most marketable product, and pressure swing adsorption (PSA) purification systems work better at higher pressures. The product mixture is known as "synthesis gas" because it is often used directly for the production of methanol and many other compounds. Hydrocarbons other than methane can be used to produce synthesis gas with varying product ratios. One of the many complications to this highly optimized technology is the formation of coke or carbon:
- CH4 → C + 2 H2
Consequently, steam reforming typically employs an excess of H2O. Additional hydrogen can be recovered from the steam by use of carbon monoxide through the water gas shift reaction. This process requires an iron oxide catalyst:[101]
- CO + H2O → CO2 + H2
Hydrogen is sometimes produced and consumed in the same industrial process, without being separated. In the Haber process for the production of ammonia, hydrogen is generated from natural gas.[102]
Partial oxidation of hydrocarbons[edit]
Other methods for CO and H2 production include partial oxidation of hydrocarbons:[103]
- 2 CH4 + O2 → 2 CO + 4 H2
Although less important commercially, coal can serve as a prelude to the shift reaction above:[101]
- C + H2O → CO + H2
Olefin production units may produce substantial quantities of byproduct hydrogen particularly from cracking light feedstocks like ethane or propane.[104]
Water electrolysis[edit]

This method is currently the most common approach to producing hydrogen and commercial electrolysers are being installed in many places throughout the world. Electrolysers can be operated from renewable energy such as solar, wind or hydro and when coupled with a fuel cell are a key component of the Advanced Solid State Hydrogen Battery[44].
The electrolysis of water is a conceptually simple method of producing hydrogen.
- 2 H2O(l) → 2 H2(g) + O2(g)
Commercial electrolyzers use nickel-based catalysts in strongly alkaline solution. Platinum is a superior catalyst but is expensive.[105]
Electrolysis of brine to yield chlorine also produces hydrogen as a co-product.[106]
Methane pyrolysis[edit]
Hydrogen can be produced by pyrolysis of natural gas (methane).
This route has a lower carbon footprint than commercial hydrogen production processes.[107][108][109][110] Developing a commercial methane pyrolysis process could expedite the expanded use of hydrogen in industrial and transportation applications. Methane pyrolysis is accomplished by passing methane through a molten metal catalyst containing dissolved nickel. Methane is converted to hydrogen gas and solid carbon.[111][112]
- CH4(g) → C(s) + 2 H2(g) (ΔH° = 74 kJ/mol)
The carbon may be sold as a manufacturing feedstock or fuel, or landfilled.
Further research continues in several laboratories, including at Karlsruhe Liquid-metal Laboratory [113] and at University of California – Santa Barbara.[114] BASF built a methane pyrolysis pilot plant.[115]
Thermochemical[edit]
More than 200 thermochemical cycles can be used for water splitting. Many of these cycles such as the iron oxide cycle, cerium(IV) oxide–cerium(III) oxide cycle, zinc zinc-oxide cycle, sulfur-iodine cycle, copper-chlorine cycle and hybrid sulfur cycle have been evaluated for their commercial potential to produce hydrogen and oxygen from water and heat without using electricity.[116] A number of laboratories (including in France, Germany, Greece, Japan, and the United States) are developing thermochemical methods to produce hydrogen from solar energy and water.[117]
Laboratory methods[edit]
H2 is produced in laboratories, often as a by-product of other reactions. Many metals react with water to produce H2, but the rate of hydrogen evolution depends on the metal, the pH, and the presence of alloying agents. Most commonly, hydrogen evolution is induced by acids. The alkali and alkaline earth metals, aluminium, zinc, manganese, and iron react readily with aqueous acids. This reaction is the basis of the Kipp's apparatus, which once was used as a laboratory gas source:
- Zn + 2 H+ → Zn2+ + H2
In the absence of acid, the evolution of H2 is slower. Because iron is widely used structural material, its anaerobic corrosion is of technological significance:
- Fe + 2 H2O → Fe(OH)2 + H2
Many metals, such as aluminium, are slow to react with water because they form passivated coatings of oxides. An alloy of aluminium and gallium, however, does react with water.[118] At high pH, aluminium can produce H2:
- 2 Al + 6 H2O + 2 OH− → 2 [Al(OH)4]− + 3 H2
Some metal-containing compounds react with acids to evolve H2. Under anaerobic conditions, ferrous hydroxide (Fe(OH)
2) can be oxidized by the protons of water to form magnetite and H2. This process is described by the Schikorr reaction:
- 3 Fe(OH)2 → Fe3O4 + 2 H2O + H2
This process occurs during the anaerobic corrosion of iron and steel in oxygen-free groundwater and in reducing soils below the water table.
Biohydrogen[edit]
H2 is produced by hydrogenase enzymes in some fermentation.[119]
Applications[edit]
Petrochemical industry[edit]
Large quantities of H2 are used in the "upgrading" of fossil fuels. Key consumers of H2 include hydrodealkylation, hydrodesulfurization, and hydrocracking. Many of these reactions can be classified as hydrogenolysis, i.e., the cleavage of bonds by hydrogen. Illustrative is the separation of sulfur from liquid fossil fuels:[98]
- R2S + 2 H2 → H2S + 2 RH
Hydrogenation[edit]
Hydrogenation, the addition of H2 to various substrates is conducted on a large scale. The hydrogenation of N2 to produce ammonia by the Haber–Bosch process consumes a few percent of the energy budget in the entire industry. The resulting ammonia is used to supply the majority of the protein consumed by humans.[120] Hydrogenation is used to convert unsaturated fats and oils to saturated (trans) fats and oils. The major application is the production of margarine. Methanol is produced by hydrogenation of carbon dioxide. It is similarly the source of hydrogen in the manufacture of hydrochloric acid. H2 is also used as a reducing agent for the conversion of some ores to the metals.[121]
Coolant[edit]
Hydrogen is commonly used in power stations as a coolant in generators due to a number of favorable properties that are a direct result of its light diatomic molecules. These include low density, low viscosity, and the highest specific heat and thermal conductivity of all gases.
