Naloxone
This article needs more reliable medical references for verification or relies too heavily on primary sources. (September 2023) |  |
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Narcan, Evzio, Nyxoid, others |
| Other names | EN-1530; N-Allylnoroxymorphone; 17-Allyl-4,5α-epoxy-3,14-dihydroxymorphinan-6-one, naloxone hydrochloride (USAN US) |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a612022 |
| License data | |
| Pregnancy category |
|
| Routes of administration | Nasal, intravenous, intramuscular |
| Drug class | Opioid antagonist |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 2% (by mouth, 90% absorption but high first-pass metabolism) 43–54% (intranasally) 98% (intramuscular, subcutaneous)[12][13] |
| Metabolism | Liver |
| Onset of action | 2 min (IV), 5 min (IM)[13] |
| Elimination half-life | 1–1.5 h |
| Duration of action | 30–60 min[13] |
| Excretion | Urine, bile |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.006.697 |
| Chemical and physical data | |
| Formula | C19H21NO4 |
| Molar mass | 327.380 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Naloxone is an opioid antagonist: a medication used to reverse or reduce the effects of opioids.[13] For example, it is used to restore breathing after an opioid overdose.[13] Effects begin within two minutes when given intravenously, five minutes when injected into a muscle,[13] and ten minutes as a nasal spray.[14] Naloxone blocks the effects of opioids for 30 to 90 minutes.[15]
Administration to opioid-dependent individuals may cause symptoms of opioid withdrawal, including restlessness, agitation, nausea, vomiting, a fast heart rate, and sweating.[13] To prevent this, small doses every few minutes can be given until the desired effect is reached.[13] In those with previous heart disease or taking medications that negatively affect the heart, further heart problems have occurred.[13] It appears to be safe in pregnancy, after having been given to a limited number of women.[16] Naloxone is a non-selective and competitive opioid receptor antagonist.[6][17] It works by reversing the depression of the central nervous system and respiratory system caused by opioids.[13]
Naloxone was patented in 1961 and approved for opioid overdose in the United States in 1971.[18][19] It is on the World Health Organization's List of Essential Medicines.[20] Naloxone is available as a generic medication.[13][21]
Medical uses[edit]
Opioid overdose[edit]
Naloxone is useful in treating both acute opioid overdose and respiratory or mental depression due to opioids.[13] Whether it is useful in those in cardiac arrest due to an opioid overdose is unclear.[22]
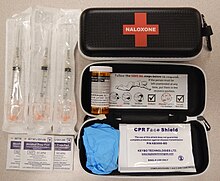
It is included as a part of emergency overdose response kits distributed to heroin and other opioid drug users, and to emergency responders. This has been shown to reduce rates of deaths due to overdose.[23] A prescription for naloxone is recommended if a person is on a high dose of opioid (>100 mg of morphine equivalence/day), is prescribed any dose of opioid accompanied by a benzodiazepine, or is suspected or known to use opioids nonmedically.[24] Prescribing naloxone should be accompanied by standard education that includes preventing, identifying, and responding to an overdose; rescue breathing; and calling emergency services.[25]
Distribution of naloxone to individuals likely to encounter people who overdose is one aspect of harm reduction strategies.[26]
However, with opioids that have longer half-lives, respiratory depression returns after naloxone has worn off; therefore, adequate dosing and continuous monitoring may be necessary.[27]
Symptoms of Opioid Overdose[edit]
Signs and symptoms of opioid overdose primarily stems from respiratory depression and it can lead to unconscious or death if the overdose is severe or not reversed in critical moments with naloxone administration. Symptoms can also include slowed breathing rate, blue-ish discolouration of the lips and/or fingernails and unresponsive from stimulation by those other people around them.[28] Even if the suspected individual is showing signs and symptoms of opioid overdose, you can still administer naloxone even if the suspected drug is not an opioid as it is deem to be safe and can only reverse opioid binding to its receptors.[28]
Clonidine overdose[edit]
Naloxone can also be used as an antidote in overdose of clonidine, a medication that lowers blood pressure.[29] Clonidine overdoses are of special relevance for children, in whom even small doses can cause significant harm.[30] However, there is controversy regarding naloxone's efficacy in treating the symptoms of clonidine overdose, namely slow heart rate, low blood pressure, and confusion/somnolence.[30] Case reports that used doses of 0.1 mg/kg (maximum of 2 mg/dose) repeated every 1–2 minutes (10 mg total dose) have shown inconsistent benefit.[30] As the doses used throughout the literature vary, it is difficult to form a conclusion regarding the benefit of naloxone in this setting.[31] The mechanism for naloxone's proposed benefit in clonidine overdose is unclear, but it has been suggested that endogenous opioid receptors mediate the sympathetic nervous system in the brain and elsewhere in the body.[31]
Preventing recreational opioid use[edit]
Naloxone is poorly absorbed when taken orally or sublingually, so it is often combined with a number of oral or sublingual opioid preparations, including buprenorphine and pentazocine, so that when swallowed or taken sublingually, only the non-naloxone opioid has an effect.[13][32] However, if the combination is injected (such as by dissolving a pill or sublingual strip in water), the naloxone is believed to block the effect of the other opioid.[13][32] This combination is used in an effort to prevent non-medical use.[32]
However, SAMHSA’s clinical guidelines state that if the combination of buprenorphine and naloxone is injected by a regular user of buprenorphine or buprenorphine/naloxone, then the buprenorphine would still produce an agonist effect but the naloxone would fail to produce an antagonist effect. This is because the amount of naloxone that would be required to block the buprenorphine after injection is much larger than the amount that is contained in buprenorphine/naloxone (Suboxone) pills and strips.[33] If someone who is not physically dependent on opioids were to inject the buprenorphine/naloxone combination, then the effects of the buprenorphine may at most be slightly lessened, but the individual would still be expected to experience euphoric effects.[33]
Other uses[edit]
A 2003 meta-analysis of existing research showed naloxone to improve blood flow in patients with shock, including septic, cardiogenic, hemorrhagic, or spinal shock, but could not determine if this reduced patient deaths.[34]
Special populations[edit]
Pregnancy and breastfeeding[edit]
Whether naloxone is excreted in breast milk is unknown, however, it is not orally bioavailable and therefore is unlikely to affect a breastfeeding infant.[35] Naloxone administration in pregnant individuals are still recommended in life-threatening opioid overdose despite the potential to cause maternal rapid-onset opioid withdrawal.[28]
Children[edit]
Naloxone can be used on infants who were exposed to intrauterine opiates administered to mothers during delivery. However, there is insufficient evidence for the use of naloxone to lower cardiorespiratory and neurological depression in these infants.[36] Infants exposed to high concentrations of opiates during pregnancy may have CNS damage in the setting of perinatal asphyxia. Naloxone has been studied to improve outcomes in this population, however the evidence is currently weak.[37][36]
Intravenous, intramuscular, or subcutaneous administration of naloxone can be given to children and neonates to reverse opiate effects. The American Academy of Pediatrics recommends only intravenous administration as the other two forms can cause unpredictable absorption. After a dose is given, the child should be monitored for at least 24 hours. For children with low blood pressure due to septic shock, naloxone safety and effectiveness are not established.[38]
Geriatric use[edit]
For patients 65 years and older, it is unclear if there is a difference in response. However, older people often have decreased liver and kidney function that may lead to an increased level of naloxone in their body.[6]
Available forms[edit]
Dosing[edit]
Administration of naloxone doses is the same regardless of different age populations (infant, children, teen, adults or elderly). Intramuscular naloxone can be administered as 0.4mg/ml per vial or intranasal naloxone, Narcan®, as 4mg (1 available spray per can). The overdosed opioid patient may require subsequent administration in 2-3 minute intervals if they remain unconscious/ unresponsive/ until emergency team arrives.[28]
