Zanoterone
 | |
| Clinical data | |
|---|---|
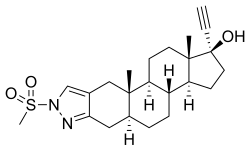
| Other names | WIN-49596; (5α,17α)-1'-(methylsulfonyl)-1'-H-pregn-20-yno[3,2-c]pyrazol-17-ol |
| Routes of administration | By mouth |
| Drug class | Steroidal antiandrogen |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
| Formula | C23H32N2O3S |
| Molar mass | 416.58 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Zanoterone (INN, USAN) (former developmental code name WIN-49596), also known as (5α,17α)-1'-(methylsulfonyl)-1'-H-pregn-20-yno[3,2-c]pyrazol-17-ol,[1] is a steroidal antiandrogen which was never marketed.[2][3][4] It was investigated for the treatment of benign prostatic hyperplasia (BPH) but failed to demonstrate sufficient efficacy in phase II clinical trials, and also showed an unacceptable incidence rate and severity of side effects (e.g., breast pain and gynecomastia).[4][5] As such, it was not further developed.[4][5]
Zanoterone was derived from 5α-dihydroethisterone (5α-dihydro-17α-ethynyltestosterone).[6][7] It is an antagonist of the androgen receptor (Ki = 2.2 μM; RBA compared to metribolone = 2.2%), and with the exception of antiprogestogenic activity in rat and rabbit models, is devoid of other hormonal activities.[6][8] Zanoterone does not inhibit 5α-reductase, aromatase, or 3α- or 3β-hydroxysteroid dehydrogenase in vitro.[6] The drug significantly increases testosterone and estradiol levels in men.[9] Zanoterone has been found to not significantly inhibit mating performance or fertility in adult male rats at high dosages for an extended period of time.[6] It has been found to act as an inducer of the enzyme CYP3A4 in vivo in rats.[10]
| Antiandrogen | Relative potency |
|---|---|
| Bicalutamide | 4.3 |
| Hydroxyflutamide | 3.5 |
| Flutamide | 3.3 |
| Cyproterone acetate | 1.0 |
| Zanoterone | 0.4 |
| Description: Relative potencies of orally administered antiandrogens in antagonizing 0.8 to 1.0 mg/kg s.c. testosterone propionate-induced ventral prostate weight increase in castrated immature male rats. Higher values mean greater potency. Sources: See template. | |
See also[edit]
References[edit]
- ^ William Andrew Publishing (22 October 2013). Pharmaceutical Manufacturing Encyclopedia, 3rd Edition. Elsevier. pp. 3517–3518. ISBN 978-0-8155-1856-3.
- ^ Morton IK, Hall JM (31 October 1999). Concise Dictionary of Pharmacological Agents: Properties and Synonyms. Springer Science & Business Media. pp. 294–. ISBN 978-0-7514-0499-9.
- ^ Ganellin CR, Triggle DJ (1997). Dictionary of Pharmacological Agents. Taylor & Francis. pp. 540–. ISBN 978-0-412-46630-4.
- ^ a b c Schröder FH, Radlmaier A (2009). "Steroidal Antiandrogens". In Jordan VC, Furr BJ (eds.). Hormone Therapy in Breast and Prostate Cancer. Cancer Drug Discovery and Development. Humana Press. pp. 325–346. doi:10.1007/978-1-59259-152-7_15. ISBN 978-1-60761-471-5.
- ^ a b Wein AJ, Kavoussi LR, Novick AC, Partin AW, Peters CA (28 September 2011). Campbell-Walsh Urology. Elsevier Health Sciences. pp. 2637–. ISBN 978-1-4557-2298-3.
- ^ a b c d Mallamo JP, Juniewicz PE (8 September 1989). "New horizons in the treatment of proliferative prostatic disease.". Annual Reports in Medicinal Chemistry. Academic Press. pp. 197-206 (200). ISBN 978-0-08-058368-6.
- ^ Lednicer D, Mitscher LA (5 November 1998). The Organic Chemistry of Drug Synthesis. John Wiley & Sons. p. 65. ISBN 978-0-471-24510-0.
- ^ Winneker RC, Wagner MM, Batzold FH (December 1989). "Studies on the mechanism of action of Win 49596: a steroidal androgen receptor antagonist". Journal of Steroid Biochemistry. 33 (6): 1133–1138. doi:10.1016/0022-4731(89)90420-2. PMID 2615358.
- ^ Berger BM, Naadimuthu A, Boddy A, Fisher HA, McConnell JD, Milam D, et al. (September 1995). "The effect of zanoterone, a steroidal androgen receptor antagonist, in men with benign prostatic hyperplasia. The Zanoterone Study Group". The Journal of Urology. 154 (3): 1060–1064. doi:10.1016/S0022-5347(01)66976-3. PMID 7543598.
- ^ Roberts AE, Ritz MA, Hoekstra S, Descotes G, Hincks JR (1996). "Induction of liver cytochrome P-450 (CYP) 3A in male and female rats by a steroidal androgen receptor antagonist, zanoterone". Journal of Biochemical Toxicology. 11 (3): 101–110. doi:10.1002/(SICI)1522-7146(1996)11:3<101::AID-JBT1>3.0.CO;2-O. PMID 9029268.