Energy carrier[edit]
Elemental hydrogen is widely discussed in the context of energy, as an energy carrier with potential to help the decarbonisation of economies to mitigate greenhouse gas emissions.[122][123] This therefore requires hydrogen to be produced cleanly, in quantities to supply in sectors and applications where cheaper and more energy efficient alternatives are limited. These include microgrids, standalone power supplies, heavy industry and long-distance transport.[122] Hydrogen is a ''carrier'' of energy rather than an energy resource, because there is minimal naturally occurring source of hydrogen in useful quantities.[124]
Hydrogen can be deployed as an energy source in fuel cells to produce electricity, or via combustion to generate heat by either gas engines and turbines or as process heat in industrial process industry.[7] When hydrogen is consumed in fuel cells, the only emission is water vapour.[7] The overall lifecycle emissions of hydrogen depend on how it is produced. The most common method is steam methane reforming, in which hydrogen is produced from a chemical reaction between steam and methane, the main component of natural gas. Producing one tonne of hydrogen through this process emits 6.6–9.3 tonnes of carbon dioxide.[125] While carbon capture and storage (CCS) could remove a large fraction of these emissions, the overall carbon footprint of hydrogen from natural gas is difficult to assess as of 2021[update], in part because of emissions (including vented and fugitive methane) created in the production of the natural gas itself.[126]
Electricity can be used to split water molecules, producing sustainable hydrogen provided the electricity is generated sustainably. Hydrogen can be produced when there is a surplus of renewable electricity, then stored and used to generate heat or to re-generate electricity.[127] Hydrogen created through electrolysis using renewable energy is commonly referred to as "green hydrogen."[128] It can be further stored in metal hydride and reused through a fuel cell[44], used directly in Hydrogen combustion engines or transformed into synthetic fuels such as ammonia and methanol.[129]
Innovation in hydrogen electrolysers now make large-scale production of hydrogen from electricity more cost-competitive.[130] There is potential for hydrogen produced this way to play a significant role in decarbonising energy systems where there are challenges and limitations to replacing fossil fuels with direct use of electricity.[122]
Hydrogen Energy Storage Systems (HESS)[edit]
The most advanced form of HESS today is the use of Electrolysers coupled to Solid State Hydrogen storage in Metal Hydride (FeTi) and then being converted to electricity for consumption via Fuel Cells or Hydrogen combustion engines (Hydrogen Generators). The HESS is becoming a competitor to other BESS technologies due to its long life, (up to 30 years), 100% recyclability, ability to retain its capacity without degradation (unlike Lithium Batteries), its much broader operating temperature capability and its ability to provide near 100% cycling of the battery without impacting the battery life. The latter allows the entire capacity of the HESS to be available at all times and for up to 30 years.[46]
These Advanced Solid State Batteries using Hydrogen Carrier[44]
are commercially available and being economically deployed in microgrids
[131], Stand-Alone Power Supplies[132], and e-car charging[133], diesel generator substitution[134] and direct storage of hydrogen for industrial processes.Cite error: A <ref> tag is missing the closing </ref> (see the help page). However, it is likely to play a larger role in providing industrial feedstock for cleaner production of ammonia and organic chemicals.[122] For example, in steelmaking, hydrogen could function as a clean energy carrier and also as a low-carbon catalyst replacing coal-derived coke.[135] Hydrogen used to decarbonise transportation is likely to find its largest applications in shipping, aviation and to a lesser extent heavy goods vehicles, through the use of hydrogen-derived synthetic fuels such as ammonia and methanol, and fuel cell technology.[122] For light duty vehicles including passenger cars, hydrogen is far behind other alternative fuel vehicles, especially compared with the rate of adoption of battery electric vehicles, and may not play a significant role in future.[136]
Semiconductor industry[edit]
Hydrogen is employed to saturate broken ("dangling") bonds of amorphous silicon and amorphous carbon that helps stabilizing material properties.[137] It is also a potential electron donor in various oxide materials, including ZnO,[138][139] SnO2, CdO, MgO,[140] ZrO2, HfO2, La2O3, Y2O3, TiO2, SrTiO3, LaAlO3, SiO2, Al2O3, ZrSiO4, HfSiO4, and SrZrO3.[141]
Niche and evolving uses[edit]
- Shielding gas: Hydrogen is used as a shielding gas in welding methods such as atomic hydrogen welding.[142][143]
- Cryogenic research: Liquid H2 is used in cryogenic research, including superconductivity studies.[144]
- Buoyant lifting: Because H2 is lighter than air, having only 7% of the density of air, it was once widely used as a lifting gas in balloons and airships.[145]
- Leak detection: Pure or mixed with nitrogen (sometimes called forming gas), hydrogen is a tracer gas for detection of minute leaks. Applications can be found in the automotive, chemical, power generation, aerospace, and telecommunications industries.[146] Hydrogen is an authorized food additive (E 949) that allows food package leak testing, as well as having anti-oxidizing properties.[147]
- Neutron moderation: Deuterium (hydrogen-2) is used in nuclear fission applications as a moderator to slow neutrons.
- Nuclear fusion fuel: Deuterium is used in nuclear fusion reactions.[77]
- Isotopic labeling: Deuterium compounds have applications in chemistry and biology in studies of isotope effects on reaction rates.[148]
- Rocket propellant: Liquid hydrogen and liquid oxygen together serve as cryogenic fuel in liquid-propellant rockets, as in the Space Shuttle main engines. NASA has investigated the use of rocket propellant made from atomic hydrogen, boron or carbon that is frozen into solid molecular hydrogen particles that are suspended in liquid helium. Upon warming, the mixture vaporizes to allow the atomic species to recombine, heating the mixture to high temperature.[149]
- Tritium uses: Tritium (hydrogen-3), produced in nuclear reactors, is used in the production of hydrogen bombs,[150] as an isotopic label in the biosciences,[61] and as a source of beta radiation in radioluminescent paint for instrument dials and emergency signage.[56]
Biological reactions[edit]
H2 is a product of some types of anaerobic metabolism and is produced by several microorganisms, usually via reactions catalyzed by iron- or nickel-containing enzymes called hydrogenases. These enzymes catalyze the reversible redox reaction between H2 and its component two protons and two electrons. Creation of hydrogen gas occurs in the transfer of reducing equivalents, produced during pyruvate fermentation, to water.[151] The natural cycle of hydrogen production and consumption by organisms is called the hydrogen cycle.[152] Bacteria such as Mycobacterium smegmatis can utilize the small amount of hydrogen in the atmosphere as a source of energy when other sources are lacking, using a hydrogenase with small channels that exclude oxygen and so permits the reaction to occur even though the hydrogen concentration is very low and the oxygen concentration is as in normal air.[94][153]
Hydrogen is the most abundant element in the human body in terms of numbers of atoms of the element but the third most abundant element by mass. H2 occurs in the breath of humans due to the metabolic activity of hydrogenase-containing microorganisms in the large intestine and is a natural component of flatus. The concentration in the breath of fasting people at rest is typically less than 5 parts per million (ppm) but can be 50 ppm when people with intestinal disorders consume molecules they cannot absorb during diagnostic hydrogen breath tests.[154]
Water splitting, in which water is decomposed into its component protons, electrons, and oxygen, occurs in the light reactions in all photosynthetic organisms. Some such organisms, including the alga Chlamydomonas reinhardtii and cyanobacteria, have evolved a second step in the dark reactions in which protons and electrons are reduced to form H2 gas by specialized hydrogenases in the chloroplast.[155] Efforts have been undertaken to genetically modify cyanobacterial hydrogenases to efficiently synthesize H2 gas even in the presence of oxygen.[156] Efforts have also been undertaken with genetically modified alga in a bioreactor.[157]
Safety and precautions[edit]
| Hazards | |
|---|---|
| GHS labelling: | |

| |
| Danger | |
| H220 | |
| P202, P210, P271, P377, P381, P403[158] | |
| NFPA 704 (fire diamond) | |
Hydrogen poses a number of hazards to human safety, from potential detonations and fires when mixed with air to being an asphyxiant in its pure, oxygen-free form.[159] In addition, liquid hydrogen is a cryogen and presents dangers (such as frostbite) associated with very cold liquids.[160] Hydrogen dissolves in many metals and in addition to leaking out, may have adverse effects on them, such as hydrogen embrittlement,[161] leading to cracks and explosions.[162] Hydrogen gas leaking into external air may spontaneously ignite. Moreover, hydrogen fire, while being extremely hot, is almost invisible, and thus can lead to accidental burns.[163]
Even interpreting the hydrogen data (including safety data) is confounded by a number of phenomena. Many physical and chemical properties of hydrogen depend on the parahydrogen/orthohydrogen ratio (it often takes days or weeks at a given temperature to reach the equilibrium ratio, for which the data is usually given). Hydrogen detonation parameters, such as critical detonation pressure and temperature, strongly depend on the container geometry.[159]
See also[edit]
- Combined cycle hydrogen power plant
- Hydrogen economy – Using hydrogen to decarbonize sectors which are hard to electrify
- Hydrogen production – Industrial production of molecular hydrogen
- Hydrogen safety – Procedures for safe production, handling and use of hydrogen
- Hydrogen technologies – Technologies that relating to the production & use of hydrogen
- Hydrogen transport
- Liquid hydrogen – Liquid state of the element hydrogen
- Methane pyrolysis – Thermal decomposition of materials (for hydrogen)
- Natural hydrogen – Molecular hydrogen naturally occurring on Earth
- Pyrolysis – Thermal decomposition of materials
Notes[edit]
- ^ However, most of the universe's mass is not in the form of baryons or chemical elements. See dark matter and dark energy.