Intravenous[edit]
In hospital settings, naloxone is injected intravenously, with an onset of 1–2 minutes and a duration of up to 45 minutes.[39]
Intramuscular or subcutaneous[edit]
Naloxone can also be administered via intramuscular or subcutaneous injection. The onset of naloxone provided through this route is 2 to 5 minutes with a duration of around 30–120min.[40] Naloxone administered intramuscularly are provided through pre-filled syringes, vials, and auto-injector. A hand-held auto-injector is pocket-sized and can be used in non-medical settings such as in the home.[22] It is designed for use by laypersons, including family members and caregivers of opioid users at-risk for an opioid emergency, such as an overdose.[41] According to the FDA's National Drug Code Directory, a generic version of the auto-injector began to be marketed at the end of 2019.[42]
Intranasal[edit]
Narcan nasal spray was approved in the US in 2015, and is the first FDA-approved nasal spray for emergency treatment or suspected overdose.[43][44] It was developed in a partnership between LightLake Therapeutics and the National Institute on Drug Abuse.[45] The approval process was fast-tracked.[46] A generic version of the nasal spray was approved in the United States in 2019, though did not come to market until 2021.[43][47]
In 2021, the FDA approved Kloxxado, an 8 mg dose of intranasal naloxone developed by Hikma Pharmaceuticals.[48] Citing the frequent need for multiple 4 mg doses of Narcan to successfully reverse overdose, packs of Kloxxado Nasal Spray contain two pre-packaged nasal spray devices, each containing 8 mg of naloxone.[49][9]
However, a wedge device (nasal atomizer) can also be attached to a syringe that may also be used to create a mist to deliver the drug to the nasal mucosa.[50] This is useful near facilities where many overdoses occur that already stock injectors.[51]
Contraindications[edit]
Main contraindications for naloxone use is hypersensitivity to the drug itself or any elements of its formulation.[52]
Side effects[edit]
Administration of naloxone to somebody who has used opioids may cause rapid-onset opioid withdrawal.[53]
Naloxone has little to no effect if opioids are not present.[54] In people with opioids in their system, it may cause increased sweating, nausea, restlessness, trembling, vomiting, flushing, and headache, and has in rare cases been associated with heart rhythm changes, seizures, and pulmonary edema.[55][56]
Naloxone has been shown to block the action of pain-lowering endorphins the body produces naturally. These endorphins likely operate on the same opioid receptors that naloxone blocks. It is capable of blocking a placebo pain-lowering response, if the placebo is administered together with a hidden or blind injection of naloxone.[57] Other studies have found that placebo alone can activate the body's μ-opioid endorphin system, delivering pain relief by the same receptor mechanism as morphine.[58][59]
Naloxone should be used with caution in people with cardiovascular disease as well as those that are currently taking medications that could have adverse effects on the cardiovascular system such as causing low blood pressure, fluid accumulation in the lungs (pulmonary edema), and abnormal heart rhythms. There have been reports of abrupt reversals with opioid antagonists leading to pulmonary edema and ventricular fibrillation.[60] Administration of naloxone may increase the risk of cardiac arrest for patients who are experiencing heart problems.[28]
Use of naloxone to treat people who have been using opioids recreationally may cause acute opioid withdrawal with distressing physiological symptoms such as shivering, tachycardia and nausea; these in turn may lead to aggression and reluctance to receive further treatment.[61]
Parenteral administration of naloxone can result in redness locally at the site of injection.[52]
Pharmacology[edit]
Pharmacodynamics[edit]
| Compound | Affinities (Ki) | Ratios | Refs | ||
|---|---|---|---|---|---|
| MOR | DOR | KOR | MOR:DOR:KOR | ||
| Naloxone | 1.1 nM 1.4 nM |
16 nM 67.5 nM |
12 nM 2.5 nM |
1:15:11 1:48:1.8 |
[62] [63][64] |
| (−)-Naloxone | 0.559 nM 0.93 nM |
36.5 nM 17 nM |
4.91 nM 2.3 nM |
1:65:9 1:18:2 |
[65] [66] |
| (+)-Naloxone | 3,550 nM >1,000 nM |
122,000 nM >1,000 nM |
8,950 nM >1,000 nM |
1:34:3 ND |
[65] [66] |
Naloxone is a lipophilic compound that acts as a non-selective and competitive opioid receptor antagonist.[17][6] The pharmacologically active isomer of naloxone is (−)-naloxone.[65][67] Naloxone's binding affinity is highest for the μ-opioid receptor (MOR), then the δ-opioid receptor (DOR), and lowest for the κ-opioid receptor (KOR);[17] naloxone has negligible affinity for the nociceptin receptor.[68]
If naloxone is administered in the absence of concomitant opioid use, no functional pharmacological activity occurs, except the inability of the body to combat pain naturally.[citation needed] In contrast to direct opiate agonists, which elicit opiate withdrawal symptoms when discontinued in opiate-tolerant people, no evidence indicates the development of tolerance or dependence on naloxone. The mechanism of action is not completely understood, but studies suggest it functions to produce withdrawal symptoms by competing for opioid receptors within the brain (a competitive antagonist, not a direct agonist), thereby preventing the action of both endogenous and xenobiotic opioids on these receptors without directly producing any effects itself.[69]
A single administration of naloxone at a relatively high dose of 2 mg by intravenous injection has been found to produce brain MOR blockade of 80% at 5 minutes, 47% at 2 hours, 44% at 4 hours, and 8% at 8 hours.[70] A low dose (2 μg/kg) produced brain MOR blockade of 42% at 5 minutes, 36% at 2 hours, 33% at 4 hours, and 10% at 8 hours.[70] Intranasal administration of naloxone via nasal spray has likewise been found to rapidly occupy brain MORs, with peak occupancy occurring at 20 minutes, peak occupancies of 67% at a dose of 2 mg and 85% with 4 mg, and an estimated half-life of occupancy disappearance of approximately 100 minutes (1.67 hours).[71][72]
Pharmacokinetics[edit]
When administered parenterally (non-orally or non-rectally, e.g., intravenously or by injection), as is most common, naloxone has a rapid distribution throughout the body. The mean serum half-life has been shown to range from 30 to 81 minutes, shorter than the average half-life of some opiates, necessitating repeat dosing if opioid receptors must be stopped from triggering for an extended period. Naloxone is primarily metabolized by the liver. Its major metabolite is naloxone-3-glucuronide, which is excreted in the urine.[69] For people with liver diseases such as alcoholic liver disease or hepatitis, naloxone usage has not been shown to increase serum liver enzyme levels.[73]
Naloxone has low systemic bioavailability when taken by mouth due to hepatic first-pass metabolism, but it does block opioid receptors that are located in the intestine.[74]
Chemistry[edit]
Naloxone, also known as N-allylnoroxymorphone or as 17-allyl-4,5α-epoxy-3,14-dihydroxymorphinan-6-one, is a synthetic morphinan derivative and was derived from oxymorphone (14-hydroxydihydromorphinone), an opioid analgesic.[75][76][77] Oxymorphone, in turn, was derived from morphine, an opioid analgesic and naturally occurring constituent of the opium poppy.[78] Naloxone is a racemic mixture of two enantiomers, (–)-naloxone (levonaloxone) and (+)-naloxone (dextronaloxone), only the former of which is active at opioid receptors.[79][80] The drug is highly lipophilic, allowing it to rapidly penetrate the brain and to achieve a far greater brain to serum ratio than that of morphine.[75] Opioid antagonists related to naloxone include cyprodime, nalmefene, nalodeine, naloxol, and naltrexone.[81]
History[edit]
Naloxone was patented in 1961 by Mozes J. Lewenstein, Jack Fishman, and the company Sankyo.[18] It was approved for opioid use disorder treatment in the United States in 1971.[82]
Society and culture[edit]
Misinformation[edit]
Naloxone has been subject to much inaccurate media reporting and a number of urban legends about it have become prevalent.[83]
One such myth is that naloxone makes the recipient violent.[84] Another is that events called "Lazarus parties" have taken place, in which people reportedly took fatal overdoses in anticipation of being treated with naloxone; in reality this was a fiction spread by the police.[83] Yet another is the claim that people have indulged in "yo-yoing", whereby they would take naloxone and opioids simultaneously to enjoy an extreme "high" and subsequent revival; the idea is scientifically nonsensical. This however, has never been scientifically studied. [83]
Names[edit]
