Citalopram
 (R)-(−)-citalopram (top), (S)-(+)-citalopram (bottom) | |
| Clinical data | |
|---|---|
| Pronunciation | /saɪˈtæləˌpræm, sɪ-/;[1] |
| Trade names | Celexa, Cipramil, others[2] |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a699001 |
| License data |
|
| Pregnancy category |
|
| Routes of administration | oral, intravenous[4][5][6] |
| Drug class | Selective serotonin reuptake inhibitor (SSRI)[7] |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 80% peak at 4 hours[7] |
| Protein binding | <80%[10] |
| Metabolism | Liver (CYP3A4 and CYP2C19) |
| Metabolites | Desmethylcitalopram (DCT) and didesmethylcitalopram (DDCT) |
| Elimination half-life | 35 hours |
| Excretion | Mostly as unmetabolized citalopram, partly DCT and traces of DDCT in urine |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.056.247 |
| Chemical and physical data | |
| Formula | C20H21FN2O |
| Molar mass | 324.399 g·mol−1 |
| 3D model (JSmol) | |
| Chirality | Racemic mixture |
| |
| |
| (verify) | |
Citalopram, sold under the brand name Celexa among others, is an antidepressant of the selective serotonin reuptake inhibitor (SSRI) class.[7][10] It is used to treat major depressive disorder, obsessive compulsive disorder, panic disorder, and social phobia.[7] The antidepressant effects may take one to four weeks to occur.[7] It is typically taken orally (swallowed by mouth).[7][10] In some European countries, it is sometimes given intravenously (injected into a vein) to initiate treatment, before switching to the oral route of administration for continuation of treatment.[4] It has also been used intravenously in other parts of the world in some other circumstances.[5][6]
Common side effects include nausea, trouble sleeping, sexual problems, shakiness, feeling tired, and sweating.[7] Serious side effects include an increased risk of suicide in those under the age of 25, serotonin syndrome, glaucoma, and QT prolongation.[7] It should not be used in persons who take or have recently taken an MAO inhibitor.[7] There are concerns that use during pregnancy may harm the fetus.[3]
Citalopram was approved for medical use in the United States in 1998.[7] It is on the World Health Organization's List of Essential Medicines.[11] It is available as a generic medication.[12] In 2021, it was the 31st most commonly prescribed medication in the United States, with more than 18 million prescriptions.[13][14]
Medical uses[edit]

Depression[edit]
In the United States, citalopram is approved to treat major depressive disorder.[15] Citalopram appears to have comparable efficacy and superior tolerability relative to other antidepressants.[16][17] In the National Institute for Health and Clinical Excellence ranking of ten antidepressants for efficacy and cost-effectiveness, citalopram is fifth in effectiveness (after mirtazapine, escitalopram, venlafaxine, and sertraline) and fourth in cost-effectiveness.[18]
Evidence for effectiveness of citalopram for treating depression in children is uncertain.[19][20]
Panic disorder[edit]
Citalopram is licensed in the UK[21] and other European countries[22] for panic disorder, with or without agoraphobia.
Other[edit]
Citalopram may be used off-label to treat anxiety, and dysthymia,[23] premenstrual dysphoric disorder, body dysmorphic disorder, and obsessive–compulsive disorder.[24]
It appears to be as effective as fluvoxamine and paroxetine in obsessive–compulsive disorder.[25] Some data suggest the effectiveness of intravenous infusion of citalopram in resistant OCD.[5] Citalopram is well tolerated and as effective as moclobemide in social anxiety disorder.[26] There are studies suggesting that citalopram can be useful in reducing aggressive and impulsive behavior.[27][28] It appears to be superior to placebo for behavioural disturbances associated with dementia.[29] It has also been used successfully for hypersexuality in early Alzheimer's disease.[30]
A meta-analysis, including studies with fluoxetine, paroxetine, sertraline, escitalopram, and citalopram versus placebo, showed SSRIs to be effective in reducing symptoms of premenstrual syndrome, whether taken continuously or just in the luteal phase.[31] For alcoholism, citalopram has produced a modest reduction in alcoholic drink intake and increase in drink-free days in studies of alcoholics, possibly by decreasing desire or reducing the reward.[32]
While on its own citalopram is less effective than amitriptyline in the prevention of migraines, in refractory cases, combination therapy may be more effective.[33]
Citalopram and other SSRIs can be used to treat hot flashes.[34]: 107
A 2009 multisite randomized controlled study found no benefit and some adverse effects in autistic children from citalopram, raising doubts whether SSRIs are effective for treating repetitive behavior in children with autism.[35]
Some research suggests citalopram interacts with cannabinoid protein-couplings in the rat brain, and this is put forward as a potential cause of some of the drug's antidepressant effect.[36]
Administration[edit]
Citalopram is typically taken in one dose, either in the morning or evening. It can be taken with or without food. Its absorption does not increase when taken with food,[37][10] but doing so can help prevent nausea. Nausea is often caused when the 5-HT3 receptors actively absorb free serotonin, as this receptor is present within the digestive tract.[38]
Adverse effects[edit]
Citalopram theoretically causes side effects by increasing the concentration of serotonin in other parts of the body (e.g., the intestines). Other side effects, such as increased apathy and emotional flattening, may be caused by the decrease in dopamine release associated with increased serotonin. Citalopram is also a mild antihistamine, which may be responsible for some of its sedating properties.[34]: 104