- ^ 286 kJ/mol: energy per mole of the combustible material (molecular hydrogen).
References[edit]
- ^ "Hydrogen". Encyclopædia Britannica. Archived from the original on 24 December 2021. Retrieved 25 December 2021.
- ^ Boyd, Padi (19 July 2014). "What is the chemical composition of stars?". NASA. Archived from the original on 15 January 2015. Retrieved 5 February 2008.
- ^ Laursen, S.; Chang, J.; Medlin, W.; Gürmen, N.; Fogler, H. S. (27 July 2004). "An extremely brief introduction to computational quantum chemistry". Molecular Modeling in Chemical Engineering. University of Michigan. Archived from the original on 20 May 2015. Retrieved 4 May 2015.
- ^ Presenter: Professor Jim Al-Khalili (21 January 2010). "Discovering the Elements". Chemistry: A Volatile History. 25:40 minutes in. BBC. BBC Four. Archived from the original on 25 January 2010. Retrieved 9 February 2010.
- ^ Dincer, Ibrahim; Acar, Canan (14 September 2015). "Review and evaluation of hydrogen production methods for better sustainability". International Journal of Hydrogen Energy. 40 (34): 11094–11111. doi:10.1016/j.ijhydene.2014.12.035. ISSN 0360-3199. Archived from the original on 15 February 2022. Retrieved 4 February 2022.
- ^ "Hydrogen Basics – Production". Florida Solar Energy Center. 2007. Archived from the original on 18 February 2008. Retrieved 5 February 2008.
- ^ a b c d e Lewis, Alastair C. (10 June 2021). "Optimising air quality co-benefits in a hydrogen economy: a case for hydrogen-specific standards for NO x emissions". Environmental Science: Atmospheres. 1 (5): 201–207. doi:10.1039/D1EA00037C. S2CID 236732702.
 This article incorporates text from this source, which is available under the CC BY 3.0 license.
This article incorporates text from this source, which is available under the CC BY 3.0 license.
- ^ a b Rogers, H. C. (1999). "Hydrogen Embrittlement of Metals". Science. 159 (3819): 1057–1064. Bibcode:1968Sci...159.1057R. doi:10.1126/science.159.3819.1057. PMID 17775040. S2CID 19429952.
- ^ "Dihydrogen". O=CHem Directory. University of Southern Maine. Archived from the original on 13 February 2009. Retrieved 6 April 2009.
- ^ Committee on Alternatives and Strategies for Future Hydrogen Production and Use (2004). The Hydrogen Economy: Opportunities, Costs, Barriers, and R&D Needs. National Academies Press. p. 240. ISBN 978-0-309-09163-3. Archived from the original on 29 January 2021. Retrieved 3 September 2020.
- ^ Carcassi, M. N.; Fineschi, F. (2005). "Deflagrations of H2–air and CH4–air lean mixtures in a vented multi-compartment environment". Energy. 30 (8): 1439–1451. doi:10.1016/j.energy.2004.02.012.
- ^ Patnaik, P. (2007). A Comprehensive Guide to the Hazardous Properties of Chemical Substances. Wiley-Interscience. p. 402. ISBN 978-0-471-71458-3. Archived from the original on 26 January 2021. Retrieved 3 September 2020.
- ^ Schefer, E. W.; Kulatilaka, W. D.; Patterson, B. D.; Settersten, T. B. (June 2009). "Visible emission of hydrogen flames". Combustion and Flame. 156 (6): 1234–1241. doi:10.1016/j.combustflame.2009.01.011. Archived from the original on 29 January 2021. Retrieved 30 June 2019.
- ^ "Myths about the Hindenburg Crash". Airships.net. Archived from the original on 20 April 2021. Retrieved 29 March 2021.
- ^ Lide, David R., ed. (2006). CRC Handbook of Chemistry and Physics (87th ed.). Boca Raton, FL: CRC Press. ISBN 0-8493-0487-3.
- ^ Clayton, D. D. (2003). Handbook of Isotopes in the Cosmos: Hydrogen to Gallium. Cambridge University Press. ISBN 978-0-521-82381-4.
- ^ NAAP Labs (2009). "Energy Levels". University of Nebraska Lincoln. Archived from the original on 11 May 2015. Retrieved 20 May 2015.
- ^ "photon wavelength 13.6 eV". Wolfram Alpha. 20 May 2015. Archived from the original on 12 May 2016. Retrieved 20 May 2015.
- ^ Stern, D. P. (16 May 2005). "The Atomic Nucleus and Bohr's Early Model of the Atom". NASA Goddard Space Flight Center (mirror). Archived from the original on 17 October 2008. Retrieved 20 December 2007.
- ^ Stern, D. P. (13 February 2005). "Wave Mechanics". NASA Goddard Space Flight Center. Archived from the original on 13 May 2008. Retrieved 16 April 2008.
- ^ Staff (2003). "Hydrogen (H2) Properties, Uses, Applications: Hydrogen Gas and Liquid Hydrogen". Universal Industrial Gases, Inc. Archived from the original on 19 February 2008. Retrieved 5 February 2008.
- ^ Green, Richard A.; et al. (2012). "The theory and practice of hyperpolarization in magnetic resonance using parahydrogen". Prog. Nucl. Magn. Reson. Spectrosc. 67: 1–48. doi:10.1016/j.pnmrs.2012.03.001. PMID 23101588. Archived from the original on 28 August 2021. Retrieved 28 August 2021.
- ^ "Die Entdeckung des para-Wasserstoffs (The discovery of para-hydrogen)". Max-Planck-Institut für Biophysikalische Chemie (in German). Archived from the original on 16 November 2020. Retrieved 9 November 2020.
- ^ Milenko, Yu. Ya.; Sibileva, R. M.; Strzhemechny, M. A. (1997). "Natural ortho-para conversion rate in liquid and gaseous hydrogen". Journal of Low Temperature Physics. 107 (1–2): 77–92. Bibcode:1997JLTP..107...77M. doi:10.1007/BF02396837. S2CID 120832814.
- ^ Hritz, J. (March 2006). "CH. 6 – Hydrogen" (PDF). NASA Glenn Research Center Glenn Safety Manual, Document GRC-MQSA.001. NASA. Archived from the original (PDF) on 16 February 2008. Retrieved 5 February 2008.
- ^ Amos, Wade A. (1 November 1998). "Costs of Storing and Transporting Hydrogen" (PDF). National Renewable Energy Laboratory. pp. 6–9. Archived (PDF) from the original on 26 December 2014. Retrieved 19 May 2015.