Naloxone is its international nonproprietary name, British Approved Name, Dénomination Commune Française, Denominazione Comune Italiana, and Japanese Accepted Name, while naloxone hydrochloride is its United States Adopted Name and British Approved Name (Modified).[85][86][87][88]
The patent has expired and it is available as a generic medication. Several of formulations use patented dispensers (spray mechanisms or autoinjectors), and patent disputes instead of the generic forms of the nasal spray were litigated between 2016 and 2020, when a judge ruled in favor of Teva, the generic manufacturer.[89] Teva announced entry of the first generic nasal spray formulation in December 2021.[47] Brand names of naloxone include Narcan, Kloxxado, Nalone, Evzio, Prenoxad Injection, Narcanti, Narcotan, and Zimhi, among others.
Legal status and availability to law enforcement and emergency personnel[edit]
The examples and perspective in this section deal primarily with the United States and Canada and do not represent a worldwide view of the subject. (September 2022) |
Naloxone (Nyxoid) was approved for use in the European Union in September 2017.[90]
In March 2023, the US FDA approved naloxone hydrochloride nasal spray (Narcan) for over-the-counter (OTC), nonprescription, use – the first naloxone product approved for use without a prescription.[91] Other formulations and dosages of naloxone remain available by prescription.[91] In July 2023, the FDA approved Rivive nasal spray for OTC use.[92][93]
In the United States, naloxone is available without a prescription.[91][92][94][95] Naloxone may not be available at all pharmacies.[96][97]
As of 2019, officials in 29 states had issued standing orders to enable licensed pharmacists to provide naloxone to patients without the individual first visiting a prescriber.[98] Prescribers working with harm reduction or low threshold treatment programs have also issued standing orders to enable these organizations to distribute naloxone to their clients.[99] A standing order, also referred to as a "non-patient specific prescription" is written by a physician, nurse or other prescriber to authorize medicine distribution outside the doctor-patient relationship.[100] In the case of naloxone, these orders are meant to facilitate naloxone distribution to people using opioids, and their family members and friends.[98] Over 200 naloxone distribution programs utilize licensed prescribers to distribute the drug through such orders, or through the authority of pharmacists (as with California's legal provision, AB1535).[101][102]
Laws and policies in many US jurisdictions have been changed to allow wider distribution of naloxone.[103][104] In addition to laws or regulations permitting distribution of medicine to at risk individuals and families, some 36 states have passed laws that provide naloxone prescribers with immunity against both civil and criminal liabilities.[105] While paramedics in the US have carried naloxone for decades, law enforcement officers in many states throughout the country carry naloxone to reverse the effects of heroin overdoses when reaching the location before paramedics. As of 12 July 2015, law enforcement departments in 28 US states are allowed to or required to carry naloxone to quickly respond to opioid overdoses.[106] Programs training fire personnel in opioid overdose response using naloxone have also shown promise in the US, and efforts to integrate opioid fatality prevention into emergency response have grown due to the US overdose crisis.[107][108][109][110]
Following the use of the nasal spray device by police officers on Staten Island in New York, an additional 20,000 police officers will begin carrying naloxone in mid-2014. The state's Office of the Attorney General will provide US$1.2 million to supply nearly 20,000 kits. Police Commissioner William Bratton said: "Naloxone gives individuals a second chance to get help".[111] Emergency Medical Service Providers (EMS) routinely administer naloxone, except where basic Emergency Medical Technicians are prohibited by policy or by state law.[112] In efforts to encourage citizens to seek help for possible opioid overdoses, many states have adopted Good Samaritan laws that provide immunity against certain criminal liabilities for anybody who, in good faith, seeks emergency medical care for either themselves or someone around them who may be experiencing an opioid overdose.[113]
States including Vermont and Virginia have developed programs that mandate the prescription of naloxone when a prescription has exceeded a certain level of morphine milliequivalents per day as preventative measures against overdose.[114] Healthcare institution-based naloxone prescription programs have also helped reduce rates of opioid overdose in North Carolina, and have been replicated in the US military.[101][115]
In Canada, naloxone single-use syringe kits are distributed and available at various clinics and emergency rooms. Alberta Health Services is increasing the distribution points for naloxone kits at all emergency rooms, and various pharmacies and clinics province-wide. All Edmonton Police Service and Calgary Police Service patrol cars carry an emergency single-use naloxone syringe kit. Some Royal Canadian Mounted Police patrol vehicles also carry the drug, occasionally in excess to help distribute naloxone among users and concerned family/friends. Nurses, paramedics, medical technicians, and emergency medical responders can also prescribe and distribute the drug. As of February 2016, pharmacies across Alberta and some other Canadian jurisdictions are allowed to distribute single-use take-home naloxone kits or prescribe the drug to people using opioids.[116]
Following Alberta Health Services, Health Canada reviewed the prescription-only status of naloxone, resulting in plans to remove it in 2016, making naloxone more accessible.[117][118] Due to the rising number of drug deaths across the country, Health Canada proposed a change to make naloxone more widely available to Canadians in support of efforts to address the growing number of opioid overdoses.[119] In March 2016, Health Canada did change the prescription status of naloxone, as "pharmacies are now able to proactively give out naloxone to those who might experience or witness an opioid overdose."[120]
Community access[edit]

In a survey of US laypersons in December 2021, most people believed the scientifically supported idea that trained bystanders can reverse overdoses with naloxone.[121]
A survey of US naloxone prescription programs in 2010, revealed that 21 out of 48 programs reported challenges in obtaining naloxone in the months leading up to the survey, due mainly to either cost increases that outstripped allocated funding or the suppliers' inability to fill orders.[122] The approximate cost of a 1 ml ampoule of naloxone in the US is estimated to be significantly higher than in most other countries.[101]
Take-home naloxone programs for people who use opioids is under way in many North American cities.[122][123] CDC estimates that the US programs for drug users and their caregivers prescribing take-home doses of naloxone and training on its use prevented 10,000 opioid overdose deaths by 2014.[122]
In Australia, as of February 2016, some forms of naloxone are available "over the counter" in pharmacies without a prescription.[2][124][125] It comes in single-use filled syringe similar to law enforcement kits. A single dose costs AU$20; for those with a prescription, five doses can bought for AU$40, amounting to a rate of eight dollars per dose (2019).[126]
In Alberta, in addition to pharmacy distribution, take-home naloxone kits are available and distributed in most drug treatment or rehabilitation centres.[116]
In the European Union, take home naloxone pilots were launched in the Channel Islands and in Berlin in the late 1990s.[127] In 2008, the Welsh Assembly government announced its intention to establish demonstration sites for take-home naloxone,[128] and in 2010, Scotland instituted a national naloxone program.[129] Inspired by North American and European efforts, non-governmental organizations running programs to train drug users as overdose responders and supply them with naloxone are now operational in Russia, Ukraine, Georgia, Kazakhstan, Tajikistan, Afghanistan, China, Vietnam, and Thailand.[130]
In 2018, a maker of naloxone announced it would provide a free kit including two doses of the nasal spray, as well as educational materials, to each of the 16,568 public libraries and 2,700 YMCAs in the U.S.[131]
Criticism[edit]
Narcan's manufacturer charges $150 for the nasal spray and aggressively sues competitors looking to market a cheaper unauthorized generic version of the drug.[132] The public relations effort[133] to raise awareness of naloxone and promote policies such as bulk purchases by police departments obviously increases sales.
