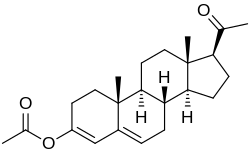
Progesterone 3-acetyl enol ether
 | |
| Clinical data | |
|---|---|
| Other names | Progesterone acetate; Progesterone 3-acetate; 3-Acetoxypregna-3,5-diene-20-one; 20-Oxopregna-3,5-dien-3-yl acetate; 3,5-Progesterol acetate; NSC-124740 |
| Drug class | Progestogen; Progestogen ether |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| Chemical and physical data | |
| Formula | C23H32O3 |
| Molar mass | 356.506 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Progesterone 3-acetyl enol ether, also known as progesterone acetate,[1] as well as 3-acetoxypregna-3,5-dien-20-one, is a progestin which was never marketed.[2][3][4][5] It was reported to possess similar potency to progesterone and hydroxyprogesterone caproate in the rabbit endometrial carbonic anhydrase test, a bioassay of progestogenic activity.[2][3] In addition, it was able to maintain pregnancy in animals.[2] Progesterone 3-acetyl enol ether is closely related to quingestrone, which is also known as progesterone 3-cyclopentyl enol ether and was formerly marketed as an oral contraceptive.[6]
The 3-acetyl ether may be cleaved from progesterone 3-acetyl enol ether in vivo and, based on its chemical structure, this may result in the transformation of progesterone 3-acetyl enol ether into 3α-dihydroprogesterone and/or 3β-dihydroprogesterone. 3β-Dihydroprogesterone has been reported to possess about the same progestogenic potency as progesterone in the Clauberg test, whereas 3α-dihydroprogesterone was not assessed.[7]
The C3 enol ethers of progesterone are less suited for use via depot injection relative to progestogen esters like hydroxyprogesterone caproate due to their susceptibility to oxidative metabolism.[8]
See also[edit]
- Progestogen ester § Progestogen ethers
- List of progestogen esters § Ethers of progesterone derivatives
References[edit]
- ^ "ChemIDplus - 0004954067 - YIPYJRPRHUJJDP-WKOLOUIMSA-N - Pregna-3,5-dien-20-one, 3-(acetyloxy)- - Similar structures search, synonyms, formulas, resource links, and other chemical information".
- ^ a b c Lutwak-Mann C, Adams CE (April 1957). "Carbonic anhydrase in the female reproductive tract. II. Endometrial carbonic anhydrase as indicator of luteoid potency: correlation with progestational proliferation". J. Endocrinol. 15 (1): 43–55. doi:10.1677/joe.0.0150043. PMID 13439082.
- ^ a b Pincus G, Miyake T, Merrill AP, Longo P (November 1957). "The bioassay of progesterone". Endocrinology. 61 (5): 528–33. doi:10.1210/endo-61-5-528. PMID 13480263.
- ^ Ralph I. Dorfman (3 February 2016). Bioassay. Elsevier. pp. 153–. ISBN 978-1-4832-7276-4.
- ^ Rao, P. N., & Edwards, B. E. (1967). U.S. Patent No. 3,321,495. Washington, DC: U.S. Patent and Trademark Office.
- ^ Gaunt R, Steinetz BG, Chart JJ (1968). "Pharmacologic alteration of steroid hormone functions". Clin. Pharmacol. Ther. 9 (5): 657–81. doi:10.1002/cpt196895657. PMID 4175595. S2CID 38695246.
An interesting substance which has received little attention is the 3-cyclopentyl enol ether of progesterone (quingestrone). [...]
- ^ Junkermann H, Runnebaum B, Lisboa BP (July 1977). "New progesterone metabolites in human myometrium". Steroids. 30 (1): 1–14. doi:10.1016/0039-128X(77)90131-3. PMID 919010. S2CID 28420255.
In the Clauberg bioassay the 3β-hydroxy-4-pregnen-20-one shows about the same potency as progesterone (34). In regard to the biological activity of the 3α epimer no data are available.
- ^ Junkmann, Karl (1954). "Gestagens of prolonged action". Naunyn-Schmiedebergs Archiv für Pharmakologie und Experimentelle Pathologie. 223: 244–53. ISSN 0365-5423.
Among a large no. of pregnane derivs. the esters of 17-α-hydroxyprogesterone (I), itself of weak lutein hormone action, have a strong and long-lasting gestagen action. The optimal results are obtained with I caproate. It permits the administration of depot doses in clear solns. Within the range of dosage used no androgenic effect was noted. It has no influence on growth and on the secondary sex characteristics in infantile and adult castrate male rats. The 3-enol esters of progesterone, which have a somewhat prolonged action, are less suited for depot administration because of their oxidizability.