Other common side effects of citalopram include drowsiness, insomnia, nausea, weight changes (usually weight gain), increase in appetite, vivid dreaming, frequent urination, dry mouth,[37] increased sweating, trembling, diarrhea, excessive yawning, severe tinnitus, and fatigue. Less common side effects include bruxism, vomiting, cardiac arrhythmia, blood pressure changes, dilated pupils, anxiety, mood swings, headache, hyperactivity and dizziness. Rare side effects include convulsions, hallucinations, severe allergic reactions and photosensitivity.[37] If sedation occurs, the dose may be taken at bedtime rather than in the morning. Some data suggests citalopram may cause nightmares.[39] Citalopram is associated with a higher risk of arrhythmia than other SSRIs.[40][41]
Citalopram and other SSRIs can induce a mixed state, especially in those with undiagnosed bipolar disorder.[34]: 105 According to an article published in 2020, one of the other rare side effects of Citalopram could be triggering visual snow syndrome; which does not resolve after the discontinuation of the medicine.[42]
Sexual dysfunction[edit]
Sexual dysfunction is often a side effect with SSRIs.[43] Some people experience persistent sexual side effects when taking SSRIs or after discontinuing them.[44] Symptoms of medication-induced sexual dysfunction from antidepressants include difficulty with orgasm, erection, or ejaculation.[44] Other symptoms may be genital anesthesia, anhedonia, decreased libido, vaginal lubrication issues, and nipple insensitivity in women. Rates are unknown, and there is no established treatment.[45]
Abnormal heart rhythm[edit]
In August 2011, the FDA announced, "Citalopram causes dose-dependent QT interval prolongation. Citalopram should no longer be prescribed at doses greater than 40 mg per day".[46] A further clarification, issued in March 2012, restricted the maximum dose to 20 mg for subgroups of patients, including those older than 60 years and those taking an inhibitor of cytochrome P450 2C19.7.[47]
Endocrine effects[edit]
As with other SSRIs, citalopram can cause an increase in serum prolactin level.[48] Citalopram has no significant effect on insulin sensitivity in women of reproductive age[49] and no changes in glycaemic control were seen in another trial.[50]
Exposure in pregnancy[edit]
Antidepressant exposure (including citalopram) during pregnancy is associated with shorter duration of gestation (by three days), increased risk of preterm delivery (by 55%), lower birth weight (by 75 g), and lower Apgar scores (by <0.4 points). Antidepressant exposure is not associated with an increased risk of spontaneous abortion.[51] It is uncertain whether there is an increased prevalence of septal heart defects among children whose mothers were prescribed an SSRI in early pregnancy.[52][53]
Overdose[edit]
Overdosage may result in vomiting, sedation, disturbances in heart rhythm, dizziness, sweating, nausea, tremor, and rarely amnesia, confusion, coma, or convulsions.[34]: 105 Overdose deaths have occurred, sometimes involving other drugs, but also with citalopram as the sole agent. Citalopram and N-desmethylcitalopram may be quantified in blood or plasma to confirm a diagnosis of poisoning in hospitalized patients or to assist in a medicolegal death investigation. Blood or plasma citalopram concentrations are usually in a range of 50-400 μg/L in persons receiving the drug therapeutically, 1000–3000 μg/L in patients who survive acute overdosage and 3–30 mg/L in those who do not survive.[46][54][55] It is the most dangerous of SSRIs in overdose.[56]
Suicidality[edit]
In the United States, citalopram carries a boxed warning stating it may increase suicidal thinking and behavior in those under age 24.[37]
Discontinuation Syndrome[edit]
SSRI discontinuation syndrome has been reported when treatment is stopped. It includes sensory, gastrointestinal symptoms, dizziness, lethargy, and sleep disturbances, as well as psychological symptoms such as anxiety/agitation, irritability, and poor concentration.[57] Electric shock-like sensations are typical for SSRI discontinuation.[58] Withdrawal symptoms can occur when this medicine is suddenly stopped, such as paraesthesiae, sleeping problems (difficulty sleeping and intense dreams), feeling dizzy, agitated or anxious, nausea, vomiting, tremors, confusion, sweating, headache, diarrhea, palpitations, changes in emotions, irritability, and eye or eyesight problems. Treatment with citalopram should be reduced gradually when treatment is finished.[59]
Interactions[edit]
Serotonin Syndrome[edit]
Citalopram should not be taken with St John's wort, tryptophan or 5-HTP as the resulting drug interaction could lead to serotonin syndrome.[60] With St John's wort, this may be caused by compounds in the plant extract reducing the efficacy of the hepatic cytochrome P450 enzymes that process citalopram.[61] Tryptophan and 5-HTP are precursors to serotonin.[62] When taken with an SSRI, such as citalopram, this can lead to levels of serotonin that can be lethal. This may also be the case when SSRIs are taken with SRAs (serotonin releasing agents) such as in the case of MDMA. It is possible that SSRIs could reduce the effects associated due to an SRA, since SSRIs stop the reuptake of Serotonin by blocking SERT. This would allow less serotonin in and out of the transporters, thus decreasing the likelihood of neurotoxic effects. However, these concerns are still disputed as the exact pharmacodynamic effects of citalopram and MDMA have yet to be fully identified.[citation needed] Citalopram is contraindicated in individuals taking MAOIs, owing to a potential for serotonin syndrome.