References[edit]
- ^ "Naloxone Use During Pregnancy". Drugs.com. 2 September 2019. Archived from the original on 25 April 2020. Retrieved 13 May 2020.
- ^ a b Lenton SR, Dietze PM, Jauncey M (March 2016). "Australia reschedules naloxone for opioid overdose". The Medical Journal of Australia. 204 (4): 146–147. doi:10.5694/mja15.01181. PMID 26937664. S2CID 9320372. Archived from the original on 19 July 2020. Retrieved 19 July 2020.
- ^ Anvisa (31 March 2023). "RDC Nº 784 - Listas de Substâncias Entorpecentes, Psicotrópicas, Precursoras e Outras sob Controle Especial" [Collegiate Board Resolution No. 784 - Lists of Narcotic, Psychotropic, Precursor, and Other Substances under Special Control] (in Brazilian Portuguese). Diário Oficial da União (published 4 April 2023). Archived from the original on 3 August 2023. Retrieved 16 August 2023.
- ^ "Frequently Asked Questions: Access to naloxone in Canada (including Narcan Nasal Spray)". Health Canada. 6 July 2016. Archived from the original on 16 August 2021. Retrieved 16 August 2021.
- ^ "Naloxone 400 micrograms/ml solution for Injection/Infusion – Summary of Product Characteristics (SmPC)". (emc). 6 February 2019. Archived from the original on 4 August 2020. Retrieved 13 May 2020.
- ^ a b c d "Narcan- naloxone hydrochloride spray Narcan- naloxone hydrochloride spray". DailyMed. 7 October 2019. Archived from the original on 30 January 2016. Retrieved 12 May 2020.
- ^ "Evzio- naloxone hydrochloride injection, solution". DailyMed. 1 February 2018. Archived from the original on 8 October 2020. Retrieved 5 October 2020.
- ^ "Zimhi- naloxone hydrochloride injection, solution". DailyMed. 29 September 2022. Archived from the original on 7 January 2023. Retrieved 7 January 2023.
- ^ a b "Kloxxado- naloxone hcl spray". DailyMed. 10 May 2022. Archived from the original on 7 January 2023. Retrieved 7 January 2023.
- ^ "NDA 208411/S-006 Supplemental Approval letter" (PDF). U.S. Food and Drug Administration (FDA). Archived (PDF) from the original on 25 June 2023. Retrieved 29 July 2023.
- ^ "RiVive: Naloxone HCl Nasal Spray 3 mg Emergency Treatment of Opioid Overdose" (PDF). Front Actuator (nasal spray device) Label. U.S. Food and Drug Administration. Archived (PDF) from the original on 2 August 2023. Retrieved 2 August 2023.
- ^ Ryan SA, Dunne RB (May 2018). "Pharmacokinetic properties of intranasal and injectable formulations of naloxone for community use: a systematic review". Pain Management. 8 (3): 231–245. doi:10.2217/pmt-2017-0060. PMID 29683378.
- ^ a b c d e f g h i j k l m n "Naloxone Hydrochloride". The American Society of Health-System Pharmacists. Archived from the original on 2 January 2015. Retrieved 2 January 2015.
- ^ McDonald R, Lorch U, Woodward J, Bosse B, Dooner H, Mundin G, et al. (March 2018). "Pharmacokinetics of concentrated naloxone nasal spray for opioid overdose reversal: Phase I healthy volunteer study". Addiction. 113 (3): 484–493. doi:10.1111/add.14033. PMC 5836974. PMID 29143400.
- ^ "Naloxone DrugFacts". National Institute on Drug Abuse. 1 June 2021. Archived from the original on 6 January 2022. Retrieved 5 January 2022.
- ^ "Prescribing medicines in pregnancy database". Australian Government. 3 March 2014. Archived from the original on 8 April 2014. Retrieved 22 April 2014.
- ^ a b c Malenka RC, Nestler EJ, Hyman SE (2009). Sydor A, Brown RY (eds.). Molecular Neuropharmacology: A Foundation for Clinical Neuroscience (2nd ed.). New York: McGraw-Hill Medical. pp. 190–191, 287. ISBN 9780071481274.
Products of this research include the discovery of lipophilic, small-molecule opioid receptor antagonists, such as naloxone and naltrexone, which have been critical tools for investigating the physiology and behavioral actions of opiates. ... A competitive antagonist of opiate action (naloxone) had been identified in early studies. ... Opiate antagonists have clinical utility as well. Naloxone, a nonselective antagonist with a relative affinity of μ > δ > κ, is used to treat heroin and other opiate overdoses.
- ^ a b Yardley W (14 December 2013). "Jack Fishman Dies at 83; Saved Many From Overdose". The New York Times. Archived from the original on 15 December 2013. Retrieved 6 July 2015.
- ^ US patent 3493657, Jack Fishman & Mozes Juda Lewenstein, "Therapeutic compositions of n-allyl-14-hydroxy - dihydronormorphinane and morphine", published 1970-02-03, issued 1970-02-03, assigned to Mozes Juda Lewenstein Archived 7 December 2022 at the Wayback Machine
- ^ World Health Organization (2019). World Health Organization model list of essential medicines: 21st list 2019. Geneva: World Health Organization. hdl:10665/325771. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
- ^ "Competitive Generic Therapy Approvals". U.S. Food and Drug Administration (FDA). 29 June 2023. Archived from the original on 29 June 2023. Retrieved 29 June 2023.