- ^ Svadlenak, R. E.; Scott, A. B. (1957). "The Conversion of Ortho- to Parahydrogen on Iron Oxide-Zinc Oxide Catalysts". Journal of the American Chemical Society. 79 (20): 5385–5388. doi:10.1021/ja01577a013.
- ^ Clark, J. (2002). "The Acidity of the Hydrogen Halides". Chemguide. Archived from the original on 20 February 2008. Retrieved 9 March 2008.
- ^ Kimball, J. W. (7 August 2003). "Hydrogen". Kimball's Biology Pages. Archived from the original on 4 March 2008. Retrieved 4 March 2008.
- ^ IUPAC Compendium of Chemical Terminology, Electronic version, Hydrogen Bond Archived 19 March 2008 at the Wayback Machine
- ^ Sandrock, G. (2 May 2002). "Metal-Hydrogen Systems". Sandia National Laboratories. Archived from the original on 24 February 2008. Retrieved 23 March 2008.
- ^ a b "Structure and Nomenclature of Hydrocarbons". Purdue University. Archived from the original on 11 June 2012. Retrieved 23 March 2008.
- ^ "Organic Chemistry". Dictionary.com. Lexico Publishing Group. 2008. Archived from the original on 18 April 2008. Retrieved 23 March 2008.
- ^ "Biochemistry". Dictionary.com. Lexico Publishing Group. 2008. Archived from the original on 29 March 2008. Retrieved 23 March 2008.
- ^ Takeshita, T.; Wallace, W. E.; Craig, R. S. (1974). "Hydrogen solubility in 1:5 compounds between yttrium or thorium and nickel or cobalt". Inorganic Chemistry. 13 (9): 2282–2283. doi:10.1021/ic50139a050.
- ^ Kirchheim, R.; Mutschele, T.; Kieninger, W.; Gleiter, H.; Birringer, R.; Koble, T. (1988). "Hydrogen in amorphous and nanocrystalline metals". Materials Science and Engineering. 99 (1–2): 457–462. doi:10.1016/0025-5416(88)90377-1.
- ^ Kirchheim, R. (1988). "Hydrogen solubility and diffusivity in defective and amorphous metals". Progress in Materials Science. 32 (4): 262–325. doi:10.1016/0079-6425(88)90010-2.
- ^ Christensen, C. H.; Nørskov, J. K.; Johannessen, T. (9 July 2005). "Making society independent of fossil fuels – Danish researchers reveal new technology". Technical University of Denmark. Archived from the original on 21 May 2015. Retrieved 19 May 2015.
- ^ Moers, K. (1920). "Investigations on the Salt Character of Lithium Hydride". Zeitschrift für Anorganische und Allgemeine Chemie. 113 (191): 179–228. doi:10.1002/zaac.19201130116. Archived (PDF) from the original on 24 August 2019. Retrieved 24 August 2019.
- ^ Downs, A. J.; Pulham, C. R. (1994). "The hydrides of aluminium, gallium, indium, and thallium: a re-evaluation". Chemical Society Reviews. 23 (3): 175–184. doi:10.1039/CS9942300175.
- ^ Hibbs, D. E.; Jones, C.; Smithies, N. A. (1999). "A remarkably stable indium trihydride complex: synthesis and characterisation of [InH3P(C6H11)3]". Chemical Communications (2): 185–186. doi:10.1039/a809279f.
- ^ a b c Miessler, G. L.; Tarr, D. A. (2003). Inorganic Chemistry (3rd ed.). Prentice Hall. ISBN 978-0-13-035471-6.
- ^ a b c d e f g h Application of hydrides in hydrogen storage and compression: Achievements, outlook and perspectives, Jose Bellosta von Colbe a, Jose-Ram_on Ares b, Jussara Barale i, et al, Department of Nanotechnology, Helmholtz-Zentrum Geesthacht, Max-Plank-Str. 59, 21502 Geesthacht, Germany,
- ^ a b c d "Advanced Solid State Hydrogen Battery". greenhy2.com.au. Retrieved 2024-02-12.
- ^ a b Hydrogen - A sustainable energy carrier Kasper T. Møllera, Torben R. Jensena,⁎, Etsuo Akibab,c,d, Hai-wen Lib,ca Interdisciplinary Nanoscience Center (iNANO) and Department of Chemistry, Aarhus University, DK-8000 Aarhus, Denmark
- ^ a b Hydrogen storage methods The Science of Nature • April 2004 Andreas Züttel École Polytechnique Fédérale de Lausanne
- ^ a b c d e f g Cite error: The named reference
TETwas invoked but never defined (see the help page). - ^ Okumura, A. M.; Yeh, L. I.; Myers, J. D.; Lee, Y. T. (1990). "Infrared spectra of the solvated hydronium ion: vibrational predissociation spectroscopy of mass-selected H3O+•(H2O)n•(H2)m". Journal of Physical Chemistry. 94 (9): 3416–3427. doi:10.1021/j100372a014.
- ^ Perdoncin, G.; Scorrano, G. (1977). "Protonation Equilibria in Water at Several Temperatures of Alcohols, Ethers, Acetone, Dimethyl Sulfide, and Dimethyl Sulfoxide". Journal of the American Chemical Society. 99 (21): 6983–6986. doi:10.1021/ja00463a035.
- ^ Carrington, A.; McNab, I. R. (1989). "The infrared predissociation spectrum of triatomic hydrogen cation (H3+)". Accounts of Chemical Research. 22 (6): 218–222. doi:10.1021/ar00162a004.
- ^ Gurov, Y. B.; Aleshkin, D. V.; Behr, M. N.; Lapushkin, S. V.; Morokhov, P. V.; Pechkurov, V. A.; Poroshin, N. O.; Sandukovsky, V. G.; Tel'kushev, M. V.; Chernyshev, B. A.; Tschurenkova, T. D. (2004). "Spectroscopy of superheavy hydrogen isotopes in stopped-pion absorption by nuclei". Physics of Atomic Nuclei. 68 (3): 491–97. Bibcode:2005PAN....68..491G. doi:10.1134/1.1891200. S2CID 122902571.
- ^ Korsheninnikov, A.; Nikolskii, E.; Kuzmin, E.; Ozawa, A.; Morimoto, K.; Tokanai, F.; Kanungo, R.; Tanihata, I.; et al. (2003). "Experimental Evidence for the Existence of 7H and for a Specific Structure of 8He". Physical Review Letters. 90 (8): 082501. Bibcode:2003PhRvL..90h2501K. doi:10.1103/PhysRevLett.90.082501. PMID 12633420.
- ^ Urey, H. C.; Brickwedde, F. G.; Murphy, G. M. (1933). "Names for the Hydrogen Isotopes". Science. 78 (2035): 602–603. Bibcode:1933Sci....78..602U. doi:10.1126/science.78.2035.602. PMID 17797765.
- ^ Oda, Y.; Nakamura, H.; Yamazaki, T.; Nagayama, K.; Yoshida, M.; Kanaya, S.; Ikehara, M. (1992). "1H NMR studies of deuterated ribonuclease HI selectively labeled with protonated amino acids". Journal of Biomolecular NMR. 2 (2): 137–47. doi:10.1007/BF01875525. PMID 1330130. S2CID 28027551.