Other interactions[edit]
SSRIs, including citalopram, can increase the risk of bleeding, especially when coupled with aspirin, NSAIDs, warfarin, or other anticoagulants.[37] Taking citalopram with omeprazole may cause higher blood levels of citalopram. This is a potentially dangerous interaction, so dosage adjustments may be needed or alternatives may be prescribed.[63][64][10]
Pharmacokinetics[edit]
Citalopram is considered safe and well tolerated in the therapeutic dose range. Distinct from some other agents in its class, it exhibits linear pharmacokinetics and minimal drug interaction potential, making it a better choice for the elderly or comorbid patients.[65]
Stereochemistry[edit]
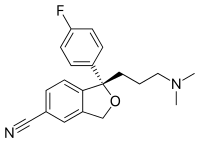
Citalopram has one stereocenter, to which a 4-fluoro phenyl group and an N, N-dimethyl-3-aminopropyl group bind. As a result of this chirality, the molecule exists in (two) enantiomeric forms (mirror images). They are termed S-(+)-citalopram and R-(–)-citalopram.

|

|
|

|
| (S)-(+)-citalopram | (R)-(–)-citalopram |
Citalopram is sold as a racemic mixture, consisting of 50% (R)-(−)-citalopram and 50% (S)-(+)-citalopram. Only the (S)-(+) enantiomer has the desired antidepressant effect.[66] Lundbeck now markets the (S)-(+) enantiomer, the generic name of which is escitalopram. Whereas citalopram is supplied as the hydrobromide, escitalopram is sold as the oxalate salt (hydrooxalate).[37] In both cases, the salt forms of the amine make these otherwise lipophilic compounds water-soluble.
Metabolism[edit]
Citalopram is metabolized in the liver mostly by CYP2C19, but also by CYP3A4 and CYP2D6. Metabolites desmethylcitalopram and didesmethylcitalopram are significantly less energetic and their contribution to the overall action of citalopram is negligible. The half-life of citalopram is about 35 hours. Approximately 80% is cleared by the liver and 20% by the kidneys.[67] The elimination process is slower in the elderly and in patients with liver or kidney failure. With once-daily dosing, steady plasma concentrations are achieved in about a week. Potent inhibitors of CYP2C19 and 3A4 might decrease citalopram clearance.[10] Tobacco smoke exposure was found to inhibit the biotransformation of citalopram in animals, suggesting that the elimination rate of citalopram is decreased after tobacco smoke exposure. After intragastric administration, the half-life of the racemic mixture of citalopram was increased by about 287%.[68]

| Receptor | Ki (nM) |
|---|---|
| SERT | 1.6 |
| NET | 6190 |
| 5-HT2C | 617 |
| α1 |
1211 |
| M1 | 1430 |
| H1 | 283 |
History[edit]
Citalopram was first synthesized in 1972 by chemist Klaus Bøgesø[73] and his research group at the pharmaceutical company Lundbeck and was first marketed in 1989 in Denmark. It was first marketed in the US in 1998.[74] The original patent expired in 2003, allowing other companies to legally produce and market generic versions.
Society and culture[edit]
Brand names[edit]
Citalopram is sold under these brand names:
- Akarin (Denmark, Nycomed)
- C Pram S (India)
- Celapram (Australia,[75] New Zealand),
- Celexa (U.S. and Canada, Forest Laboratories, Inc.)