- ^ a b Lavonas EJ, Drennan IR, Gabrielli A, Heffner AC, Hoyte CO, Orkin AM, et al. (November 2015). "Part 10: Special Circumstances of Resuscitation: 2015 American Heart Association Guidelines Update for Cardiopulmonary Resuscitation and Emergency Cardiovascular Care". Circulation. 132 (18 Suppl 2): S501–S518. doi:10.1161/cir.0000000000000264. PMID 26472998.
- ^ Maxwell S, Bigg D, Stanczykiewicz K, Carlberg-Racich S (2006). "Prescribing naloxone to actively injecting heroin users: a program to reduce heroin overdose deaths". Journal of Addictive Diseases. 25 (3): 89–96. doi:10.1300/J069v25n03_11. PMID 16956873. S2CID 17246459.
- ^ "Project Lazarus, Wilkes County, North Carolina" (PDF). Policy Briefing Document Prepared for the North Carolina Medical Board in Advance of the Public Hearing Regarding Prescription Naloxone. Raleigh, NC. 2007. Archived (PDF) from the original on 26 May 2022. Retrieved 20 March 2022.[page needed][verification needed]
- ^ Bowman S, Eiserman J, Beletsky L, Stancliff S, Bruce RD (July 2013). "Reducing the health consequences of opioid addiction in primary care". The American Journal of Medicine. 126 (7): 565–571. doi:10.1016/j.amjmed.2012.11.031. PMID 23664112.
- ^ Marlatt GA, Larimer ME, Witkiewitz K, eds. (2011). Harm reduction: Pragmatic strategies for managing high-risk behaviors (2nd ed.). New York: Guilford Press. ISBN 978-1-4625-0256-1.
- ^ van Dorp E, Yassen A, Dahan A (March 2007). "Naloxone treatment in opioid addiction: the risks and benefits". Expert Opinion on Drug Safety. 6 (2): 125–132. doi:10.1517/14740338.6.2.125. PMID 17367258. S2CID 11650530.
- ^ a b c d e "RxFiles" (PDF). www.rxfiles.ca. Retrieved 2 May 2024.
- ^ Niemann JT, Getzug T, Murphy W (October 1986). "Reversal of clonidine toxicity by naloxone". Annals of Emergency Medicine. 15 (10): 1229–1231. doi:10.1016/s0196-0644(86)80874-5. PMID 3752658.
- ^ a b c Ahmad SA, Scolnik D, Snehal V, Glatstein M (2015). "Use of naloxone for clonidine intoxication in the pediatric age group: case report and review of the literature". American Journal of Therapeutics. 22 (1): e14–e16. doi:10.1097/MJT.0b013e318293b0e8. PMID 23782760.
- ^ a b Seger DL (2002). "Clonidine toxicity revisited". Journal of Toxicology. Clinical Toxicology. 40 (2): 145–155. doi:10.1081/CLT-120004402. PMID 12126186. S2CID 2730597.
- ^ a b c Orman JS, Keating GM (2009). "Buprenorphine/naloxone: a review of its use in the treatment of opioid dependence". Drugs. 69 (5): 577–607. doi:10.2165/00003495-200969050-00006. PMID 19368419. S2CID 209147406.
- ^ a b McNicholas, M.D., Ph.D., Laura (2004). Clinical Guidelines for the Use of Buprenorphine in the Treatment of Opioid Addiction. 1 Choke Cherry Road Rockville, MD 20857: U.S. Department of Health and Human Services - Substance Abuse and Mental Health Services Administration. p. 23.
{{cite book}}: CS1 maint: location (link) CS1 maint: multiple names: authors list (link) - ^ Boeuf B, Poirier V, Gauvin F, Guerguerian AM, Roy C, Farrell CA, Lacroix J (2003). "Naloxone for shock". The Cochrane Database of Systematic Reviews. 2010 (4): CD004443. doi:10.1002/14651858.CD004443. PMC 9036847. PMID 14584016.
- ^ "Naloxone use while Breastfeeding". Drugs.com. Archived from the original on 2 November 2020. Retrieved 15 August 2018.
- ^ a b Moe-Byrne T, Brown JV, McGuire W (October 2018). "Naloxone for opioid-exposed newborn infants". The Cochrane Database of Systematic Reviews. 2018 (10): CD003483. doi:10.1002/14651858.CD003483.pub3. PMC 6517169. PMID 30311212.
- ^ McGuire W, Fowlie PW, Evans DJ (26 January 2004). "Naloxone for preventing morbidity and mortality in newborn infants of greater than 34 weeks' gestation with suspected perinatal asphyxia". The Cochrane Database of Systematic Reviews. 2010 (1). John Wiley & Sons, Ltd: CD003955. doi:10.1002/14651858.CD003955.pub2. PMC 6485479. PMID 14974047.
- ^ "Narcan (Naloxone Hydrochloride Injection): Side Effects, Interactions, Warning, Dosage & Uses". RxList. Archived from the original on 11 November 2020. Retrieved 24 October 2019.
- ^ Drug information handbook for advanced practice nursing: a comprehensive resource for nurse practitioners, nurse midwives and clinical specialists, including selected disease management guidelines. Lexicomp. 2013. ISBN 978-1591953234. OCLC 827841946.
- ^ Naloxone for Treatment of Opioid Overdose Oct. 2016
- ^ "FDA approves new hand-held auto-injector to reverse opioid overdose" (Press release). U.S. Food and Drug Administration (FDA). Archived from the original on 16 July 2015. Retrieved 20 July 2015.
- ^ "NDC 72853-051-02 Naloxone Hydrochloride Auto-injector". NDClist.com. Archived from the original on 21 March 2020. Retrieved 21 March 2020.
- ^ a b "FDA approves first generic naloxone nasal spray to treat opioid overdose" (Press release). U.S. Food and Drug Administration (FDA). 11 September 2019. Archived from the original on 14 September 2019. Retrieved 23 October 2019.
- ^ "FDA Approves Narcan Nasal Spray". www.jems.com. Archived from the original on 20 November 2015. Retrieved 21 November 2015.
- ^ Volkow N (18 November 2015). "Narcan Nasal Spray: Life Saving Science at NIDA". National Institute on Drug Abuse (NIDA). Archived from the original on 26 February 2017.
- ^ Dennis B (3 April 2014). "FDA approves device to combat opioid drug overdose". The Washington Post. Archived from the original on 7 April 2014. Retrieved 8 April 2014.
- ^ a b "Teva Announces Launch of First-to-Market Generic Version of Narcan (Naloxone Hydrochloride Nasal Spray), in the U.S." (Press release). Teva Pharmaceuticals. 22 December 2021. Retrieved 2 August 2023 – via Business Wire.
- ^ "FDA Approves Higher Dosage of Naloxone Nasal Spray to Treat Opioid Overdose" (Press release). U.S. Food and Drug Administration (FDA). 11 May 2021. Archived from the original on 1 September 2022. Retrieved 21 September 2022.
- ^ Abdelal R, Raja Banerjee A, Carlberg-Racich S, Darwaza N, Ito D, Shoaff J, Epstein J (May 2022). "Real-world study of multiple naloxone administration for opioid overdose reversal among bystanders". Harm Reduction Journal. 19 (1): 49. doi:10.1186/s12954-022-00627-3. PMC 9122081. PMID 35596213.