- ^ Broad, W. J. (11 November 1991). "Breakthrough in Nuclear Fusion Offers Hope for Power of Future". The New York Times. Archived from the original on 29 January 2021. Retrieved 12 February 2008.
- ^ a b Traub, R. J.; Jensen, J. A. (June 1995). "Tritium radioluminescent devices, Health and Safety Manual" (PDF). International Atomic Energy Agency. p. 2.4. Archived (PDF) from the original on 6 September 2015. Retrieved 20 May 2015.
- ^ Staff (15 November 2007). "Tritium". U.S. Environmental Protection Agency. Archived from the original on 2 January 2008. Retrieved 12 February 2008.
- ^ Nave, C. R. (2006). "Deuterium-Tritium Fusion". HyperPhysics. Georgia State University. Archived from the original on 16 March 2008. Retrieved 8 March 2008.
- ^ Kendall, C.; Caldwell, E. (1998). C. Kendall; J. J. McDonnell (eds.). "Chapter 2: Fundamentals of Isotope Geochemistry". Isotope Tracers in Catchment Hydrology. US Geological Survey: 51–86. doi:10.1016/B978-0-444-81546-0.50009-4. Archived from the original on 14 March 2008. Retrieved 8 March 2008.
- ^ "The Tritium Laboratory". University of Miami. 2008. Archived from the original on 28 February 2008. Retrieved 8 March 2008.
- ^ a b Holte, A. E.; Houck, M. A.; Collie, N. L. (2004). "Potential Role of Parasitism in the Evolution of Mutualism in Astigmatid Mites". Experimental and Applied Acarology. 25 (2): 97–107. doi:10.1023/A:1010655610575. PMID 11513367. S2CID 13159020.
- ^ van der Krogt, P. (5 May 2005). "Hydrogen". Elementymology & Elements Multidict. Archived from the original on 23 January 2010. Retrieved 20 December 2010.
- ^ § IR-3.3.2, Provisional Recommendations Archived 9 February 2016 at the Wayback Machine, Nomenclature of Inorganic Chemistry, Chemical Nomenclature and Structure Representation Division, IUPAC. Accessed on line 3 October 2007.
- ^ IUPAC (1997). "Muonium". In A.D. McNaught, A. Wilkinson (ed.). Compendium of Chemical Terminology (2nd ed.). Blackwell Scientific Publications. doi:10.1351/goldbook.M04069. ISBN 978-0-86542-684-9. Archived from the original on 13 March 2008. Retrieved 15 November 2016.
- ^ V.W. Hughes; et al. (1960). "Formation of Muonium and Observation of its Larmor Precession". Physical Review Letters. 5 (2): 63–65. Bibcode:1960PhRvL...5...63H. doi:10.1103/PhysRevLett.5.63.
- ^ Bondi, D.K.; Connor, J.N.L.; Manz, J.; Römelt, J. (20 October 1983). "Exact quantum and vibrationally adiabatic quantum, semiclassical and quasiclassical study of the collinear reactions Cl + MuCl, Cl + HCl, Cl + DCl". Molecular Physics. 50 (3): 467–488. Bibcode:1983MolPh..50..467B. doi:10.1080/00268978300102491. ISSN 0026-8976.
- ^ W.H. Koppenol; IUPAC (2001). "Names for muonium and hydrogen atoms and their ions" (PDF). Pure and Applied Chemistry. 73 (2): 377–380. doi:10.1351/pac200173020377. S2CID 97138983. Archived (PDF) from the original on 14 May 2011. Retrieved 15 November 2016.
- ^ Holman, Jack P. (2002). Heat transfer (9th ed.). New York, NY: McGraw-Hill. pp. 600–606. ISBN 0-07-240655-0. OCLC 46959719.
- ^ Incropera 1 Dewitt 2 Bergman 3 Lavigne 4, Frank P. 1 David P. 2 Theodore L. 3 Adrienne S. 4 (2007). Fundamentals of heat and mass transfer (6th ed.). Hoboken, NJ: John Wiley and Sons, Inc. pp. 941–950. ISBN 978-0-471-45728-2. OCLC 62532755.
{{cite book}}: CS1 maint: numeric names: authors list (link) - ^ Boyle, R. (1672). Tracts written by the Honourable Robert Boyle containing new experiments, touching the relation betwixt flame and air, and about explosions, an hydrostatical discourse occasion'd by some objections of Dr. Henry More against some explications of new experiments made by the author of these tracts: To which is annex't, an hydrostatical letter, dilucidating an experiment about a way of weighing water in water, new experiments, of the positive or relative levity of bodies under water, of the air's spring on bodies under water, about the differing pressure of heavy solids and fluids. Printed for Richard Davis. pp. 64–65.
- ^ Winter, M. (2007). "Hydrogen: historical information". WebElements Ltd. Archived from the original on 10 April 2008. Retrieved 5 February 2008.
- ^ Szydło, Z. A. (2020). "Hydrogen - Some Historical Highlights". Chemistry-Didactics-Ecology-Metrology. 25 (1–2): 5–34. doi:10.2478/cdem-2020-0001. S2CID 231776282.
- ^ Ramsay, W. (1896). The gases of the atmosphere: The history of their discovery. Macmillan. p. 19.
- ^ Musgrave, A. (1976). "Why did oxygen supplant phlogiston? Research programmes in the Chemical Revolution". In Howson, C. (ed.). Method and appraisal in the physical sciences. The Critical Background to Modern Science, 1800–1905. Cambridge University Press. doi:10.1017/CBO9780511760013. ISBN 978-0-521-21110-9. Retrieved 22 October 2011.
- ^ Cavendish, Henry (12 May 1766). "Three Papers, Containing Experiments on Factitious Air, by the Hon. Henry Cavendish, F. R. S." Philosophical Transactions. 56: 141–184. Bibcode:1766RSPT...56..141C. doi:10.1098/rstl.1766.0019. JSTOR 105491.
- ^ a b "Hydrogen". Van Nostrand's Encyclopedia of Chemistry. Wylie-Interscience. 2005. pp. 797–799. ISBN 978-0-471-61525-5.
- ^ a b c d e f g h i j k Emsley, John (2001). Nature's Building Blocks. Oxford: Oxford University Press. pp. 183–191. ISBN 978-0-19-850341-5.
- ^ Stwertka, Albert (1996). A Guide to the Elements. Oxford University Press. pp. 16–21. ISBN 978-0-19-508083-4.
- ^ National Electrical Manufacturers Association (1946). A chronological history of electrical development from 600 B.C. New York, N.Y., National Electrical Manufacturers Association. p. 102. Archived from the original on 4 March 2016. Retrieved 9 February 2016.
- ^ Stockel, J.F; j.d. Dunlop; Betz, F (1980). "NTS-2 Nickel-Hydrogen Battery Performance 31". Journal of Spacecraft and Rockets. 17: 31–34. Bibcode:1980JSpRo..17...31S. doi:10.2514/3.57704.
- ^ Jannette, A. G.; Hojnicki, J. S.; McKissock, D. B.; Fincannon, J.; Kerslake, T. W.; Rodriguez, C. D. (July 2002). Validation of international space station electrical performance model via on-orbit telemetry (PDF). IECEC '02. 2002 37th Intersociety Energy Conversion Engineering Conference, 2002. pp. 45–50. doi:10.1109/IECEC.2002.1391972. hdl:2060/20020070612. ISBN 0-7803-7296-4. Archived (PDF) from the original on 14 May 2010. Retrieved 11 November 2011.