- Celica (Australia)[75]
- Ciazil (Australia,[75] New Zealand)
- Cilate (South Africa)
- Cilift (South Africa)
- Cimal (South America, by Roemmers and Recalcine)
- Cipralex (Europe, South Africa)
- Cipram (Denmark, Turkey, H. Lundbeck A/S)
- Cipramil (Australia,[75] Brazil, Belgium, Chile, Finland, Germany, Netherlands, Iceland, Ireland, Israel, New Zealand, Norway, Russia, South Africa, Sweden, United Kingdom)
- Cipraned, Cinapen (Greece)
- Ciprapine (Ireland)
- Ciprotan (Ireland)
- Citabax, Citaxin (Poland)
- Cital (Poland)
- Citalec (Czech Republic, Slovakia)
- Citalex (Iran, Serbia)
- Citalo (Australia,[75] Egypt, Pakistan)
- Citalopram (Canada, Denmark, Finland, Germany, Ireland, New Zealand, Spain, Sweden, Switzerland, United Kingdom, U.S.)
- Citol (Russia, Turkey)
- Citox (Mexico)
- Citrol (Europe and Australia)[75]
- Citta (Brazil)
- Dalsan (Eastern Europe)
- Denyl (Brazil)
- Elopram (Italy)
- Estar (Pakistan)
- Humorup (Argentina)
- Humorap (Peru, Bolivia)
- Lopraxer (Greece)[76]
- Oropram (Iceland, Actavis),
- Opra (Russia)
- Pram (Russia)
- Pramcit (Pakistan)
- Procimax (Brazil)
- Recital (Israel, Thrima Inc. for Unipharm Ltd.)
- Sepram (Finland)
- Seropram (various European countries, including Czech Republic)
- Szetalo (India)
- Talam (Europe and Australia)[75]
- Temperax (Argentina, Chile, Peru)
- Vodelax (Turkey)
- Zentius (South America, by Roemmers and Recalcine)
- Zetalo (India)
- Cipratal (Kuwait, GCC)
- Zylotex (Portugal)[2]
European Commission fine[edit]
On 19 June 2013, the European Commission imposed a fine of €93.8 million on the Danish pharmaceutical company Lundbeck, plus a total of €52.2 million on several generic pharmaceutical-producing companies. This was in response to Lundbeck entering an agreement with the companies to delay their sales of generic citalopram after Lundbeck's patent on the drug had expired, thus reducing competition in breach of European antitrust law.[77]
Other uses[edit]
Citalopram is also a parasiticide.[78] Schistosomula have high mortality when treated with citalopram.[78]
See also[edit]
References[edit]
- ^ "Citalopram". Merriam-Webster.com Dictionary.
- ^ a b "Citalopram". International. Drugs.com.
- ^ a b "Citalopram (Celexa) Use During Pregnancy". Drugs.com. Retrieved 23 December 2018.
- ^ a b Kasper S, Müller-Spahn F (June 2002). "Intravenous antidepressant treatment: focus on citalopram". European Archives of Psychiatry and Clinical Neuroscience. 252 (3): 105–109. doi:10.1007/s00406-002-0363-8. PMID 12192466. S2CID 24991131.
- ^ a b c Pallanti S, Quercioli L, Koran LM (September 2002). "Citalopram intravenous infusion in resistant obsessive-compulsive disorder: an open trial". The Journal of Clinical Psychiatry. 63 (9): 796–801. doi:10.4088/JCP.v63n0908. PMID 12363120.
- ^ a b Altamura AC, Dell'Osso B, Buoli M, Zanoni S, Mundo E (August 2008). "Intravenous augmentative citalopram versus clomipramine in partial/nonresponder depressed patients: a short-term, low dose, randomized, placebo-controlled study". J Clin Psychopharmacol. 28 (4): 406–410. doi:10.1097/JCP.0b013e31817d5931. PMID 18626267. S2CID 25013120. Retrieved 12 August 2023.
- ^ a b c d e f g h i j "Citalopram Hydrobromide Monograph for Professionals". Drugs.com. AHFS. Retrieved 23 December 2018.
- ^ "FDA-sourced list of all drugs with black box warnings (Use Download Full Results and View Query links.)". nctr-crs.fda.gov. FDA. Retrieved 22 October 2023.
- ^ Anvisa (31 March 2023). "RDC Nº 784 - Listas de Substâncias Entorpecentes, Psicotrópicas, Precursoras e Outras sob Controle Especial" [Collegiate Board Resolution No. 784 - Lists of Narcotic, Psychotropic, Precursor, and Other Substances under Special Control] (in Brazilian Portuguese). Diário Oficial da União (published 4 April 2023). Archived from the original on 3 August 2023. Retrieved 16 August 2023.
