User:Gjpallen46/Sandbox
Aldosterone synthase (or 18-hydroxylase) is a steroid hydroxylase cytochrome P450 enzyme involved in the biosynthesis of the mineralocorticoid aldosterone. Officially named cytochrome P450, family 11, subfamily B, polypeptide 2, it is a protein which is only expressed in the zona glomerulosa[1] of the adrenal cortex and is primarily regulated by the renin-angiotensin system.[2]
Genetics[edit]
Aldosterone synthase is encoded on chromosome 8q22[1] by the CYP11B2 gene.[1] The gene contains 9 exons and spans roughly 7000 base pairs of DNA.[1] Research has shown that calcium ions act as a transcription factor for CYP11B2 through well defined interactions at the 5'-flanking region of CYP11B2.[1]
Aldosterone synthase is a member of the cytochrome P450 superfamily of enzymes.[3] The cytochrome P450 proteins are monooxygenases that catalyze many reactions involved in drug metabolism and synthesis of cholesterol, steroids, and other lipids.
Aldosterone Activity[edit]
Aldosterone, when present, binds to intracellular mineralocorticoid receptors which can then bind to DNA and influence transcription of genes encoding serum and glucocorticoid induced kinase, sgk. Serum and glucocorticoid induced kinase (sgk) can phosphorylate a uniquitin ligase (nedd4) which inactivates its ability to remove and degrade sodium channels from apical membranes.[4] Aldosterone activity is primarily regulated by the renin-angiotensin system[2].
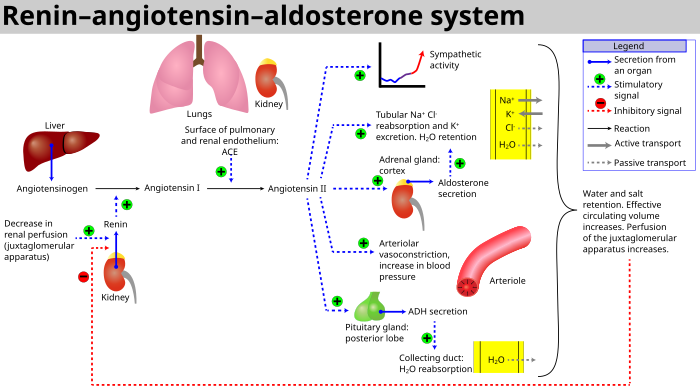
Metabolism[edit]
Aldosterone synthase converts 11-deoxycorticosterone to corticosterone, to 18-hydroxycorticosterone, and finally to aldosterone:
In human metabolism the biosynthesis of aldosterone largely depends on the metabolism of cholesterol. Cholesterol is hydroxylated becoming (20R,22R)-dihydroxycholesterol which is then metabolized as a direct precursor to pregnenolone. Pregnenolone can then followed one of two pathways which involve the metabolism of progesterone or the testosterone and estradiol biosynthesis. Aldosterone is synthesized by following the metabolism of progesterone.

Methyl Oxidase Deficiency[edit]
Lack of metabolically active aldosterone synthase leads to corticosterone methyl oxidase deficiency type I and II. The deficiency is characterized clinically by salt-wasting, failure to thrive, and growth retardation [5]. The in-active proteins are caused by the autosomal recessive inheritance of defective CYP11B2 genes in which genetic mutations destroy the enzymatic activity of aldosterone synthase[5]. Deficient aldosterone synthase activity results in impaired biosynthesis of aldosterone while corticosterone in the zona glomerulosa is excessively produced in both corticosterone methyl oxidase deficiency type I and II. The corticosterone methyl oxidase deficiencies both share this effect however type I causes an overall deficiency of 18-hydroxycorticosterone while type II overproduces it[5].
See also[edit]
Additional images[edit]

-
Corticosteroid biosynthetic pathway in rat
References[edit]
- ^ a b c d e Bassett MH; White PC; Rainey WE (2004). "The regulation of aldosterone synthase expression". Molecular and Cellular Endocrinology. 217 (1–2): 67–74. doi:10.1016. ISSN 0303-7207.
{{cite journal}}: Check|doi=value (help); Invalid|display-authors=3(help); Unknown parameter|author-separator=ignored (help); Unknown parameter|month=ignored (help) - ^ a b Peter M; Dubuis JW; Sippell WG (1999). "Disorders of the Aldosterone Synthase and Steroid 11β-Hydroxylase Deficiencies". Hormone Research in Paediatrics (51): 211–222. doi:10.1159/000023374.
{{cite journal}}: Invalid|display-authors=3(help); Unknown parameter|author-separator=ignored (help) - ^ "CYP11B2". Retrieved 17 September 2013.
- ^ White PC (2004). "Aldosterone synthase deficiency and related disorders". Molecular and Cellular Endocrinology. 217 (1–2): 81–87. doi:10.1016. ISSN 0303-7207.
{{cite journal}}: Check|doi=value (help); Invalid|display-authors=1(help); Unknown parameter|month=ignored (help) - ^ a b c Peter M; Fawaz L; Drop SL; Visser HKA; Sippell WG (1997). "Hereditary defect in biosynthesis of aldosterone: aldosterone synthase deficiency 1964-1997". 82 (11): 3525–3528. doi:10.1210/jc.82.11.3525.
{{cite journal}}: Cite journal requires|journal=(help); Invalid|display-authors=5(help); Unknown parameter|author-separator=ignored (help); Unknown parameter|month=ignored (help)
Further reading[edit]
External links[edit]
- Aldosterone+synthase at the U.S. National Library of Medicine Medical Subject Headings (MeSH)