- ^ Wolfe TR, Bernstone T (April 2004). "Intranasal drug delivery: an alternative to intravenous administration in selected emergency cases". Journal of Emergency Nursing. 30 (2): 141–147. doi:10.1016/j.jen.2004.01.006. PMID 15039670.
- ^ Fiore K (13 June 2015). "On-Label Nasal Naloxone in the Works". MedPage Today. Archived from the original on 1 August 2015. Retrieved 20 July 2015.
- ^ a b login.uml.idm.oclc.org http://online.lexi.com/lco/action/doc/retrieve/docid/patch_f/7338?cesid=10E6wyzfs7g&searchUrl=/lco/action/search?q=naloxone&t=name&acs=false&acq=naloxone. Retrieved 2 May 2024.
{{cite web}}: Missing or empty|title=(help) - ^ Britch SC, Walsh SL (July 2022). "Treatment of opioid overdose: current approaches and recent advances". Psychopharmacology (Berl) (Review). 239 (7): 2063–2081. doi:10.1007/s00213-022-06125-5. PMC 8986509. PMID 35385972.
- ^ "Opioid overdose". www.who.int. 29 August 2023. Retrieved 17 November 2023.
- ^ "Naloxone Side Effects in Detail". Drugs.com. Archived from the original on 7 May 2015. Retrieved 5 May 2015.
- ^ Schwartz JA, Koenigsberg MD (November 1987). "Naloxone-induced pulmonary edema". Annals of Emergency Medicine. 16 (11): 1294–1296. doi:10.1016/S0196-0644(87)80244-5. PMID 3662194.
- ^ Sauro MD, Greenberg RP (February 2005). "Endogenous opiates and the placebo effect: a meta-analytic review". Journal of Psychosomatic Research. 58 (2): 115–120. doi:10.1016/j.jpsychores.2004.07.001. PMID 15820838.
- ^ "More Than Just a Sugar Pill: Why the placebo effect is real - Science in the News". Science in the News. 14 September 2016. Archived from the original on 15 November 2017. Retrieved 14 November 2017.
- ^ Carvalho C, Caetano JM, Cunha L, Rebouta P, Kaptchuk TJ, Kirsch I (December 2016). "Open-label placebo treatment in chronic low back pain: a randomized controlled trial". Pain. 157 (12): 2766–2772. doi:10.1097/j.pain.0000000000000700. PMC 5113234. PMID 27755279.
- ^ "Naloxone: Contraindications". UpToDate. Archived from the original on 20 May 2016. Retrieved 31 October 2017.
- ^ "Drug misuse and dependence: UK guidelines on clinical management" (PDF) (Practice guideline). Public Health England. November 2017. p. 181.
- ^ Tam SW (February 1985). "(+)-[3H]SKF 10,047, (+)-[3H]ethylketocyclazocine, mu, kappa, delta and phencyclidine binding sites in guinea pig brain membranes". European Journal of Pharmacology. 109 (1): 33–41. doi:10.1016/0014-2999(85)90536-9. PMID 2986989.
- ^ Toll L, Berzetei-Gurske IP, Polgar WE, Brandt SR, Adapa ID, Rodriguez L, et al. (March 1998). "Standard binding and functional assays related to medications development division testing for potential cocaine and opiate narcotic treatment medications". NIDA Research Monograph. 178: 440–466. PMID 9686407.
- ^ Clark SD, Abi-Dargham A (October 2019). "The Role of Dynorphin and the Kappa Opioid Receptor in the Symptomatology of Schizophrenia: A Review of the Evidence". Biological Psychiatry. 86 (7): 502–511. doi:10.1016/j.biopsych.2019.05.012. PMID 31376930. S2CID 162168648.
- ^ a b c Codd EE, Shank RP, Schupsky JJ, Raffa RB (September 1995). "Serotonin and norepinephrine uptake inhibiting activity of centrally acting analgesics: structural determinants and role in antinociception". The Journal of Pharmacology and Experimental Therapeutics. 274 (3): 1263–1270. PMID 7562497. Archived from the original on 12 March 2020. Retrieved 13 December 2014.
- ^ a b Raynor K, Kong H, Chen Y, Yasuda K, Yu L, Bell GI, Reisine T (February 1994). "Pharmacological characterization of the cloned kappa-, delta-, and mu-opioid receptors". Molecular Pharmacology. 45 (2): 330–334. PMID 8114680. Archived from the original on 22 June 2018. Retrieved 22 June 2018.
- ^ "Naloxone: Summary". IUPHAR/BPS Guide to Pharmacology. International Union of Basic and Clinical Pharmacology. Archived from the original on 16 November 2017. Retrieved 15 November 2017.
The approved drug naloxone INN-assigned preparation is the (-)-enantiomer. ... The (+) isomer is inactive at the opioid receptors. Marketed formulations may contain naloxone hydrochloride
- ^ "Opioid receptors: Introduction". IUPHAR/BPS Guide to Pharmacology. International Union of Basic and Clinical Pharmacology. Archived from the original on 21 October 2017. Retrieved 15 November 2017.
The opioid antagonist, naloxone, which binds to μ, δ and κ receptors (with differing affinities), does not have significant affinity for the ORL1/LC132 receptor. These studies indicate that, from a pharmacological perspective, there are two major branches in the opioid peptide-N/OFQ receptor family: the main branch comprising the μ, δ and κ receptors, where naloxone acts as an antagonist; and a second branch with the receptor for N/OFQ, which has negligible affinity for naloxone.
- ^ a b "Naloxone Hydrochloride injection, solution". DailyMed. Archived from the original on 22 April 2014. Retrieved 21 April 2014.
- ^ a b Colasanti A, Lingford-Hughes A, Nutt D (2013). "Opioids Neuroimaging". In Miller PM (ed.). Biological Research on Addiction. Comprehensive Addictive Behaviors and Disorders. Vol. 2. Elsevier. pp. 675–687. doi:10.1016/B978-0-12-398335-0.00066-2. ISBN 9780123983350.
- ^ Waarde AV, Absalom AR, Visser AK, Dierckx RA (30 September 2020). "Positron Emission Tomography (PET) Imaging of Opioid Receptors". In Dierckx RA, Otte A, De Vries EF, Van Waarde A, Luiten PG (eds.). PET and SPECT of Neurobiological Systems. Springer International Publishing. pp. 749–807. doi:10.1007/978-3-030-53176-8_21. ISBN 978-3-030-53175-1. S2CID 241535315. Archived from the original on 9 March 2023. Retrieved 23 January 2023.
- ^ Johansson J, Hirvonen J, Lovró Z, Ekblad L, Kaasinen V, Rajasilta O, et al. (August 2019). "Intranasal naloxone rapidly occupies brain mu-opioid receptors in human subjects". Neuropsychopharmacology. 44 (9): 1667–1673. doi:10.1038/s41386-019-0368-x. PMC 6785104. PMID 30867551.