- ^ a b c d e f g "Celexa- citalopram tablet, film coated". DailyMed. 15 August 2019. Retrieved 28 October 2020.
- ^ World Health Organization (2021). World Health Organization model list of essential medicines: 22nd list (2021). Geneva: World Health Organization. hdl:10665/345533. WHO/MHP/HPS/EML/2021.02.
- ^ British national formulary : BNF 76 (76 ed.). Pharmaceutical Press. 2018. p. 361. ISBN 9780857113382.
- ^ "The Top 300 of 2021". ClinCalc. Archived from the original on 15 January 2024. Retrieved 14 January 2024.
- ^ "Citalopram Drug Usage Statistics". ClinCalc. Retrieved 14 January 2024.
- ^ Sharbaf Shoar N, Fariba KA, Padhy RK (2020). "Citalopram". StatPearls. Treasure Island (FL): StatPearls Publishing. PMID 29489221. Retrieved 23 October 2020.
- ^ Cipriani A, Furukawa TA, Salanti G, Chaimani A, Atkinson LZ, Ogawa Y, et al. (April 2018). "Comparative efficacy and acceptability of 21 antidepressant drugs for the acute treatment of adults with major depressive disorder: a systematic review and network meta-analysis". Lancet. 391 (10128): 1357–1366. doi:10.1016/S0140-6736(17)32802-7. PMC 5889788. PMID 29477251.
- ^ Khoo AL, Zhou HJ, Teng M, Lin L, Zhao YJ, Soh LB, et al. (August 2015). "Network Meta-Analysis and Cost-Effectiveness Analysis of New Generation Antidepressants". CNS Drugs. 29 (8): 695–712. doi:10.1007/s40263-015-0267-6. PMID 26293743. S2CID 207486435.
- ^ See p410 of "National Clinical Practice Guideline 90. Depression: The treatment and management of depression in adults, updated edition (2010)". National Institute for Health and Clinical Excellence (UK). Archived from the original on 15 December 2017. Retrieved 27 November 2016.
- ^ Cohen D (2007). "Should the use of selective serotonin reuptake inhibitors in child and adolescent depression be banned?". Psychotherapy and Psychosomatics. 76 (1): 5–14. doi:10.1159/000096360. PMID 17170559. S2CID 1112192.
- ^ Carandang C, Jabbal R, Macbride A, Elbe D (November 2011). "A review of escitalopram and citalopram in child and adolescent depression". Journal of the Canadian Academy of Child and Adolescent Psychiatry. 20 (4): 315–324. PMC 3222577. PMID 22114615.
- ^ "Citalopram 20mg Tablets - Summary of Product Characteristics (SmPC) - (emc)". www.medicines.org.uk. Retrieved 15 January 2023.
- ^ Urząd Rejestracji Produktów Leczniczych, Wyrobów Medycznych i Produktów Biobójczych (Office for Registration of Medicinal Products, Medical Devices and Biocides) "Charakterystyka Produktu Leczniczego" [Characteristic Product Leczniczego] (PDF). The Office for Registration of Medicinal Products, Medical Devices and Biocidal Products (in Polish). Archived from the original (PDF) on 5 November 2013. Retrieved 24 September 2013.
- ^ Hellerstein DJ, Batchelder S, Miozzo R, Kreditor D, Hyler S, Gangure D, et al. (May 2004). "Citalopram in the treatment of dysthymic disorder". International Clinical Psychopharmacology. 19 (3): 143–148. doi:10.1097/00004850-200405000-00004. PMID 15107656. S2CID 24416470.
- ^ Pittenger C, Bloch MH (September 2014). "Pharmacological treatment of obsessive-compulsive disorder". The Psychiatric Clinics of North America. 37 (3): 375–391. doi:10.1016/j.psc.2014.05.006. PMC 4143776. PMID 25150568.
- ^ Stein DJ, Montgomery SA, Kasper S, Tanghoj P (November 2001). "Predictors of response to pharmacotherapy with citalopram in obsessive-compulsive disorder". International Clinical Psychopharmacology. 16 (6): 357–361. doi:10.1097/00004850-200111000-00007. PMID 11712625. S2CID 38416051.
- ^ Atmaca M, Kuloglu M, Tezcan E, Unal A (December 2002). "Efficacy of citalopram and moclobemide in patients with social phobia: some preliminary findings". Human Psychopharmacology. 17 (8): 401–405. doi:10.1002/hup.436. PMID 12457375. S2CID 34395742.