- ^ Anderson, P. M.; Coyne, J. W. (2002). "A lightweight, high reliability, single battery power system for interplanetary spacecraft". Proceedings, IEEE Aerospace Conference. Vol. 5. pp. 5–2433. doi:10.1109/AERO.2002.1035418. ISBN 978-0-7803-7231-3. S2CID 108678345.
- ^ "Mars Global Surveyor". Astronautix.com. Archived from the original on 10 August 2009. Retrieved 6 April 2009.
- ^ Lori Tyahla, ed. (7 May 2009). "Hubble servicing mission 4 essentials". NASA. Archived from the original on 13 March 2015. Retrieved 19 May 2015.
- ^ Hendrix, Susan (25 November 2008). Lori Tyahla (ed.). "Extending Hubble's mission life with new batteries". NASA. Archived from the original on 5 March 2016. Retrieved 19 May 2015.
- ^ Crepeau, R. (1 January 2006). Niels Bohr: The Atomic Model. ISBN 978-1-4298-0723-4.
{{cite book}}:|journal=ignored (help) - ^ Berman, R.; Cooke, A. H.; Hill, R. W. (1956). "Cryogenics". Annual Review of Physical Chemistry. 7: 1–20. Bibcode:1956ARPC....7....1B. doi:10.1146/annurev.pc.07.100156.000245.
- ^ Charlton, Mike; Van Der Werf, Dirk Peter (1 March 2015). "Advances in antihydrogen physics". Science Progress. 98 (1): 34–62. doi:10.3184/003685015X14234978376369. PMC 10365473. PMID 25942774. S2CID 23581065.
- ^ Kellerbauer, Alban (29 January 2015). "Why Antimatter Matters". European Review. 23 (1): 45–56. doi:10.1017/S1062798714000532. S2CID 58906869.
- ^ Gagnon, S. "Hydrogen". Jefferson Lab. Archived from the original on 10 April 2008. Retrieved 5 February 2008.
- ^ Haubold, H.; Mathai, A. M. (15 November 2007). "Solar Thermonuclear Energy Generation". Columbia University. Archived from the original on 11 December 2011. Retrieved 12 February 2008.
- ^ "Hydrogen". mysite.du.edu. Archived from the original on 18 April 2009. Retrieved 20 April 2008.
- ^ Storrie-Lombardi, L. J.; Wolfe, A. M. (2000). "Surveys for z > 3 Damped Lyman-alpha Absorption Systems: the Evolution of Neutral Gas". Astrophysical Journal. 543 (2): 552–576. arXiv:astro-ph/0006044. Bibcode:2000ApJ...543..552S. doi:10.1086/317138. S2CID 120150880.
- ^ a b Rhys Grinter; Kropp, A.; Venugopal; et al. (2023). "Structural basis for bacterial energy extraction from atmospheric hydrogen". Nature. 615 (7952): 541–547. Bibcode:2023Natur.615..541G. doi:10.1038/s41586-023-05781-7. PMC 10017518. PMID 36890228.
- ^ Dresselhaus, M.; et al. (15 May 2003). "Basic Research Needs for the Hydrogen Economy" (PDF). APS March Meeting Abstracts. 2004. Argonne National Laboratory, U.S. Department of Energy, Office of Science Laboratory: m1.001. Bibcode:2004APS..MAR.m1001D. Archived from the original (PDF) on 13 February 2008. Retrieved 5 February 2008.
- ^ McCall Group; Oka Group (22 April 2005). "H3+ Resource Center". Universities of Illinois and Chicago. Archived from the original on 11 October 2007. Retrieved 5 February 2008.
- ^ Helm, H.; et al. (2003), "Coupling of Bound States to Continuum States in Neutral Triatomic Hydrogen", Dissociative Recombination of Molecular Ions with Electrons, Department of Molecular and Optical Physics, University of Freiburg, Germany, pp. 275–288, doi:10.1007/978-1-4615-0083-4_27, ISBN 978-1-4613-4915-0
- ^ a b Baade, William F.; Parekh, Uday N.; Raman, Venkat S. (2001). "Hydrogen". Kirk-Othmer Encyclopedia of Chemical Technology. doi:10.1002/0471238961.0825041803262116.a01.pub2. ISBN 9780471484943.
- ^ Freyermuth, George H. "1934 Patent: "The manufacture of hydrogen from methane hydrocarbons by the action of steam at elevated temperature"". Patent Full-Text Databases. United States Patent and Trademark Office. Archived from the original on 1 October 2021. Retrieved 30 October 2020.
- ^ Press, Roman J.; Santhanam, K. S. V.; Miri, Massoud J.; Bailey, Alla V.; Takacs, Gerald A. (2008). Introduction to Hydrogen Technology. John Wiley & Sons. p. 249. ISBN 978-0-471-77985-8.
- ^ a b c Oxtoby, D. W. (2002). Principles of Modern Chemistry (5th ed.). Thomson Brooks/Cole. ISBN 978-0-03-035373-4.
- ^ Funderburg, E. (2008). "Why Are Nitrogen Prices So High?". The Samuel Roberts Noble Foundation. Archived from the original on 9 May 2001. Retrieved 11 March 2008.
- ^ "Hydrogen Properties, Uses, Applications". Universal Industrial Gases, Inc. 2007. Archived from the original on 27 March 2008. Retrieved 11 March 2008.
- ^ Hannula, Ilkka (2015). "Co-production of synthetic fuels and district heat from biomass residues, carbon dioxide and electricity: Performance and cost analysis". Biomass and Bioenergy. 74: 26–46. doi:10.1016/j.biombioe.2015.01.006. ISSN 0961-9534.
- ^ Gong, Ming; Zhou, Wu; Tsai, Mon-Che; Zhou, Jigang; Guan, Mingyun; Lin, Meng-Chang; Zhang, Bo; Hu, Yongfeng; Wang, Di-Yan; Yang, Jiang; Pennycook, Stephen J.; Hwang, Bing-Joe; Dai, Hongjie (2014). "Nanoscale nickel oxide/Nickel heterostructures for active hydrogen evolution electrocatalysis". Nature Communications. 5: 4695. Bibcode:2014NatCo...5.4695G. doi:10.1038/ncomms5695. PMID 25146255. S2CID 205329127.
- ^ Lees, A. (2007). "Chemicals from salt". BBC. Archived from the original on 26 October 2007. Retrieved 11 March 2008.
- ^ Von Wald, Gregory A. (2020). "Optimization-based technoeconomic analysis of molten-media methane pyrolysis for reducing industrial sector CO2 emissions". Sustainable Energy & Fuels. 4 (9). Royal Society of Chemistry: 4598–4613. doi:10.1039/D0SE00427H. S2CID 225676190. Archived from the original on 8 November 2020. Retrieved 31 October 2020.
- ^ Schneider, Stefan (2020). "State of the Art of Hydrogen Production via Pyrolysis of Natural Gas". ChemBioEng Reviews. 7 (5). Wiley Online Library: 150–158. doi:10.1002/cben.202000014.
- ^ Cartwright, Jon. "The reaction that would give us clean fossil fuels forever". New Scientist. Archived from the original on 26 October 2020. Retrieved 30 October 2020.
- ^ Karlsruhe Institute of Technology. "Hydrogen from methane without CO2 emissions". Phys.Org. Archived from the original on 21 October 2020. Retrieved 30 October 2020.