- ^ "Naloxone", LiverTox: Clinical and Research Information on Drug-Induced Liver Injury, National Institute of Diabetes and Digestive and Kidney Diseases, 2012, PMID 31643568, archived from the original on 28 August 2021, retrieved 30 October 2019
- ^ Meissner W, Schmidt U, Hartmann M, Kath R, Reinhart K (January 2000). "Oral naloxone reverses opioid-associated constipation". Pain. 84 (1): 105–109. doi:10.1016/S0304-3959(99)00185-2. PMID 10601678. S2CID 42230143.
- ^ a b Dean R, Bilsky EJ, Negus SS (12 March 2009). Opiate Receptors and Antagonists: From Bench to Clinic. Springer Science & Business Media. pp. 514–. ISBN 978-1-59745-197-0.
- ^ Nagase H (21 January 2011). Chemistry of Opioids. Springer. pp. 93–. ISBN 978-3-642-18107-8.
- ^ "Morphinan-6-one, 4,5-epoxy-3,14-dihydroxy-17-(2-propenyl)-, (5α)-". Archived from the original on 1 December 2017. Retrieved 20 November 2017.
- ^ Seppala MD, Rose ME (25 January 2011). Prescription Painkillers: History, Pharmacology, and Treatment. Hazelden Publishing. pp. 143–. ISBN 978-1-59285-993-1. Archived from the original on 13 January 2023. Retrieved 20 November 2017.
- ^ Bennett LA (2006). New Topics in Substance Abuse Treatment. Nova Publishers. pp. 9–. ISBN 978-1-59454-831-4. Archived from the original on 13 January 2023. Retrieved 20 November 2017.
- ^ Wang JQ (2003). Drugs of Abuse: Neurological Reviews and Protocols. Springer Science & Business Media. pp. 44–. ISBN 978-1-59259-358-3. Archived from the original on 13 January 2023. Retrieved 20 November 2017.
- ^ Brunton L, Chabner B, Knollman B (20 December 2010). Goodman and Gilman's The Pharmacological Basis of Therapeutics, Twelfth Edition. McGraw Hill Professional. p. 510. ISBN 978-0-07-162442-8. Archived from the original on 13 January 2023. Retrieved 20 November 2017.
- ^ "Naloxone: FDA-Approved Drugs". U.S. Food and Drug Administration (FDA). Archived from the original on 4 August 2020. Retrieved 13 May 2020.
- ^ a b c Crabtree A, Masuda JR (May 2019). "Naloxone urban legends and the opioid crisis: what is the role of public health?". BMC Public Health. 19 (1): 670. doi:10.1186/s12889-019-7033-5. PMC 6543555. PMID 31146721.
- ^ "Naloxone myths debunked" (PDF) (pdf). Indiana State Department of Health. Retrieved 7 September 2023.
- ^ Elks J (14 November 2014). The Dictionary of Drugs: Chemical Data: Chemical Data, Structures and Bibliographies. Springer. pp. 851–. ISBN 978-1-4757-2085-3. Archived from the original on 13 January 2023. Retrieved 20 November 2017.
- ^ Index Nominum 2000: International Drug Directory. Taylor & Francis. 2000. pp. 715–. ISBN 978-3-88763-075-1. Archived from the original on 13 January 2023. Retrieved 20 November 2017.
- ^ Morton IK, Hall JM (6 December 2012). Concise Dictionary of Pharmacological Agents: Properties and Synonyms. Springer Science & Business Media. pp. 189–. ISBN 978-94-011-4439-1.
- ^ "Naloxone". Archived from the original on 1 December 2017. Retrieved 20 November 2017.
- ^ "Teva Invalidates Opiant Patents In Narcan Suit - Law360". www.law360.com. Archived from the original on 7 January 2022. Retrieved 5 January 2022.
- ^ "Nyxoid EPAR". European Medicines Agency (EMA). 17 September 2018. Archived from the original on 5 April 2020. Retrieved 12 May 2020.
- ^ a b c "FDA Approves First Over-the-Counter Naloxone Nasal Spray" (Press release). U.S. Food and Drug Administration (FDA). 29 March 2023. Retrieved 29 March 2023.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.
- ^ a b "FDA Approves Second Over-the-Counter Naloxone Nasal Spray Product". U.S. Food and Drug Administration (FDA) (Press release). 28 July 2023. Retrieved 29 July 2023.
- ^ "FDA Approves Harm Reduction Therapeutics' Over-the-Counter Opioid Overdose Reversal Medication" (Press release). Purdue Pharma. 1 August 2023. Retrieved 1 August 2023 – via Business Wire.
- ^ "Naloxone Opioid Overdose Reversal Medication". CVS Health. Archived from the original on 19 September 2018. Retrieved 19 September 2018.
- ^ Suttles C. "Wyoming's Albertsons, Safeway pharmacies to offer Narcan over the counter". Wyoming Tribune Eagle. Archived from the original on 3 February 2018. Retrieved 19 September 2018.
- ^ Meyerson BE, Agley JD, Davis A, Jayawardene W, Hoss A, Shannon DJ, et al. (July 2018). "Predicting pharmacy naloxone stocking and dispensing following a statewide standing order, Indiana 2016". Drug and Alcohol Dependence. 188: 187–192. doi:10.1016/j.drugalcdep.2018.03.032. PMC 6375076. PMID 29778772.
- ^ Meyerson BE, Agley JD, Jayawardene W, Eldridge LA, Arora P, Smith C, et al. (May 2020). "Feasibility and acceptability of a proposed pharmacy-based harm reduction intervention to reduce opioid overdose, HIV and hepatitis C". Research in Social & Administrative Pharmacy. 16 (5): 699–709. doi:10.1016/j.sapharm.2019.08.026. hdl:10150/641340. PMID 31611071.
- ^ a b "Addressing Opioid Overdose through Statewide Standing Orders for Naloxone Distribution". Network for Public Health Law. Archived from the original on 6 January 2022. Retrieved 5 January 2022.
- ^ Wheeler E, Jones TS, Gilbert MK, Davidson PJ (June 2015). "Opioid Overdose Prevention Programs Providing Naloxone to Laypersons - United States, 2014". MMWR. Morbidity and Mortality Weekly Report. 64 (23): 631–635. PMC 4584734. PMID 26086633.
- ^ "Guide: Treating Heroin and Opioid Use Disorder". PA.Gov. Archived from the original on 1 October 2020. Retrieved 1 October 2020.
- ^ a b c Beletsky L, Burris SC, Kral AH (2009). "Closing Death's Door: Action Steps to Facilitate Emergency Opioid Drug Overdose Reversal in the United States" (PDF). doi:10.2139/ssrn.1437163. SSRN 1437163. Archived from the original (PDF) on 27 January 2023 – via Boonshoft School of Medicine.
{{cite journal}}: Cite journal requires|journal=(help) - ^ Burris SC, Beletsky L, Castagna CA, Coyle C, Crowe C, McLaughlin JM (2009). "Stopping an Invisible Epidemic: Legal Issues in the Provision of Naloxone to Prevent Opioid Overdose". SSRN 1434381.
- ^ Davis C. "Legal interventions to reduce overdose mortality: Naloxone access and overdose good samaritan laws" (PDF). Network for Public Health Law. Archived (PDF) from the original on 3 September 2014.