- ^ Armenteros JL, Lewis JE (May 2002). "Citalopram treatment for impulsive aggression in children and adolescents: an open pilot study". Journal of the American Academy of Child and Adolescent Psychiatry. 41 (5): 522–529. doi:10.1097/00004583-200205000-00009. PMID 12014784.
- ^ Reist C, Nakamura K, Sagart E, Sokolski KN, Fujimoto KA (January 2003). "Impulsive aggressive behavior: open-label treatment with citalopram". The Journal of Clinical Psychiatry. 64 (1): 81–85. doi:10.4088/jcp.v64n0115. PMID 12590628.
- ^ Pollock BG, Mulsant BH, Rosen J, Sweet RA, Mazumdar S, Bharucha A, et al. (March 2002). "Comparison of citalopram, perphenazine, and placebo for the acute treatment of psychosis and behavioral disturbances in hospitalized, demented patients". The American Journal of Psychiatry. 159 (3): 460–465. doi:10.1176/appi.ajp.159.3.460. PMID 11870012.
- ^ Tosto G, Talarico G, Lenzi GL, Bruno G (September 2008). "Effect of citalopram in treating hypersexuality in an Alzheimer's disease case". Neurological Sciences. 29 (4): 269–270. doi:10.1007/s10072-008-0979-1. hdl:11573/22581. PMID 18810603. S2CID 11432563.
- ^ Marjoribanks J, Brown J, O'Brien PM, Wyatt K (June 2013). "Selective serotonin reuptake inhibitors for premenstrual syndrome". The Cochrane Database of Systematic Reviews. 2013 (6): CD001396. doi:10.1002/14651858.CD001396.pub3. PMC 7073417. PMID 23744611.
- ^ Tiihonen J, Ryynänen OP, Kauhanen J, Hakola HP, Salaspuro M (January 1996). "Citalopram in the treatment of alcoholism: a double-blind placebo-controlled study". Pharmacopsychiatry. 29 (1): 27–29. doi:10.1055/s-2007-979538. PMID 8852531. S2CID 260242756.
- ^ Rampello L, Alvano A, Chiechio S, Malaguarnera M, Raffaele R, Vecchio I, et al. (2004). "Evaluation of the prophylactic efficacy of amitriptyline and citalopram, alone or in combination, in patients with comorbidity of depression, migraine, and tension-type headache". Neuropsychobiology. 50 (4): 322–328. doi:10.1159/000080960. PMID 15539864. S2CID 46362166.
- ^ a b c d Stahl SM (2011). The Prescriber's Guide (Stahl's Essential Psychopharmacology). Cambridge, UK: Cambridge University Press. ISBN 978-0-521-17364-3.
- ^ King BH, Hollander E, Sikich L, McCracken JT, Scahill L, Bregman JD, et al. (June 2009). "Lack of efficacy of citalopram in children with autism spectrum disorders and high levels of repetitive behavior: citalopram ineffective in children with autism". Archives of General Psychiatry. 66 (6): 583–590. doi:10.1001/archgenpsychiatry.2009.30. PMC 4112556. PMID 19487623.
- Lay summary in: Kaplan K (2 June 2009). "Study finds antidepressant doesn't help autistic children". Los Angeles Times.
- ^ Hesketh SA, Brennan AK, Jessop DS, Finn DP (May 2008). "Effects of chronic treatment with citalopram on cannabinoid and opioid receptor-mediated G-protein coupling in discrete rat brain regions". Psychopharmacology. 198 (1): 29–36. doi:10.1007/s00213-007-1033-3. PMID 18084745. S2CID 23357324.
- ^ a b c d e f "Celexa (citalopram hydrobromide) Tablets/Oral Solution" (PDF). Prescribing Information. Forest Laboratories, Inc.
- ^ Rang HP (2003). Pharmacology. Edinburgh: Churchill Livingstone. p. 187. ISBN 978-0-443-07145-4.
- ^ Arora G, Sandhu G, Fleser C (Spring 2012). "Citalopram and nightmares". The Journal of Neuropsychiatry and Clinical Neurosciences. 24 (2): E43. doi:10.1176/appi.neuropsych.11040096. PMID 22772700.
- ^ Qirjazi E, McArthur E, Nash DM, Dixon SN, Weir MA, Vasudev A, et al. (11 August 2016). "Risk of Ventricular Arrhythmia with Citalopram and Escitalopram: A Population-Based Study". PLOS ONE. 11 (8): e0160768. Bibcode:2016PLoSO..1160768Q. doi:10.1371/journal.pone.0160768. PMC 4981428. PMID 27513855.