- ^ Upham, D. Chester (2017). "Catalytic molten metals for the direct conversion of methane to hydrogen and separable carbon". Science. 358 (6365). American Association for Advancement of Science: 917–921. Bibcode:2017Sci...358..917U. doi:10.1126/science.aao5023. PMID 29146810. S2CID 206663568.
- ^ Clarke, Palmer (2020). "Dry reforming of methane catalyzed by molten metal alloys". Nature Catalysis. 3: 83–89. doi:10.1038/s41929-019-0416-2. S2CID 210862772. Archived from the original on 29 January 2021. Retrieved 31 October 2020.
- ^ Gusev, Alexander. "KITT/IASS – Producing CO2 Free Hydrogen From Natural Gas For Energy Usage". European Energy Innovation. Institute for Advanced Sustainability Studies. Archived from the original on 29 January 2021. Retrieved 30 October 2020.
- ^ Fernandez, Sonia. "Researchers develop potentially low-cost, low-emissions technology that can convert methane without forming CO2". Phys-Org. American Institute of Physics. Archived from the original on 19 October 2020. Retrieved 19 October 2020.
- ^ BASF. "BASF researchers working on fundamentally new, low-carbon production processes, Methane Pyrolysis". United States Sustainability. BASF. Archived from the original on 19 October 2020. Retrieved 19 October 2020.
- ^ Weimer, Al (25 May 2005). "Development of solar-powered thermochemical production of hydrogen from water" (PDF). Solar Thermochemical Hydrogen Generation Project. Archived (PDF) from the original on 17 April 2007. Retrieved 21 December 2008.
- ^ Perret, R. "Development of Solar-Powered Thermochemical Production of Hydrogen from Water, DOE Hydrogen Program, 2007" (PDF). Archived from the original (PDF) on 27 May 2010. Retrieved 17 May 2008.
- ^ Parmuzina, A.V.; Kravchenko, O.V. (2008). "Activation of aluminium metal to evolve hydrogen from water". International Journal of Hydrogen Energy. 33 (12): 3073–3076. doi:10.1016/j.ijhydene.2008.02.025.
- ^ Lubitz, Wolfgang; Reijerse, Eduard; Van Gastel, Maurice (2007). "[NiFe] and [FeFe] Hydrogenases Studied by Advanced Magnetic Resonance Techniques". Chemical Reviews. 107 (10): 4331–4365. doi:10.1021/cr050186q. PMID 17845059.
- ^ Smil, Vaclav (2004). Enriching the Earth: Fritz Haber, Carl Bosch, and the Transformation of World Food Production (1st ed.). Cambridge, MA: MIT. ISBN 978-0-262-69313-4.
- ^ Chemistry Operations (15 December 2003). "Hydrogen". Los Alamos National Laboratory. Archived from the original on 4 March 2011. Retrieved 5 February 2008.
- ^ a b c d e IPCC (2022). Shukla, P.R.; Skea, J.; Slade, R.; Al Khourdajie, A.; et al. (eds.). Climate Change 2022: Mitigation of Climate Change (PDF). Contribution of Working Group III to the Sixth Assessment Report of the Intergovernmental Panel on Climate Change. Cambridge, UK and New York, NY, US: Cambridge University Press (In Press). pp. 91–92. doi:10.1017/9781009157926. ISBN 9781009157926.
- ^ Cite error: The named reference
:18was invoked but never defined (see the help page). - ^ McCarthy, J. (31 December 1995). "Hydrogen". Stanford University. Archived from the original on 14 March 2008. Retrieved 14 March 2008.
- ^ Bonheure, Mike; Vandewalle, Laurien A.; Marin, Guy B.; Van Geem, Kevin M. (March 2021). "Dream or Reality? Electrification of the Chemical Process Industries". CEP Magazine. American Institute of Chemical Engineers. Archived from the original on 17 July 2021. Retrieved 6 July 2021.
- ^ Griffiths, Steve; Sovacool, Benjamin K.; Kim, Jinsoo; Bazilian, Morgan; et al. (2021). "Industrial decarbonization via hydrogen: A critical and systematic review of developments, socio-technical systems and policy options". Energy Research & Social Science. 80: 39. doi:10.1016/j.erss.2021.102208. ISSN 2214-6296. Archived from the original on 16 October 2021. Retrieved 11 September 2021.
- ^ Palys, Matthew J.; Daoutidis, Prodromos (2020). "Using hydrogen and ammonia for renewable energy storage: A geographically comprehensive techno-economic study". Computers & Chemical Engineering. 136: 106785. doi:10.1016/j.compchemeng.2020.106785. ISSN 0098-1354.
- ^ "Hydrogen industry must clean itself up before expanding into new…". Canary Media. 31 August 2021. Retrieved 2023-04-05.
- ^ IRENA (2021). World Energy Transitions Outlook: 1.5°C Pathway (PDF). pp. 12, 22. ISBN 978-92-9260-334-2. Archived (PDF) from the original on 11 June 2021.
- ^ IEA (2021). Net Zero by 2050: A Roadmap for the Global Energy Sector (PDF). pp. 15, 75–76. Archived (PDF) from the original on 23 May 2021.
- ^ "Technical challenge of designing a micro-grid to service a luxury resort and organic farm". greenhy2.com.au. Retrieved 2024-02-12.
- ^ "The Heart of the Solution is High Density Energy Storage". greenhy2.com.au. Retrieved 2024-02-12.
- ^ "Store Excess Daytime Solar and Charge your vehicles at night". greenhy2.com.au. Retrieved 2024-02-12.
- ^ "No Future for Diesel". greenhy2.com.au. Retrieved 2024-02-12.
- ^ Blank, Thomas; Molly, Patrick (January 2020). "Hydrogen's Decarbonization Impact for Industry" (PDF). Rocky Mountain Institute. pp. 2, 7, 8. Archived (PDF) from the original on 22 September 2020.
- ^ Plötz, Patrick (2022-01-31). "Hydrogen technology is unlikely to play a major role in sustainable road transport". Nature Electronics. 5 (1): 8–10. doi:10.1038/s41928-021-00706-6. ISSN 2520-1131. S2CID 246465284.
- ^ Le Comber, P. G.; Jones, D. I.; Spear, W. E. (1977). "Hall effect and impurity conduction in substitutionally doped amorphous silicon". Philosophical Magazine. 35 (5): 1173–1187. Bibcode:1977PMag...35.1173C. doi:10.1080/14786437708232943.
- ^ Van de Walle, C. G. (2000). "Hydrogen as a cause of doping in zinc oxide" (PDF). Physical Review Letters. 85 (5): 1012–1015. Bibcode:2000PhRvL..85.1012V. doi:10.1103/PhysRevLett.85.1012. hdl:11858/00-001M-0000-0026-D0E6-E. PMID 10991462. Archived (PDF) from the original on 15 August 2017. Retrieved 1 August 2018.
- ^ Janotti, A.; Van De Walle, C. G. (2007). "Hydrogen multicentre bonds". Nature Materials. 6 (1): 44–47. Bibcode:2007NatMa...6...44J. doi:10.1038/nmat1795. PMID 17143265.
- ^ Kilic, C.; Zunger, Alex (2002). "n-type doping of oxides by hydrogen". Applied Physics Letters. 81 (1): 73–75. Bibcode:2002ApPhL..81...73K. doi:10.1063/1.1482783. S2CID 96415065.