- ^ Davis C, Webb D, Burris S (March 2013). "Changing law from barrier to facilitator of opioid overdose prevention". The Journal of Law, Medicine & Ethics. 41 (Suppl 1): 33–36. doi:10.1111/jlme.12035. PMID 23590737. S2CID 22127036.
- ^ Cutcliff A, Stringberg A, Atkins C. "As Naloxone Accessibility Increases, Pharmacist's Role Expands". Pharmacy Times. Archived from the original on 31 October 2019. Retrieved 31 October 2019.
- ^ "US Law Enforcement Who Carry Naloxone". North Carolina Harm Reduction Coalition. Archived from the original on 13 July 2015. Retrieved 12 July 2015.
- ^ Beletsky L, Rich JD, Walley AY (November 2012). "Prevention of fatal opioid overdose". JAMA. 308 (18): 1863–1864. doi:10.1001/jama.2012.14205. PMC 3551246. PMID 23150005.
- ^ Lavoie D (April 2012). "Naloxone: Drug-Overdose Antidote Is Put In Addicts' Hands". Huffington Post. Archived from the original on 18 May 2012.
- ^ Davis CS, Beletsky L (July 2009). "Bundling occupational safety with harm reduction information as a feasible method for improving police receptiveness to syringe access programs: evidence from three U.S. cities". Harm Reduction Journal. 6 (1): 16. doi:10.1186/1477-7517-6-16. PMC 2716314. PMID 19602236.
- ^ "2013 National drug control strategy" (PDF). Office of National Drug Control Policy. 2013. Archived (PDF) from the original on 21 January 2017 – via National Archives.
- ^ Durando J (27 May 2014). "NYPD officers to carry heroin antidote". USA Today. Archived from the original on 3 July 2014. Retrieved 30 May 2014.
- ^ Faul M, Dailey MW, Sugerman DE, Sasser SM, Levy B, Paulozzi LJ (July 2015). "Disparity in naloxone administration by emergency medical service providers and the burden of drug overdose in US rural communities". American Journal of Public Health. 105 (Suppl 3): e26–e32. doi:10.2105/AJPH.2014.302520. PMC 4455515. PMID 25905856.
- ^ "Drug Overdose Immunity and Good Samaritan Laws". www.ncsl.org. Archived from the original on 13 September 2019. Retrieved 31 October 2019.
- ^ Jones CM, Compton W, Vythilingam M, Giroir B (August 2019). "Naloxone Co-prescribing to Patients Receiving Prescription Opioids in the Medicare Part D Program, United States, 2016-2017". JAMA. 322 (5): 462–464. doi:10.1001/jama.2019.7988. PMC 6686765. PMID 31386124.
- ^ Albert S, Brason FW, Sanford CK, Dasgupta N, Graham J, Lovette B (June 2011). "Project Lazarus: community-based overdose prevention in rural North Carolina". Pain Medicine. 12 (Suppl 2): S77–S85. doi:10.1111/j.1526-4637.2011.01128.x. PMID 21668761.
- ^ a b "Naloxone kits now available at drug stores as province battles fentanyl crisis". CBC News. 17 February 2016. Archived from the original on 4 March 2016. Retrieved 9 March 2023.
- ^ "Naloxone's prescription-only status to get Health Canada review". CBC News. Archived from the original on 5 February 2016. Retrieved 5 February 2016.
- ^ "Fentanyl and the take-home naloxone program Alberta Health". Archived from the original on 5 February 2016. Retrieved 5 February 2016.
- ^ "Health Canada Statement on Change in Federal Prescription Status of Naloxone". news.gc.ca. 14 January 2016. Archived from the original on 10 January 2017. Retrieved 29 February 2016 – via Wayback Machine.
- ^ "Questions and Answers - Naloxone". Health Canada. 22 March 2017. Archived from the original on 8 September 2017. Retrieved 12 June 2017.
- ^ Agley J, Xiao Y, Eldridge L, Meyerson B, Golzarri-Arroyo L (May 2022). "Beliefs and misperceptions about naloxone and overdose among U.S. laypersons: a cross-sectional study". BMC Public Health. 22 (1): 924. doi:10.1186/s12889-022-13298-3. PMC 9086153. PMID 35538566.
- ^ a b c Centers for Disease Control Prevention (CDC) (February 2012). "Community-based opioid overdose prevention programs providing naloxone - United States, 2010". MMWR. Morbidity and Mortality Weekly Report. 61 (6): 101–105. PMC 4378715. PMID 22337174. Archived from the original on 26 April 2012.
- ^ Donkin K (9 September 2012). "Toronto naloxone program reduces drug overdoses among addicts". The Toronto Star. Archived from the original on 5 December 2014. Retrieved 5 May 2015.
- ^ Davey M (29 January 2016). "Selling opioid overdose antidote Naloxone over counter 'will save lives'". The Guardian. Archived from the original on 3 December 2016.
- ^ "Why the 'heroin antidote' naloxone is now available in pharmacies". ABC. 1 February 2016. Archived from the original on 4 February 2016. Retrieved 1 February 2016.
- ^ Coulter E (27 August 2019). "This drug can temporarily reverse an opioid overdose. So why aren't people using it?". ABC News. Archived from the original on 27 August 2019. Retrieved 28 August 2019.
- ^ Dettmer K, Saunders B, Strang J (April 2001). "Take home naloxone and the prevention of deaths from opiate overdose: two pilot schemes". BMJ. 322 (7291): 895–896. doi:10.1136/bmj.322.7291.895. PMC 30585. PMID 11302902.
- ^ "IHRA 21st International Conference Liverpool, 26th April 2010 - Introducing 'take home' Naloxone in Wales" (PDF). Archived (PDF) from the original on 20 July 2011. Retrieved 9 March 2011.
- ^ McAuley A, Best D, Taylor A, Hunter C, Robertson R (1 August 2012). "From evidence to policy: The Scottish national naloxone programme". Drugs: Education, Prevention and Policy. 19 (4): 309–319. doi:10.3109/09687637.2012.682232. ISSN 0968-7637. S2CID 73263293. Archived from the original on 9 March 2023. Retrieved 6 January 2022.
- ^ "Stopping Overdose". Open Society Foundations. Archived from the original on 7 January 2022. Retrieved 6 January 2022.
- ^ "Every U.S. Public Library and YMCA Will Soon Get Narcan for Free". Time. Archived from the original on 1 April 2019. Retrieved 2 April 2019.
- ^ Gilgore S. "Emergent BioSolutions defends opioid overdose drug against generic competitor". Archived from the original on 29 May 2022. Retrieved 23 July 2022.
- ^ Ducharme J (24 October 2018). "Every U.S. Public Library and YMCA Will Soon Get Narcan for Free". Time Magazine. Archived from the original on 23 July 2022. Retrieved 23 July 2022.
Further reading[edit]
- Naloxone, Flumazenil and Dantrolene as Antidotes. IPCS/CEC Evaluation of Antidotes Series. Vol. 1. Cambridge University Press. 1993. ISBN 0-521-45459-X. EUR 14797 EN. Archived from the original on 15 December 2003. Retrieved 15 February 2004.
External links[edit]
- "Naloxone Nasal Spray". MedlinePlus.
- "Naloxone". U.S. Substance Abuse and Mental Health Services Administration (SAMHSA). 16 June 2015.