- ^ Girardin FR, Gex-Fabry M, Berney P, Shah D, Gaspoz JM, Dayer P (December 2013). "Drug-induced long QT in adult psychiatric inpatients: the 5-year cross-sectional ECG Screening Outcome in Psychiatry study". The American Journal of Psychiatry. 170 (12): 1468–1476. doi:10.1176/appi.ajp.2013.12060860. PMID 24306340.
- ^ Eren, O.E., Schöberl, F., Schankin, C.J. et al. Visual snow syndrome after start of citalopram—novel insights into underlying pathophysiology. Eur J Clin Pharmacol 77, 271–272 (2021). https://doi.org/10.1007/s00228-020-02996-9
- ^ "Side effects of citalopram". nhs.uk. 15 February 2022. Retrieved 19 December 2022.
- ^ a b American Psychiatric Association (2013). Diagnostic and Statistical Manual of Mental Disorders (5th ed.). Arlington, VA: American Psychiatric Publishing. pp. 446–449. ISBN 9780890425558.
- ^ Bala A, Nguyen HM, Hellstrom WJ (January 2018). "Post-SSRI Sexual Dysfunction: A Literature Review". Sexual Medicine Reviews. 6 (1): 29–34. doi:10.1016/j.sxmr.2017.07.002. PMID 28778697.
- ^ a b "Abnormal heart rhythms associated with high doses of Celexa (citalopram hydrobromide)". Safety Communication. United States Food and Drug Administration. 24 August 2011.
- ^ "Revised recommendations for Celexa". U.S. Food and Drug Administration (FDA). 28 March 2012. Retrieved 28 October 2020.
- ^ Trenque T, Herlem E, Auriche P, Dramé M (December 2011). "Serotonin reuptake inhibitors and hyperprolactinaemia: a case/non-case study in the French pharmacovigilance database". Drug Safety. 34 (12): 1161–1166. doi:10.2165/11595660-000000000-00000. PMID 22077504. S2CID 25532853.
- ^ Kauffman RP, Castracane VD, White DL, Baldock SD, Owens R (September 2005). "Impact of the selective serotonin reuptake inhibitor citalopram on insulin sensitivity, leptin and basal cortisol secretion in depressed and non-depressed euglycemic women of reproductive age". Gynecological Endocrinology. 21 (3): 129–137. doi:10.1080/09513590500216800. PMID 16335904. S2CID 11286706.
- ^ Sindrup SH, Bjerre U, Dejgaard A, Brøsen K, Aaes-Jørgensen T, Gram LF (November 1992). "The selective serotonin reuptake inhibitor citalopram relieves the symptoms of diabetic neuropathy". Clinical Pharmacology and Therapeutics. 52 (5): 547–552. doi:10.1038/clpt.1992.183. PMID 1424428. S2CID 6730763.
- ^ Ross LE, Grigoriadis S, Mamisashvili L, Vonderporten EH, Roerecke M, Rehm J, et al. (April 2013). "Selected pregnancy and delivery outcomes after exposure to antidepressant medication: a systematic review and meta-analysis". JAMA Psychiatry. 70 (4): 436–443. doi:10.1001/jamapsychiatry.2013.684. PMID 23446732.
- ^ Pedersen LH, Henriksen TB, Vestergaard M, Olsen J, Bech BH (September 2009). "Selective serotonin reuptake inhibitors in pregnancy and congenital malformations: population based cohort study". BMJ. 339 (sep23 1): b3569. doi:10.1136/bmj.b3569. PMC 2749925. PMID 19776103.
- ^ Huybrechts KF, Palmsten K, Avorn J, Cohen LS, Holmes LB, Franklin JM, et al. (June 2014). "Antidepressant use in pregnancy and the risk of cardiac defects". The New England Journal of Medicine. 370 (25): 2397–2407. doi:10.1056/NEJMoa1312828. PMC 4062924. PMID 24941178.
- ^ Personne M, Sjöberg G, Persson H (1997). "Citalopram overdose--review of cases treated in Swedish hospitals". Journal of Toxicology. Clinical Toxicology. 35 (3): 237–240. doi:10.3109/15563659709001206. PMID 9140316.
- ^ Luchini D, Morabito G, Centini F (December 2005). "Case report of a fatal intoxication by citalopram". The American Journal of Forensic Medicine and Pathology. 26 (4): 352–354. doi:10.1097/01.paf.0000188276.33030.dd. PMID 16304470. S2CID 44840526.
- ^ Taylor D, Paton C, Kapur S (2012). The Maudsley Prescribing Guidelines in Psychiatry (Taylor, The Maudsley Prescribing Guidelines). Hoboken, NJ, USA: Wiley-Blackwell. p. 588. ISBN 9780470979693.
- ^ Warner CH, Bobo W, Warner C, Reid S, Rachal J (August 2006). "Antidepressant discontinuation syndrome". American Family Physician. 74 (3): 449–456. PMID 16913164.