- ^ Peacock, P. W.; Robertson, J. (2003). "Behavior of hydrogen in high dielectric constant oxide gate insulators". Applied Physics Letters. 83 (10): 2025–2027. Bibcode:2003ApPhL..83.2025P. doi:10.1063/1.1609245.
- ^ Durgutlu, A. (2003). "Experimental investigation of the effect of hydrogen in argon as a shielding gas on TIG welding of austenitic stainless steel". Materials & Design. 25 (1): 19–23. doi:10.1016/j.matdes.2003.07.004.
- ^ "Atomic Hydrogen Welding". Specialty Welds. 2007. Archived from the original on 16 July 2011.
- ^ Hardy, W. N. (2003). "From H2 to cryogenic H masers to HiTc superconductors: An unlikely but rewarding path". Physica C: Superconductivity. 388–389: 1–6. Bibcode:2003PhyC..388....1H. doi:10.1016/S0921-4534(02)02591-1.
- ^ Almqvist, Ebbe (2003). History of industrial gases. New York, N.Y.: Kluwer Academic/Plenum Publishers. pp. 47–56. ISBN 978-0-306-47277-0. Retrieved 20 May 2015.
- ^ Block, M. (3 September 2004). Hydrogen as Tracer Gas for Leak Detection. 16th WCNDT 2004. Montreal, Canada: Sensistor Technologies. Archived from the original on 8 January 2009. Retrieved 25 March 2008.
- ^ "Report from the Commission on Dietary Food Additive Intake" (PDF). European Union. Archived (PDF) from the original on 16 February 2008. Retrieved 5 February 2008.
- ^ Reinsch, J.; Katz, A.; Wean, J.; Aprahamian, G.; MacFarland, J. T. (1980). "The deuterium isotope effect upon the reaction of fatty acyl-CoA dehydrogenase and butyryl-CoA". J. Biol. Chem. 255 (19): 9093–97. doi:10.1016/S0021-9258(19)70531-6. PMID 7410413.
- ^ "NASA/TM—2002-211915: Solid Hydrogen Experiments for Atomic Propellants" (PDF). Archived (PDF) from the original on 9 July 2021. Retrieved 2 July 2021.
- ^ Bergeron, K. D. (2004). "The Death of no-dual-use". Bulletin of the Atomic Scientists. 60 (1): 15–17. Bibcode:2004BuAtS..60a..15B. doi:10.2968/060001004. Archived from the original on 19 April 2008. Retrieved 13 April 2008.
- ^ Cammack, R.; Robson, R. L. (2001). Hydrogen as a Fuel: Learning from Nature. Taylor & Francis Ltd. pp. 202–203. ISBN 978-0-415-24242-4. Archived from the original on 29 January 2021. Retrieved 3 September 2020.
- ^ Rhee, T. S.; Brenninkmeijer, C. A. M.; Röckmann, T. (19 May 2006). "The overwhelming role of soils in the global atmospheric hydrogen cycle" (PDF). Atmospheric Chemistry and Physics. 6 (6): 1611–1625. Bibcode:2006ACP.....6.1611R. doi:10.5194/acp-6-1611-2006. Archived (PDF) from the original on 24 August 2019. Retrieved 24 August 2019.
- ^ Alex Wilkins (Mar 8, 2023). "Soil bacteria enzyme generates electricity from hydrogen in the air". New Scientist. 257 (3430): 13. Bibcode:2023NewSc.257...13W. doi:10.1016/S0262-4079(23)00459-1. S2CID 257625443.
- ^ Eisenmann, Alexander; Amann, Anton; Said, Michael; Datta, Bettina; Ledochowski, Maximilian (2008). "Implementation and interpretation of hydrogen breath tests" (PDF). Journal of Breath Research. 2 (4): 046002. Bibcode:2008JBR.....2d6002E. doi:10.1088/1752-7155/2/4/046002. PMID 21386189. S2CID 31706721. Archived from the original (PDF) on 29 January 2021. Retrieved 26 December 2020.
- ^ Kruse, O.; Rupprecht, J.; Bader, K.; Thomas-Hall, S.; Schenk, P. M.; Finazzi, G.; Hankamer, B. (2005). "Improved photobiological H2 production in engineered green algal cells" (PDF). The Journal of Biological Chemistry. 280 (40): 34170–7. doi:10.1074/jbc.M503840200. PMID 16100118. S2CID 5373909. Archived (PDF) from the original on 29 January 2021. Retrieved 24 August 2019.
- ^ Smith, Hamilton O.; Xu, Qing (2005). "IV.E.6 Hydrogen from Water in a Novel Recombinant Oxygen-Tolerant Cyanobacteria System" (PDF). FY2005 Progress Report. United States Department of Energy. Archived (PDF) from the original on 29 December 2016. Retrieved 6 August 2016.
- ^ Williams, C. (24 February 2006). "Pond life: the future of energy". Science. The Register. Archived from the original on 9 May 2011. Retrieved 24 March 2008.
- ^ "MyChem: Chemical" (PDF). Archived from the original (PDF) on 1 October 2018. Retrieved 1 October 2018.
- ^ a b Brown, W. J.; et al. (1997). "Safety Standard for Hydrogen and Hydrogen Systems" (PDF). NASA. NSS 1740.16. Archived (PDF) from the original on 1 May 2017. Retrieved 12 July 2017.
- ^ "Liquid Hydrogen MSDS" (PDF). Praxair, Inc. September 2004. Archived from the original (PDF) on 27 May 2008. Retrieved 16 April 2008.
- ^ "'Bugs' and hydrogen embrittlement". Science News. 128 (3): 41. 20 July 1985. doi:10.2307/3970088. JSTOR 3970088.
- ^ Hayes, B. "Union Oil Amine Absorber Tower". TWI. Archived from the original on 20 November 2008. Retrieved 29 January 2010.
- ^ Walker, James L.; Waltrip, John S.; Zanker, Adam (1988). "Lactic acid to magnesium supply-demand relationships". In John J. McKetta; William Aaron Cunningham (eds.). Encyclopedia of Chemical Processing and Design. Vol. 28. New York: Dekker. p. 186. ISBN 978-0-8247-2478-8. Retrieved 20 May 2015.
Further reading[edit]
- The Hyperfine Splitting in Hydrogen - The Feynman Lectures on Physics
- Chart of the Nuclides (17th ed.). Knolls Atomic Power Laboratory. 2010. ISBN 978-0-9843653-0-2.
- Newton, David E. (1994). The Chemical Elements. New York: Franklin Watts. ISBN 978-0-531-12501-4.
- Rigden, John S. (2002). Hydrogen: The Essential Element. Cambridge, Massachusetts: Harvard University Press. ISBN 978-0-531-12501-4.
- Romm, Joseph J. (2004). The Hype about Hydrogen, Fact and Fiction in the Race to Save the Climate. Island Press. ISBN 978-1-55963-703-9.
- Scerri, Eric (2007). The Periodic System, Its Story and Its Significance. New York: Oxford University Press. ISBN 978-0-19-530573-9.
External links[edit]
- Basic Hydrogen Calculations of Quantum Mechanics
- Hydrogen at The Periodic Table of Videos (University of Nottingham)
- High temperature hydrogen phase diagram
- Wavefunction of hydrogen
Template:Featured article is only for Wikipedia:Featured articles.