- ^ Prakash O, Dhar V (June 2008). "Emergence of electric shock-like sensations on escitalopram discontinuation". Journal of Clinical Psychopharmacology. 28 (3): 359–360. doi:10.1097/JCP.0b013e3181727534. PMID 18480703.
- ^ "Citalopram withdrawal: What to expect". www.medicalnewstoday.com. 13 October 2022. Retrieved 19 December 2022.
- ^ Karch AM (2006). Lippincott's Nursing Drug Guide. Hagerstwon, MD: Lippincott Williams & Wilkins. ISBN 978-1-58255-436-5.
- ^ "Interactions with St John's wort preparations". Prescriber Update Articles. New Zealand Medicines and Medical Devices Safety Authority. 2000.
- ^ "The History of Tryptophan, Serotonin and 5-HTP". www.oxfordvitality.co.uk. 21 November 2016. Retrieved 22 July 2018.
- ^ "Drug interactions between Celexa and omeprazole". Drugs.com. Retrieved 28 January 2014.
- ^ "citalopram (Rx) - Celexa". Medscape. Retrieved 28 January 2014.
- ^ Keller MB (December 2000). "Citalopram therapy for depression: a review of 10 years of European experience and data from U.S. clinical trials". The Journal of Clinical Psychiatry. 61 (12): 896–908. doi:10.4088/JCP.v61n1202. PMID 11206593.
- ^ Lepola U, Wade A, Andersen HF (May 2004). "Do equivalent doses of escitalopram and citalopram have similar efficacy? A pooled analysis of two positive placebo-controlled studies in major depressive disorder". International Clinical Psychopharmacology. 19 (3): 149–155. doi:10.1097/00004850-200405000-00005. PMID 15107657. S2CID 36768144.
- ^ "Citalopram". DrugBank. 17 August 2016. Retrieved 12 February 2017.
- ^ Majcherczyk J, Kulza M, Senczuk-Przybylowska M, Florek E, Jawien W, Piekoszewski W (February 2012). "Influence of tobacco smoke on the pharmacokinetics of citalopram and its enantiomers". Journal of Physiology and Pharmacology. 63 (1): 95–100. PMID 22460466.
- ^ Sangkuhl K, Klein TE, Altman RB (November 2011). "PharmGKB summary: citalopram pharmacokinetics pathway". Pharmacogenetics and Genomics. 21 (11): 769–772. doi:10.1097/FPC.0b013e328346063f. PMC 3349993. PMID 21546862.
- ^ Hiemke C, Härtter S (January 2000). "Pharmacokinetics of selective serotonin reuptake inhibitors". Pharmacology & Therapeutics. 85 (1): 11–28. doi:10.1016/s0163-7258(99)00048-0. PMID 10674711.
- ^ Budău M, Hancu G, Rusu A, Muntean DL (January 2020). "Analytical methodologies for the enantiodetermination of citalopram and its metabolites". Chirality. 32 (1): 32–41. doi:10.1002/chir.23139. PMID 31702071. S2CID 207936622.
- ^ Owens JM, Knight DL, Nemeroff CB (July–August 2002). "[Second generation SSRIS: human monoamine transporter binding profile of escitalopram and R-fluoxetine]". L'Encéphale. 28 (4): 350–355. PMID 12232544.
- ^ Bøgesø KP, Sánchez C (December 2012). "The discovery of citalopram and its refinement to escitalopram.". In Fischer J, Ganellin CR, Rotella DP (eds.). Analogue-Based Drug Discovery. Vol. III. Weinheim, Germany: Wiley-VCH Verlag GmbH & Co. KGaA. pp. 269–94. doi:10.1002/9783527651085.ch11. ISBN 978-3-527-65108-5. Retrieved 23 October 2020.
- ^ Rawe B, May P (November 2009). "Citalopram: A new treatment for depression". Molecule of the Month. University of Bristol. doi:10.6084/m9.figshare.5255050. Retrieved 16 February 2015.
- ^ a b c d e f g "Pharmaceutical Benefits Scheme (PBS) - Citalopram". Australian Government Department of Health. Australian Government.
- ^ "Lopraxer". Drugs.com.
- ^ "Antitrust: Commission fines Lundbeck and other pharma companies for delaying market entry of generic medicines" (Press release). Brussels: European Union. 19 June 2013. Retrieved 20 June 2013.
- ^ a b Ribeiro P, Geary TG (2010). "Neuronal signaling in schistosomes: current status and prospects for postgenomics". Canadian Journal of Zoology. 88 (1). Canadian Science Publishing: 1–22. doi:10.1139/z09-126. ISSN 0008-4301.
